Desmoteplase is a novel, highly fibrin-specific "clot-busting" (thrombolytic) drug in development that reached phase III clinical trials. The Danish pharmaceutical company, Lundbeck, owns the worldwide rights to Desmoteplase. In 2009, two large trials were started to test it as a safe and effective treatment for patients with acute ischaemic stroke. After disappointing results in DIAS-3, DIAS-4 was terminated, and in December 2014 Lundbeck announced that they would stop the development of desmoteplase.

Cladribine, sold under the brand name Leustatin, among others, is a medication used to treat hairy cell leukemia and B-cell chronic lymphocytic leukemia. Cladribine, sold under the brand name Mavenclad, is used for the treatment of adults with highly active forms of relapsing-remitting multiple sclerosis.

Asparaginase is an enzyme that is used as a medication and in food manufacturing. As a medication, L-asparaginase is used to treat acute lymphoblastic leukemia (ALL) and lymphoblastic lymphoma (LBL). It is given by injection into a vein, muscle, or under the skin. A pegylated version is also available. In food manufacturing it is used to decrease acrylamide.
Iduronidase, sold as Aldurazyme, is an enzyme with the systematic name glycosaminoglycan α-L-iduronohydrolase. It catalyses the hydrolysis of unsulfated α-L-iduronosidic linkages in dermatan sulfate.

Pegaptanib sodium injection is an anti-angiogenic medicine for the treatment of neovascular (wet) age-related macular degeneration (AMD). It was discovered by NeXstar Pharmaceuticals and licensed in 2000 to EyeTech Pharmaceuticals, now OSI Pharmaceuticals, for late stage development and marketing in the United States. Gilead Sciences continues to receive royalties from the drugs licensing. Outside the US pegaptanib is marketed by Pfizer. Approval was granted by the U.S. Food and Drug Administration (FDA) in December 2004.

Laquinimod is an experimental immunomodulator developed by Active Biotech and Teva. It is being investigated as an oral treatment for multiple sclerosis (MS) and Huntington's disease.
MRI contrast agents are contrast agents used to improve the visibility of internal body structures in magnetic resonance imaging (MRI). The most commonly used compounds for contrast enhancement are gadolinium-based contrast agents (GBCAs). Such MRI contrast agents shorten the relaxation times of nuclei within body tissues following oral or intravenous administration.

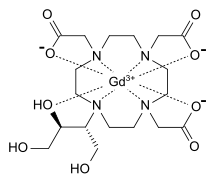
Gadoteric acid, sold under the brand name Dotarem among others, is a macrocycle-structured gadolinium-based MRI contrast agent (GBCA). It consists of the organic acid DOTA as a chelating agent, and gadolinium (Gd3+), and is used in form of the meglumine salt (gadoterate meglumine). The paramagnetic property of gadoteric acid reduces the T1 relaxation time (and to some extent the T2 and T2* relaxation times) in MRI, which is the source of its clinical utility. Because it has magnetic properties, gadoteric acid develops a magnetic moment when put under a magnetic field, which increases the signal intensity (brightness) of tissues during MRI imaging.

Secukinumab, sold under the brand name Cosentyx among others, is a human IgG1κ monoclonal antibody used for the treatment of psoriasis, ankylosing spondylitis, and psoriatic arthritis. It binds to the protein interleukin (IL)-17A and is marketed by Novartis.

Dupilumab, sold under the brand name Dupixent, is a monoclonal antibody blocking interleukin 4 and interleukin 13, used for allergic diseases such as atopic dermatitis (eczema), asthma and nasal polyps which result in chronic sinusitis. It is also used for the treatment of eosinophilic esophagitis and prurigo nodularis.
Tildrakizumab, sold under the brand names Ilumya and Ilumetri, is a monoclonal antibody designed for the treatment of immunologically mediated inflammatory disorders. It is approved for the treatment of adult patients with moderate-to-severe plaque psoriasis in the United States and the European Union.

Flutemetamol (18F) is a PET scanning radiopharmaceutical containing the radionuclide fluorine-18, used as a diagnostic tool for Alzheimer's disease.

Aripiprazole lauroxil, sold under the brand name Aristada, is a long-acting injectable atypical antipsychotic that was developed by Alkermes. It is an N-acyloxymethyl prodrug of aripiprazole that is administered via intramuscular injection once every four to eight weeks for the treatment of schizophrenia. Aripiprazole lauroxil was approved by the U.S. Food and Drug Administration (FDA) on 5 October 2015.
Risankizumab, sold under the brand name Skyrizi, is a humanized monoclonal antibody used for the treatment of plaque psoriasis, psoriatic arthritis, and Crohn's disease. It is designed to target interleukin 23A (IL-23A). It is given by subcutaneous injection.
Imipenem/cilastatin/relebactam, sold under the brand name Recarbrio, is a fixed-dose combination medication used as an antibiotic. In 2019, it was approved for use in the United States for the treatment of complicated urinary tract and complicated intra-abdominal infections. It is administered via intravenous injection.
Pertuzumab/trastuzumab/hyaluronidase, sold under the brand name Phesgo, is a fixed-dose combination medication to treat adults with HER2-positive breast cancer that has spread to other parts of the body, and for treatment of adults with early HER2-positive breast cancer. It contains pertuzumab, trastuzumab, and hyaluronidase–zzxf. It is injected under the skin via subcutaneous injection in the thigh. In the European Union, Phesgo contains the active ingredients pertuzumab and trastuzumab along with the enzyme vorhyaluronidase alfa.
Pegcetacoplan, sold under the brand name Empaveli, among others, is a medication used to treat paroxysmal nocturnal hemoglobinuria and geographic atrophy of the retina. Pegcetacoplan is a complement inhibitor.

Piflufolastat F-18, sold under the brand name Pylarify among others, is a radioactive diagnostic agent used for positron emission tomography (PET) imaging. It is given by intravenous injection.
Vutrisiran, sold under the brand name Amvuttra, is a medication used for the treatment of the polyneuropathy of hereditary transthyretin-mediated (hATTR) amyloidosis in adults. It is a double stranded small interfering RNA (siRNA) that interferes with the expression of the transthyretin (TTR) gene. Transthyretin is a serum protein made in the liver whose major function is transport of vitamin A and thyroxine. Rare mutations in the transthyretin gene result in accumulation of large amyloid deposits of misfolded transthyretin molecules most prominently in peripheral nerves and the heart. Patients with hATTR typically present with polyneuropathy or autonomic dysfunction followed by cardiomyopathy which, if untreated, is fatal within 5 to 10 years.

Gadopiclenol, sold under the brand name Elucirem among others, is a contrast agent used with magnetic resonance imaging (MRI) to detect and visualize lesions with abnormal vascularity in the central nervous system and in the body. Gadopiclenol is a paramagnetic macrocyclic non-ionic complex of gadolinium.