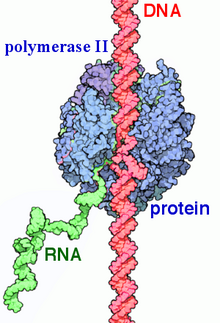
In biochemistry, a polymerase is an enzyme that synthesizes long chains of polymers or nucleic acids. DNA polymerase and RNA polymerase are used to assemble DNA and RNA molecules, respectively, by copying a DNA template strand using base-pairing interactions or RNA by half ladder replication.

Transcription is the process of copying a segment of DNA into RNA. The segments of DNA transcribed into RNA molecules that can encode proteins produce messenger RNA (mRNA). Other segments of DNA are transcribed into RNA molecules called non-coding RNAs (ncRNAs).

In molecular biology, RNA polymerase, or more specifically DNA-directed/dependent RNA polymerase (DdRP), is an enzyme that catalyzes the chemical reactions that synthesize RNA from a DNA template.
In genetics, a transcription terminator is a section of nucleic acid sequence that marks the end of a gene or operon in genomic DNA during transcription. This sequence mediates transcriptional termination by providing signals in the newly synthesized transcript RNA that trigger processes which release the transcript RNA from the transcriptional complex. These processes include the direct interaction of the mRNA secondary structure with the complex and/or the indirect activities of recruited termination factors. Release of the transcriptional complex frees RNA polymerase and related transcriptional machinery to begin transcription of new mRNAs.

The preinitiation complex is a complex of approximately 100 proteins that is necessary for the transcription of protein-coding genes in eukaryotes and archaea. The preinitiation complex positions RNA polymerase II at gene transcription start sites, denatures the DNA, and positions the DNA in the RNA polymerase II active site for transcription.
RNA polymerase 1 is, in higher eukaryotes, the polymerase that only transcribes ribosomal RNA, a type of RNA that accounts for over 50% of the total RNA synthesized in a cell.

General transcription factors (GTFs), also known as basal transcriptional factors, are a class of protein transcription factors that bind to specific sites (promoter) on DNA to activate transcription of genetic information from DNA to messenger RNA. GTFs, RNA polymerase, and the mediator constitute the basic transcriptional apparatus that first bind to the promoter, then start transcription. GTFs are also intimately involved in the process of gene regulation, and most are required for life.

A capping enzyme (CE) is an enzyme that catalyzes the attachment of the 5' cap to messenger RNA molecules that are in the process of being synthesized in the cell nucleus during the first stages of gene expression. The addition of the cap occurs co-transcriptionally, after the growing RNA molecule contains as little as 25 nucleotides. The enzymatic reaction is catalyzed specifically by the phosphorylated carboxyl-terminal domain (CTD) of RNA polymerase II. The 5' cap is therefore specific to RNAs synthesized by this polymerase rather than those synthesized by RNA polymerase I or RNA polymerase III. Pre-mRNA undergoes a series of modifications - 5' capping, splicing and 3' polyadenylation before becoming mature mRNA that exits the nucleus to be translated into functional proteins and capping of the 5' end is the first of these modifications. Three enzymes, RNA triphosphatase, guanylyltransferase, and methyltransferase are involved in the addition of the methylated 5' cap to the mRNA.
Cleavage and polyadenylation specificity factor (CPSF) is involved in the cleavage of the 3' signaling region from a newly synthesized pre-messenger RNA (pre-mRNA) molecule in the process of gene transcription. In eukaryotes, messenger RNA precursors (pre-mRNA) are transcribed in the nucleus from DNA by the enzyme, RNA polymerase II. The pre-mRNA must undergo post-transcriptional modifications, forming mature RNA (mRNA), before they can be transported into the cytoplasm for translation into proteins. The post-transcriptional modifications are: the addition of a 5' m7G cap, splicing of intronic sequences, and 3' cleavage and polyadenylation.

Eukaryotic transcription is the elaborate process that eukaryotic cells use to copy genetic information stored in DNA into units of transportable complementary RNA replica. Gene transcription occurs in both eukaryotic and prokaryotic cells. Unlike prokaryotic RNA polymerase that initiates the transcription of all different types of RNA, RNA polymerase in eukaryotes comes in three variations, each translating a different type of gene. A eukaryotic cell has a nucleus that separates the processes of transcription and translation. Eukaryotic transcription occurs within the nucleus where DNA is packaged into nucleosomes and higher order chromatin structures. The complexity of the eukaryotic genome necessitates a great variety and complexity of gene expression control.
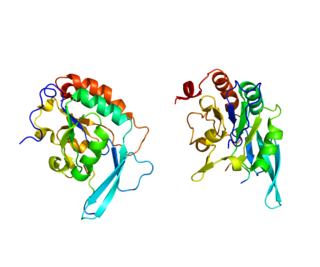
DNA-directed RNA polymerase II subunit RPB1, also known as RPB1, is an enzyme that is encoded by the POLR2A gene in humans.

DNA-directed RNA polymerase II subunit RPB4 is an enzyme that in humans is encoded by the POLR2D gene.

General transcription factor IIH subunit 1 is a protein that in humans is encoded by the GTF2H1 gene.
RNA polymerase II holoenzyme is a form of eukaryotic RNA polymerase II that is recruited to the promoters of protein-coding genes in living cells. It consists of RNA polymerase II, a subset of general transcription factors, and regulatory proteins known as SRB proteins.

DSIF is a protein complex that can either negatively or positively affect transcription by RNA polymerase II. It can interact with the negative elongation factor (NELF) to promote the stalling of Pol II at some genes, which is called promoter proximal pausing. The pause occurs soon after initiation, once 20-60 nucleotides have been transcribed. This stalling is relieved by positive transcription elongation factor b (P-TEFb) and Pol II enters productive elongation to resume synthesis till finish. In humans, DSIF is composed of hSPT4 and hSPT5. hSPT5 has a direct role in mRNA capping which occurs while the elongation is paused.
RNA polymerase IV is an enzyme that synthesizes small interfering RNA (siRNA) in plants, which silence gene expression. RNAP IV belongs to a family of enzymes that catalyze the process of transcription known as RNA Polymerases, which synthesize RNA from DNA templates. Discovered via phylogenetic studies of land plants, genes of RNAP IV are thought to have resulted from multistep evolution processes that occurred in RNA Polymerase II phylogenies. Such an evolutionary pathway is supported by the fact that RNAP IV is composed of 12 protein subunits that are either similar or identical to RNA polymerase II, and is specific to plant genomes. Via its synthesis of siRNA, RNAP IV is involved in regulation of heterochromatin formation in a process known as RNA directed DNA Methylation (RdDM).
Cryptic unstable transcripts (CUTs) are a subset of non-coding RNAs (ncRNAs) that are produced from intergenic and intragenic regions. CUTs were first observed in S. cerevisiae yeast models and are found in most eukaryotes. Some basic characteristics of CUTs include a length of around 200–800 base pairs, a 5' cap, poly-adenylated tail, and rapid degradation due to the combined activity of poly-adenylating polymerases and exosome complexes. CUT transcription occurs through RNA Polymerase II and initiates from nucleosome-depleted regions, often in an antisense orientation. To date, CUTs have a relatively uncharacterized function but have been implicated in a number of putative gene regulation and silencing pathways. Thousands of loci leading to the generation of CUTs have been described in the yeast genome. Additionally, stable uncharacterized transcripts, or SUTs, have also been detected in cells and bear many similarities to CUTs but are not degraded through the same pathways.
RNA polymerase V, previously known as RNA polymerase IVb, is a multisubunit plant specific RNA polymerase. It is required for normal function and biogenesis of small interfering RNA (siRNA). Together with RNA polymerase IV, Pol V is involved in an siRNA-dependent epigenetic pathway known as RNA-directed DNA methylation (RdDM), which establishes and maintains heterochromatic silencing in plants.
In epigenetics, proline isomerization is the effect that cis-trans isomerization of the amino acid proline has on the regulation of gene expression. Similar to aspartic acid, the amino acid proline has the rare property of being able to occupy both cis and trans isomers of its prolyl peptide bonds with ease. Peptidyl-prolyl isomerase, or PPIase, is an enzyme very commonly associated with proline isomerization due to their ability to catalyze the isomerization of prolines. PPIases are present in three types: cyclophilins, FK507-binding proteins, and the parvulins. PPIase enzymes catalyze the transition of proline between cis and trans isomers and are essential to the numerous biological functions controlled and affected by prolyl isomerization Without PPIases, prolyl peptide bonds will slowly switch between cis and trans isomers, a process that can lock proteins in a nonnative structure that can affect render the protein temporarily ineffective. Although this switch can occur on its own, PPIases are responsible for most isomerization of prolyl peptide bonds. The specific amino acid that precedes the prolyl peptide bond also can have an effect on which conformation the bond assumes. For instance, when an aromatic amino acid is bonded to a proline the bond is more favorable to the cis conformation. Cyclophilin A uses an "electrostatic handle" to pull proline into cis and trans formations. Most of these biological functions are affected by the isomerization of proline when one isomer interacts differently than the other, commonly causing an activation/deactivation relationship. As an amino acid, proline is present in many proteins. This aids in the multitude of effects that isomerization of proline can have in different biological mechanisms and functions.

Archaeal transcription is the process in which a segment of archaeal DNA is copied into a newly synthesized strand of RNA using the sole Pol II-like RNA polymerase (RNAP). The process occurs in three main steps: initiation, elongation, and termination; and the end result is a strand of RNA that is complementary to a single strand of DNA. A number of transcription factors govern this process with homologs in both bacteria and eukaryotes, with the core machinery more similar to eukaryotic transcription.