
The voltaic pile was the first electrical battery that could continuously provide an electric current to a circuit. It was invented by Italian physicist Alessandro Volta, who published his experiments in 1799. The voltaic pile then enabled a rapid series of other discoveries including the electrical decomposition (electrolysis) of water into oxygen and hydrogen by William Nicholson and Anthony Carlisle (1800) and the discovery or isolation of the chemical elements sodium (1807), potassium (1807), calcium (1808), boron (1808), barium (1808), strontium (1808), and magnesium (1808) by Humphry Davy.

The volt is the derived unit for electric potential, electric potential difference (voltage), and electromotive force. It is named after the Italian physicist Alessandro Volta (1745–1827).

The nickel–cadmium battery is a type of rechargeable battery using nickel oxide hydroxide and metallic cadmium as electrodes. The abbreviation Ni-Cd is derived from the chemical symbols of nickel (Ni) and cadmium (Cd): the abbreviation NiCad is a registered trademark of SAFT Corporation, although this brand name is commonly used to describe all Ni–Cd batteries.

The lead–acid battery is a type of rechargeable battery first invented in 1859 by French physicist Gaston Planté. It is the first type of rechargeable battery ever created. Compared to modern rechargeable batteries, lead–acid batteries have relatively low energy density. Despite this, their ability to supply high surge currents means that the cells have a relatively large power-to-weight ratio. These features, along with their low cost, make them attractive for use in motor vehicles to provide the high current required by starter motors.

A lemon battery is a simple battery often made for the purpose of education. Typically, a piece of zinc metal and a piece of copper are inserted into a lemon and connected by wires. Power generated by reaction of the metals is used to power a small device such as a light-emitting diode (LED).
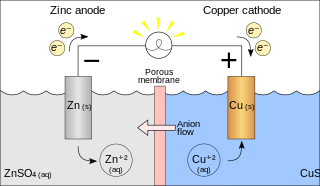
A galvanic cell or voltaic cell, named after the scientists Luigi Galvani and Alessandro Volta, respectively, is an electrochemical cell in which an electric current is generated from spontaneous Oxidation-Reduction reactions. A common apparatus generally consists of two different metals, each immersed in separate beakers containing their respective metal ions in solution that are connected by a salt bridge or separated by a porous membrane.

An alkaline battery is a type of primary battery that derives its energy from the reaction between zinc metal and manganese dioxide.

Zinc–air batteries (non-rechargeable), and zinc–air fuel cells are metal–air batteries powered by oxidizing zinc with oxygen from the air. These batteries have high energy densities and are relatively inexpensive to produce. Sizes range from very small button cells for hearing aids, larger batteries used in film cameras that previously used mercury batteries, to very large batteries used for electric vehicle propulsion and grid-scale energy storage.

The Daniell cell is a type of electrochemical cell invented in 1836 by John Frederic Daniell, a British chemist and meteorologist, and consists of a copper pot filled with a copper (II) sulfate solution, in which is immersed an unglazed earthenware container filled with sulfuric acid and a zinc electrode. He was searching for a way to eliminate the hydrogen bubble problem found in the voltaic pile, and his solution was to use a second electrolyte to consume the hydrogen produced by the first. Zinc sulfate may be substituted for the sulfuric acid. The Daniell cell was a great improvement over the existing technology used in the early days of battery development. A later variant of the Daniell cell called the gravity cell or crowfoot cell was invented in the 1860s by a Frenchman named Callaud and became a popular choice for electrical telegraphy.

A zinc–carbon battery (or carbon zinc battery in U.S. English) is a dry cell primary battery that provides direct electric current from the electrochemical reaction between zinc and manganese dioxide (MnO2) in the presence of an electrolyte. It produces a voltage of about 1.5 volts between the zinc anode, which is typically constructed as a cylindrical container for the battery cell, and a carbon rod surrounded by a compound with a higher Standard electrode potential (positive polarity), known as the cathode, that collects the current from the manganese dioxide electrode. The name "zinc-carbon" is slightly misleading as it implies that carbon is acting as the reducing agent rather than the manganese dioxide.

A mercury battery is a non-rechargeable electrochemical battery, a primary cell. Mercury batteries use a reaction between mercuric oxide and zinc electrodes in an alkaline electrolyte. The voltage during discharge remains practically constant at 1.35 volts, and the capacity is much greater than that of a similarly sized zinc-carbon battery. Mercury batteries were used in the shape of button cells for watches, hearing aids, cameras and calculators, and in larger forms for other applications.
Electrochemistry, a branch of chemistry, went through several changes during its evolution from early principles related to magnets in the early 16th and 17th centuries, to complex theories involving conductivity, electric charge and mathematical methods. The term electrochemistry was used to describe electrical phenomena in the late 19th and 20th centuries. In recent decades, electrochemistry has become an area of current research, including research in batteries and fuel cells, preventing corrosion of metals, the use of electrochemical cells to remove refractory organics and similar contaminants in wastewater electrocoagulation and improving techniques in refining chemicals with electrolysis and electrophoresis.

In the early days of electronics, vacuum tube devices were powered by batteries. Each battery had a different designation depending on which tube element it was associated with.

The Bunsen cell is a zinc-carbon primary cell composed of a zinc anode in dilute sulfuric acid separated by a porous pot from a carbon cathode in nitric or chromic acid.

A nickel–zinc battery, abbreviated NiZn, is a type of rechargeable battery similar to NiCd batteries, but with a higher voltage of 1.6 V.

The Leclanché cell is a battery invented and patented by the French scientist Georges Leclanché in 1866. The battery contained a conducting solution (electrolyte) of ammonium chloride, a cathode of carbon, a depolarizer of manganese dioxide (oxidizer), and an anode of zinc (reductant). The chemistry of this cell was later successfully adapted to manufacture a dry cell.

Batteries provided the main source of electricity before the development of electric generators and electrical grids around the end of the 19th century. Successive improvements in battery technology facilitated major electrical advances, from early scientific studies to the rise of telegraphs and telephones, eventually leading to portable computers, mobile phones, electric cars, and many other electrical devices.

An electric battery is a source of electric power consisting of one or more electrochemical cells with external connections for powering electrical devices.

The penny battery is a voltaic pile which uses various coinage as the metal disks (pennies) of a traditional voltaic pile. The coins are stacked with pieces of electrolyte soaked paper in between. The penny battery experiment is common during electrochemistry units in an educational setting.

Zinc–cerium batteries are a type of redox flow battery first developed by Plurion Inc. (UK) during the 2000s. In this rechargeable battery, both negative zinc and positive cerium electrolytes are circulated though an electrochemical flow reactor during the operation and stored in two separated reservoirs. Negative and positive electrolyte compartments in the electrochemical reactor are separated by a cation-exchange membrane, usually Nafion (DuPont). The Ce(III)/Ce(IV) and Zn(II)/Zn redox reactions take place at the positive and negative electrodes, respectively. Since zinc is electroplated during charge at the negative electrode this system is classified as a hybrid flow battery. Unlike in zinc–bromine and zinc–chlorine redox flow batteries, no condensation device is needed to dissolve halogen gases. The reagents used in the zinc-cerium system are considerably less expensive than those used in the vanadium flow battery.



















