Related Research Articles

In multicellular organisms, stem cells are undifferentiated or partially differentiated cells that can differentiate into various types of cells and proliferate indefinitely to produce more of the same stem cell. They are the earliest type of cell in a cell lineage. They are found in both embryonic and adult organisms, but they have slightly different properties in each. They are usually distinguished from progenitor cells, which cannot divide indefinitely, and precursor or blast cells, which are usually committed to differentiating into one cell type.
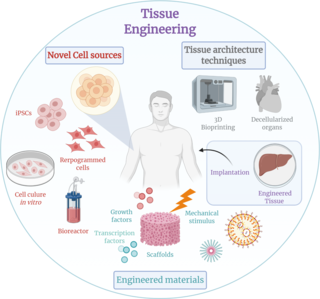
Tissue engineering is a biomedical engineering discipline that uses a combination of cells, engineering, materials methods, and suitable biochemical and physicochemical factors to restore, maintain, improve, or replace different types of biological tissues. Tissue engineering often involves the use of cells placed on tissue scaffolds in the formation of new viable tissue for a medical purpose but is not limited to applications involving cells and tissue scaffolds. While it was once categorized as a sub-field of biomaterials, having grown in scope and importance it can be considered as a field of its own.
Regenerative medicine deals with the "process of replacing, engineering or regenerating human or animal cells, tissues or organs to restore or establish normal function". This field holds the promise of engineering damaged tissues and organs by stimulating the body's own repair mechanisms to functionally heal previously irreparable tissues or organs.

Cell therapy is a therapy in which viable cells are injected, grafted or implanted into a patient in order to effectuate a medicinal effect, for example, by transplanting T-cells capable of fighting cancer cells via cell-mediated immunity in the course of immunotherapy, or grafting stem cells to regenerate diseased tissues.

Adult stem cells are undifferentiated cells, found throughout the body after development, that multiply by cell division to replenish dying cells and regenerate damaged tissues. Also known as somatic stem cells, they can be found in juvenile, adult animals, and humans, unlike embryonic stem cells.
Stem-cell therapy is the use of stem cells to treat or prevent a disease or condition. As of 2016, the only established therapy using stem cells is hematopoietic stem cell transplantation. This usually takes the form of a bone-marrow transplantation, but the cells can also be derived from umbilical cord blood. Research is underway to develop various sources for stem cells as well as to apply stem-cell treatments for neurodegenerative diseases and conditions such as diabetes and heart disease.

Mesenchyme is a type of loosely organized animal embryonic connective tissue of undifferentiated cells that give rise to most tissues, such as skin, blood or bone. The interactions between mesenchyme and epithelium help to form nearly every organ in the developing embryo.

A microcarrier is a support matrix that allows for the growth of adherent cells in bioreactors. Instead of on a flat surface, cells are cultured on the surface of spherical microcarriers so that each particle carries several hundred cells, and therefore expansion capacity can be multiplied several times over. It provides a straightforward way to scale up culture systems for industrial production of cell or protein-based therapies, or for research purposes.
Mesenchymal stem cells (MSCs) are multipotent cells found in multiple human adult tissues including bone marrow, synovial tissues, and adipose tissues. Since they are derived from the mesoderm, they have been shown to differentiate into bone, cartilage, muscle, and adipose tissue. MSCs from embryonic sources have shown promise scientifically while creating significant controversy. As a result, many researchers have focused on adult stem cells, or stem cells isolated from adult humans that can be transplanted into damaged tissue.
Amniotic stem cells are the mixture of stem cells that can be obtained from the amniotic fluid as well as the amniotic membrane. They can develop into various tissue types including skin, cartilage, cardiac tissue, nerves, muscle, and bone. The cells also have potential medical applications, especially in organ regeneration.

Mesenchymal stem cells (MSCs) also known as mesenchymal stromal cells or medicinal signaling cells are multipotent stromal cells that can differentiate into a variety of cell types, including osteoblasts, chondrocytes, myocytes and adipocytes.
Adult mesenchymal stem cells are being used by researchers in the fields of regenerative medicine and tissue engineering to artificially reconstruct human tissue which has been previously damaged. Mesenchymal stem cells are able to differentiate, or mature from a less specialized cell to a more specialized cell type, to replace damaged tissues in various organs.
Nanotopography refers to specific surface features which form or are generated at the nanoscopic scale. While the term can be used to describe a broad range of applications ranging from integrated circuits to microfluidics, in practice it typically applied to sub-micron textured surfaces as used in biomaterials research.

Regenerative endodontic procedures is defined as biologically based procedures designed to replace damaged structures such as dentin, root structures, and cells of the pulp-dentin complex. This new treatment modality aims to promote normal function of the pulp. It has become an alternative to heal apical periodontitis. Regenerative endodontics is the extension of root canal therapy. Conventional root canal therapy cleans and fills the pulp chamber with biologically inert material after destruction of the pulp due to dental caries, congenital deformity or trauma. Regenerative endodontics instead seeks to replace live tissue in the pulp chamber. The ultimate goal of regenerative endodontic procedures is to regenerate the tissues and the normal function of the dentin-pulp complex.
The in vivo bioreactor is a tissue engineering paradigm that uses bioreactor methodology to grow neotissue in vivo that augments or replaces malfunctioning native tissue. Tissue engineering principles are used to construct a confined, artificial bioreactor space in vivo that hosts a tissue scaffold and key biomolecules necessary for neotissue growth. Said space often requires inoculation with pluripotent or specific stem cells to encourage initial growth, and access to a blood source. A blood source allows for recruitment of stem cells from the body alongside nutrient delivery for continual growth. This delivery of cells and nutrients to the bioreactor eventually results in the formation of a neotissue product.
Treena Livingston Arinzeh is Professor of Biomedical Engineering at Columbia University in New York, New York, joining in 2022. She was formerly a Distinguished Professor in Biomedical Engineering at the New Jersey Institute of Technology in Newark, New Jersey. She is known for her research on adult stem-cell therapy. Arinzeh takes part in the American Chemical Society's Project Seeds program, opening up her lab for high school students from economically disadvantaged backgrounds for summer internships.
Richard Oreffo FRSB FIOR is a British–Nigerian physician and Professor of Musculoskeletal Science at the University of Southampton. His research considers skeletal biology and the fundamental mechanisms that underpin skeletal stem cell differentiation. In 2020, he launched the Cowrie Scholarship Foundation, which supports Black British students in their university studies.

Mohamadreza Baghaban Eslaminejad is the director of the Adult Stem Cell Lab and histology/embryology Professor at the Royan Institute where he held a multi-departmental professorship in bioengineering, tissue engineering, regenerative medicine, and stem cell therapy. Eslaminejad studies have been cited over 4000 times. He is best known for Hard Tissue Engineering and utilizing Mesenchymal stem cells for treatment of orthopedic diseases.
Erika Moore Taylor is a biomedical engineer, scientist, Assistant Professor, "Forbes 30 under 30 honoree," financial advisor, and the founder of a scholarship program that has been featured on CNBC.

Stem cellfat grafting is autotransplantation of adipose-derived stem cells (ADSCs) extracted from fat-abundant donor sites to other areas such as the face, breast, and hip to reconstruct the operative areas into desirable shapes. ADSCs are multipotent stem cells found in adipose tissues, displaying similar differentiation potentials to bone marrow-derived mesenchymal stem cells (BM-MSCs).
References
- ↑ "Research Institutes - Institute of Molecular, Cell and Systems Biology - All staff - Dr Matthew J Dalby". University of Glasgow. 22 October 2020. Retrieved 13 April 2021.
- ↑ McNamara, L. E.; McMurray, R. J.; Biggs, M. J. P.; Kantawong, F.; Oreffo, R. O. C.; Dalby, M. J. (2010). "Nanotopographical Control of Stem Cell Differentiation". Journal of Tissue Engineering. 1 (1): 120623. doi:10.4061/2010/120623. ISSN 2041-7314. PMC 3042612 . PMID 21350640.
- ↑ Dalby, Matthew J.; Gadegaard, Nikolaj; Tare, Rahul; Andar, Abhay; Riehle, Mathis O.; Herzyk, Pawel; Wilkinson, Chris D. W.; Oreffo, Richard O. C. (2007). "The control of human mesenchymal cell differentiation using nanoscale symmetry and disorder". Nature Materials. 6 (12): 997–1003. doi:10.1038/nmat2013. ISSN 1476-1122. PMID 17891143.
- ↑ "Professor Matthew Dalby". University of Glasgow . Retrieved 4 April 2018.
- ↑ McMurray RJ, Dalby MJ, Tsimbouri PM (May 2015). "Using biomaterials to study stem cell mechanotransduction, growth and differentiation" (PDF). Journal of Tissue Engineering and Regenerative Medicine. 9 (5): 528–39. doi: 10.1002/term.1957 . PMID 25370612. S2CID 39642567.
- ↑ "World first for dog's broken leg - BBC News" – via www.youtube.com.
- ↑ "Matthew Dalby". Google Scholar . Retrieved 1 November 2018.