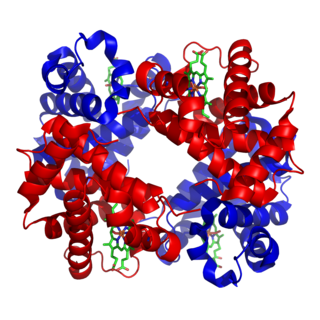
Hemoglobin is a protein containing iron that facilitates the transport of oxygen in red blood cells. Almost all vertebrates contain hemoglobin, with the exception of the fish family Channichthyidae and the tissues of some invertebrate animals. Hemoglobin in the blood carries oxygen from the respiratory organs to the other tissues of the body, where it releases the oxygen to enable aerobic respiration which powers the animal's metabolism. A healthy human has 12 to 20 grams of hemoglobin in every 100 mL of blood. Hemoglobin is a metalloprotein, a chromoprotein, and globulin.

Hemoglobinopathy is the medical term for a group of inherited blood disorders and diseases that primarily affect red blood cells. They are single-gene disorders and, in most cases, they are inherited as autosomal co-dominant traits.
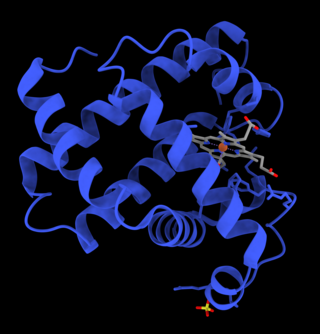
Myoglobin is an iron- and oxygen-binding protein found in the cardiac and skeletal muscle tissue of vertebrates in general and in almost all mammals. Myoglobin is distantly related to hemoglobin. Compared to hemoglobin, myoglobin has a higher affinity for oxygen and does not have cooperative binding with oxygen like hemoglobin does. Myoglobin consists of non-polar amino acids at the core of the globulin, where the heme group is non-covalently bounded with the surrounding polypeptide of myoglobin. In humans, myoglobin is only found in the bloodstream after muscle injury.

A hemeprotein, or heme protein, is a protein that contains a heme prosthetic group. They are a very large class of metalloproteins. The heme group confers functionality, which can include oxygen carrying, oxygen reduction, electron transfer, and other processes. Heme is bound to the protein either covalently or noncovalently or both.

Leghemoglobin is an oxygen-carrying phytoglobin found in the nitrogen-fixing root nodules of leguminous plants. It is produced by these plants in response to the roots being colonized by nitrogen-fixing bacteria, termed rhizobia, as part of the symbiotic interaction between plant and bacterium: roots not colonized by Rhizobium do not synthesise leghemoglobin. Leghemoglobin has close chemical and structural similarities to hemoglobin, and, like hemoglobin, is red in colour. It was originally thought that the heme prosthetic group for plant leghemoglobin was provided by the bacterial symbiont within symbiotic root nodules. However, subsequent work shows that the plant host strongly expresses heme biosynthesis genes within nodules, and that activation of those genes correlates with leghemoglobin gene expression in developing nodules.

Heme, or haem, is a precursor to hemoglobin, which is necessary to bind oxygen in the bloodstream. Heme is biosynthesized in both the bone marrow and the liver.
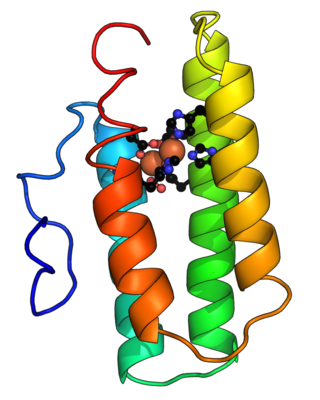
Hemerythrin (also spelled haemerythrin; Ancient Greek: αἷμα, romanized: haîma, lit. 'blood', Ancient Greek: ἐρυθρός, romanized: erythrós, lit. 'red') is an oligomeric protein responsible for oxygen (O2) transport in the marine invertebrate phyla of sipunculids, priapulids, brachiopods, and in a single annelid worm genus, Magelona. Myohemerythrin is a monomeric O2-binding protein found in the muscles of marine invertebrates. Hemerythrin and myohemerythrin are essentially colorless when deoxygenated, but turn a violet-pink in the oxygenated state.

The globins are a superfamily of heme-containing globular proteins, involved in binding and/or transporting oxygen. These proteins all incorporate the globin fold, a series of eight alpha helical segments. Two prominent members include myoglobin and hemoglobin. Both of these proteins reversibly bind oxygen via a heme prosthetic group. They are widely distributed in many organisms.
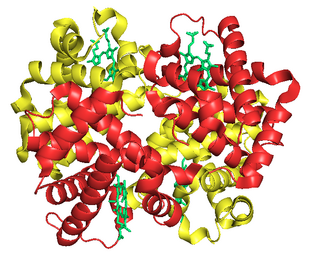
Fetal hemoglobin, or foetal haemoglobin is the main oxygen carrier protein in the human fetus. Hemoglobin F is found in fetal red blood cells, and is involved in transporting oxygen from the mother's bloodstream to organs and tissues in the fetus. It is produced at around 6 weeks of pregnancy and the levels remain high after birth until the baby is roughly 2–4 months old. Hemoglobin F has a different composition than adult forms of hemoglobin, allowing it to bind oxygen more strongly; this in turn enables the developing fetus to retrieve oxygen from the mother's bloodstream, which occurs through the placenta found in the mother's uterus.
Carboxyhemoglobin is a stable complex of carbon monoxide and hemoglobin (Hb) that forms in red blood cells upon contact with carbon monoxide. Carboxyhemoglobin is often mistaken for the compound formed by the combination of carbon dioxide (carboxyl) and hemoglobin, which is actually carbaminohemoglobin. Carboxyhemoglobin terminology emerged when carbon monoxide was known by its historic name, "carbonic oxide", and evolved through Germanic and British English etymological influences; the preferred IUPAC nomenclature is carbonylhemoglobin.
A respiratory pigment is a metalloprotein that serves a variety of important functions, its main being O2 transport. Other functions performed include O2 storage, CO2 transport, and transportation of substances other than respiratory gases. There are four major classifications of respiratory pigment: hemoglobin, hemocyanin, erythrocruorin–chlorocruorin, and hemerythrin. The heme-containing globin is the most commonly-occurring respiratory pigment, occurring in at least 9 different phyla of animals.

Hemoglobin A (HbA), also known as adult hemoglobin, hemoglobin A1 or α2β2, is the most common human hemoglobin tetramer, accounting for over 97% of the total red blood cell hemoglobin. Hemoglobin is an oxygen-binding protein, found in erythrocytes, which transports oxygen from the lungs to the tissues. Hemoglobin A is the most common adult form of hemoglobin and exists as a tetramer containing two alpha subunits and two beta subunits (α2β2). Hemoglobin A2 (HbA2) is a less common adult form of hemoglobin and is composed of two alpha and two delta-globin subunits. This hemoglobin makes up 1-3% of hemoglobin in adults.

Heme oxygenase, or haem oxygenase, is an enzyme that catalyzes the degradation of heme to produce biliverdin, ferrous ion, and carbon monoxide.
The human β-globin locus is composed of five genes located on a short region of chromosome 11, responsible for the creation of the beta parts of the oxygen transport protein Haemoglobin. This locus contains not only the beta globin gene but also delta, gamma-A, gamma-G, and epsilon globin. Expression of all of these genes is controlled by single locus control region (LCR), and the genes are differentially expressed throughout development.
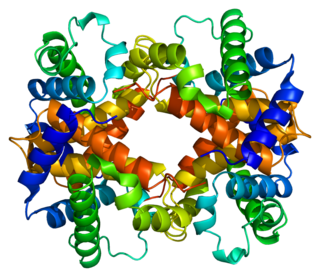
Hemoglobin subunit beta is a globin protein, coded for by the HBB gene, which along with alpha globin (HBA), makes up the most common form of haemoglobin in adult humans, hemoglobin A (HbA). It is 147 amino acids long and has a molecular weight of 15,867 Da. Normal adult human HbA is a heterotetramer consisting of two alpha chains and two beta chains.

Erythrocruorin, and the similar chlorocruorin, are large oxygen-carrying hemeprotein complexes, which have a molecular mass greater than 3.5 million daltons. Both are sometimes called giant hemoglobin or hexagonal bilayer haemoglobin. They are found in many annelids and arthropods.

Nitric oxide dioxygenase (EC 1.14.12.17) is an enzyme that catalyzes the conversion of nitric oxide (NO) to nitrate (NO−
3) . The net reaction for the reaction catalyzed by nitric oxide dioxygenase is shown below:
In enzymology, a leghemoglobin reductase (EC 1.6.2.6) is an enzyme that catalyzes the chemical reaction

Cytoglobin is the protein product of CYGB, a human and mammalian gene.
Vitreoscilla haemoglobin (VHb) is a type of haemoglobin found in the Gram-negative aerobic bacterium, Vitreoscilla. It is the first haemoglobin discovered from bacteria, but unlike classic hemoglobin it is composed only of a single globin molecule.















