Related Research Articles

RDX (abbreviation of "Research Department eXplosive") or hexogen, among other names, is an organic compound with the formula (O2N2CH2)3. It is white, odorless and tasteless, widely used as an explosive. Chemically, it is classified as a nitroamine alongside HMX, which is a more energetic explosive than TNT. It was used widely in World War II and remains common in military applications.
Nitric acid is the inorganic compound with the formula HNO3. It is a highly corrosive mineral acid. The compound is colorless, but samples tend to acquire a yellow cast over time due to decomposition into oxides of nitrogen. Most commercially available nitric acid has a concentration of 68% in water. When the solution contains more than 86% HNO3, it is referred to as fuming nitric acid. Depending on the amount of nitrogen dioxide present, fuming nitric acid is further characterized as red fuming nitric acid at concentrations above 86%, or white fuming nitric acid at concentrations above 95%.

Polysaccharides, or polycarbohydrates, are the most abundant carbohydrates found in food. They are long-chain polymeric carbohydrates composed of monosaccharide units bound together by glycosidic linkages. This carbohydrate can react with water (hydrolysis) using amylase enzymes as catalyst, which produces constituent sugars. They range in structure from linear to highly branched. Examples include storage polysaccharides such as starch, glycogen and galactogen and structural polysaccharides such as cellulose and chitin.

Starch or amylum is a polymeric carbohydrate consisting of numerous glucose units joined by glycosidic bonds. This polysaccharide is produced by most green plants for energy storage. Worldwide, it is the most common carbohydrate in human diets, and is contained in large amounts in staple foods such as wheat, potatoes, maize (corn), rice, and cassava (manioc).

Trinitrotoluene, more commonly known as TNT, more specifically 2,4,6-trinitrotoluene, and by its preferred IUPAC name 2-methyl-1,3,5-trinitrobenzene, is a chemical compound with the formula C6H2(NO2)3CH3. TNT is occasionally used as a reagent in chemical synthesis, but it is best known as an explosive material with convenient handling properties. The explosive yield of TNT is considered to be the standard comparative convention of bombs and asteroid impacts. In chemistry, TNT is used to generate charge transfer salts.
Nitrocellulose is a highly flammable compound formed by nitrating cellulose through exposure to a mixture of nitric acid and sulfuric acid. One of its first major uses was as guncotton, a replacement for gunpowder as propellant in firearms. It was also used to replace gunpowder as a low-order explosive in mining and other applications. In the form of collodion it was also a critical component in an early photographic emulsion, the use of which revolutionized photography in the 1860s.

Picric acid is an organic compound with the formula (O2N)3C6H2OH. Its IUPAC name is 2,4,6-trinitrophenol (TNP). The name "picric" comes from Greek: πικρός (pikros), meaning "bitter", due to its bitter taste. It is one of the most acidic phenols. Like other strongly nitrated organic compounds, picric acid is an explosive, which is its primary use. It has also been used as medicine (antiseptic, burn treatments) and as a dye.
Ammonium nitrate is a chemical compound with the formula NH4NO3. It is a white crystalline salt consisting of ions of ammonium and nitrate. It is highly soluble in water and hygroscopic as a solid, although it does not form hydrates. It is predominantly used in agriculture as a high-nitrogen fertilizer.

Mochi, is a Japanese rice cake made of mochigome (もち米), a short-grain japonica glutinous rice, and sometimes other ingredients such as water, sugar, and cornstarch. The steamed rice is pounded into paste and molded into the desired shape. In Japan, it is traditionally made in a ceremony called mochitsuki. While eaten year-round, mochi is a traditional food for the Japanese New Year, and is commonly sold and eaten during that time.

Lead(II) nitrate is an inorganic compound with the chemical formula Pb(NO3)2. It commonly occurs as a colourless crystal or white powder and, unlike most other lead(II) salts, is soluble in water.

Dextrins are a group of low-molecular-weight carbohydrates produced by the hydrolysis of starch and glycogen. Dextrins are mixtures of polymers of D-glucose units linked by α-(1→4) or α-(1→6) glycosidic bonds.

Amylose is a polysaccharide made of α-D-glucose units, bonded to each other through α(1→4) glycosidic bonds. It is one of the two components of starch, making up approximately 20–30%. Because of its tightly packed helical structure, amylose is more resistant to digestion than other starch molecules and is therefore an important form of resistant starch.

Amylopectin is a water-insoluble polysaccharide and highly branched polymer of α-glucose units found in plants. It is one of the two components of starch, the other being amylose.

Barium nitrate is the inorganic compound with the chemical formula Ba(NO3)2. It, like most barium salts, is colorless, toxic, and water-soluble. It burns with a green flame and is an oxidizer; the compound is commonly used in pyrotechnics.
Starch gelatinization is a process of breaking down of intermolecular bonds of starch molecules in the presence of water and heat, allowing the hydrogen bonding sites to engage more water. This irreversibly dissolves the starch granule in water. Water acts as a plasticizer.

Resistant starch (RS) is starch, including its degradation products, that escapes from digestion in the small intestine of healthy individuals. Resistant starch occurs naturally in foods, but it can also be added as part of dried raw foods, or used as an additive in manufactured foods.

Waxy corn or glutinous corn is a type of field corn characterized by its sticky texture when cooked as a result of larger amounts of amylopectin. The corn was first described from a specimen from China in 1909. As this plant showed many peculiar traits, the American breeders long used it as a genetic marker to tag the existence of hidden genes in other maize breeding programs. In 1922 a researcher found that the endosperm of waxy maize contained only amylopectin and no amylose starch molecule in opposition to normal dent corn varieties that contain both. Until World War II, the main source of starch in the United States was tapioca, but when Japan severed the supply lines of the U.S., they forced processors to turn to waxy maize. Amylopectin or waxy starch is now used mainly in food products, but also in the textile, adhesive, corrugating and paper industry.
Retrogradation is a reaction that takes place when the amylose and amylopectin chains in cooked, gelatinized starch realign themselves as the cooked starch cools.

Amflora is a genetically modified potato cultivar developed by BASF Plant Science. "Amflora" potato plants produce pure amylopectin starch that is processed to waxy potato starch. It was approved for industrial applications in the European Union on 2 March 2010 by the European Commission. In January 2012, the potato was withdrawn from the market in the EU.

Floridean starch is a type of a storage glucan found in glaucophytes and in red algae, in which it is usually the primary sink for fixed carbon from photosynthesis. It is found in grains or granules in the cell's cytoplasm and is composed of an α-linked glucose polymer with a degree of branching intermediate between amylopectin and glycogen, though more similar to the former. The polymers that make up floridean starch are sometimes referred to as "semi-amylopectin".
References
- ↑ Robert Turkington (26 October 2009). Chemicals Used for Illegal Purposes. John Wiley & Sons. pp. 295–. ISBN 978-0-470-18780-7.
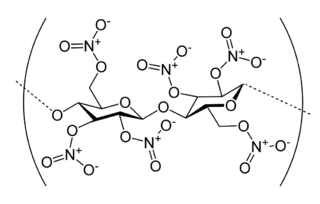
- 1 2 3 4 Liu, Jiping (2019), "Nitrostarch", Nitrate Esters Chemistry and Technology, Singapore: Springer Singapore, pp. 581–598, doi:10.1007/978-981-13-6647-5_11, ISBN 978-981-13-6645-1, S2CID 239210636 , retrieved 2022-02-25
- ↑ John Pichtel (15 September 2016). Terrorism and WMDs: Awareness and Response, Second Edition. Taylor & Francis. pp. 207–. ISBN 978-1-4987-3901-6.
- ↑ Zakaria Abdel-rahman Shalash (1955). Studies on nitration of starch stabilization and explosive prop...