
In genetics, a mutagen is a physical or chemical agent that permanently changes genetic material, usually DNA, in an organism and thus increases the frequency of mutations above the natural background level. As many mutations can cause cancer in animals, such mutagens can therefore be carcinogens, although not all necessarily are. All mutagens have characteristic mutational signatures with some chemicals becoming mutagenic through cellular processes.
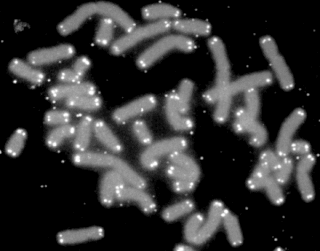
A telomere is a region of repetitive nucleotide sequences associated with specialized proteins at the ends of linear chromosomes. Telomeres are a widespread genetic feature most commonly found in eukaryotes. In most, if not all species possessing them, they protect the terminal regions of chromosomal DNA from progressive degradation and ensure the integrity of linear chromosomes by preventing DNA repair systems from mistaking the very ends of the DNA strand for a double-strand break.

Telomerase, also called terminal transferase, is a ribonucleoprotein that adds a species-dependent telomere repeat sequence to the 3' end of telomeres. A telomere is a region of repetitive sequences at each end of the chromosomes of most eukaryotes. Telomeres protect the end of the chromosome from DNA damage or from fusion with neighbouring chromosomes. The fruit fly Drosophila melanogaster lacks telomerase, but instead uses retrotransposons to maintain telomeres.
Genotoxicity is the property of chemical agents that damage the genetic information within a cell causing mutations, which may lead to cancer. While genotoxicity is often confused with mutagenicity, all mutagens are genotoxic, but some genotoxic substances are not mutagenic. The alteration can have direct or indirect effects on the DNA: the induction of mutations, mistimed event activation, and direct DNA damage leading to mutations. The permanent, heritable changes can affect either somatic cells of the organism or germ cells to be passed on to future generations. Cells prevent expression of the genotoxic mutation by either DNA repair or apoptosis; however, the damage may not always be fixed leading to mutagenesis.

Radiation hormesis is the hypothesis that low doses of ionizing radiation are beneficial, stimulating the activation of repair mechanisms that protect against disease, that are not activated in absence of ionizing radiation. The reserve repair mechanisms are hypothesized to be sufficiently effective when stimulated as to not only cancel the detrimental effects of ionizing radiation but also inhibit disease not related to radiation exposure. It has been a mainstream concept since at least 2009.

A clastogen is a mutagenic agent that disturbs normal DNA related processes or directly causes DNA strand breakages, thus causing the deletion, insertion, or rearrangement of entire chromosome sections. These processes are a form of mutagenesis which if left unrepaired, or improperly repaired, can lead to cancer. Known clastogens include acridine yellow, benzene, ethylene oxide, arsenic, phosphine, mimosine, actinomycin D, camptothecin, methotrexate, methyl acrylate, resorcinol and 5-fluorodeoxyuridine. Additionally, 1,2-dimethylhydrazine is a known colon carcinogen and shows signs of possessing clastogenic activity. There are many clastogens not listed here and research is ongoing to discover new clastogens. Some known clastogens only exhibit clastogenic activity in certain cell types, such as caffeine which exhibits clastogenic activity in plant cells. Researchers are interested in clastogens for researching cancer, as well as for other human health concerns such as the inheritability of clastogen effected paternal germ cells that lead to fetus developmental defects.

DNA repair protein XRCC4 also known as X-ray repair cross-complementing protein 4 or XRCC4 is a protein that in humans is encoded by the XRCC4 gene. In addition to humans, the XRCC4 protein is also expressed in many other metazoans, fungi and in plants. The X-ray repair cross-complementing protein 4 is one of several core proteins involved in the non-homologous end joining (NHEJ) pathway to repair DNA double strand breaks (DSBs).

Nibrin, also known as NBN or NBS1, is a protein which in humans is encoded by the NBN gene.
Particle therapy is a form of external beam radiotherapy using beams of energetic neutrons, protons, or other heavier positive ions for cancer treatment. The most common type of particle therapy as of August 2021 is proton therapy.
Health threats from cosmic rays are the dangers posed by cosmic rays to astronauts on interplanetary missions or any missions that venture through the Van-Allen Belts or outside the Earth's magnetosphere. They are one of the greatest barriers standing in the way of plans for interplanetary travel by crewed spacecraft, but space radiation health risks also occur for missions in low Earth orbit such as the International Space Station (ISS).
In radiobiology, the relative biological effectiveness is the ratio of biological effectiveness of one type of ionizing radiation relative to another, given the same amount of absorbed energy. The RBE is an empirical value that varies depending on the type of ionizing radiation, the energies involved, the biological effects being considered such as cell death, and the oxygen tension of the tissues or so-called oxygen effect.
The MRN complex is a protein complex consisting of Mre11, Rad50 and Nbs1. In eukaryotes, the MRN/X complex plays an important role in the initial processing of double-strand DNA breaks prior to repair by homologous recombination or non-homologous end joining. The MRN complex binds avidly to double-strand breaks both in vitro and in vivo and may serve to tether broken ends prior to repair by non-homologous end joining or to initiate DNA end resection prior to repair by homologous recombination. The MRN complex also participates in activating the checkpoint kinase ATM in response to DNA damage. Production of short single-strand oligonucleotides by Mre11 endonuclease activity has been implicated in ATM activation by the MRN complex.
Telomere-binding proteins function to bind telomeric DNA in various species. In particular, telomere-binding protein refers to TTAGGG repeat binding factor-1 (TERF1) and TTAGGG repeat binding factor-2 (TERF2). Telomere sequences in humans are composed of TTAGGG sequences which provide protection and replication of chromosome ends to prevent degradation. Telomere-binding proteins can generate a T-loop to protect chromosome ends. TRFs are double-stranded proteins which are known to induce bending, looping, and pairing of DNA which aids in the formation of T-loops. They directly bind to TTAGGG repeat sequence in the DNA. There are also subtelomeric regions present for regulation. However, in humans, there are six subunits forming a complex known as shelterin.

Structural maintenance of chromosomes protein 1B (SMC-1B) is a protein that in humans is encoded by the SMC1B gene. SMC proteins engage in chromosome organization and can be broken into 3 groups based on function which are cohesins, condensins, and DNA repair.SMC-1B belongs to a family of proteins required for chromatid cohesion and DNA recombination during meiosis and mitosis. SMC1ß protein appears to participate with other cohesins REC8, STAG3 and SMC3 in sister-chromatid cohesion throughout the whole meiotic process in human oocytes.

Astronauts are exposed to approximately 50-2,000 millisieverts (mSv) while on six-month-duration missions to the International Space Station (ISS), the Moon and beyond. The risk of cancer caused by ionizing radiation is well documented at radiation doses beginning at 100mSv and above.
HZE ions are the high-energy nuclei component of galactic cosmic rays (GCRs) which have an electric charge of +3 e or greater – that is, they must be the nuclei of elements heavier than hydrogen or helium.
Chromosomal instability (CIN) is a type of genomic instability in which chromosomes are unstable, such that either whole chromosomes or parts of chromosomes are duplicated or deleted. More specifically, CIN refers to the increase in rate of addition or loss of entire chromosomes or sections of them. The unequal distribution of DNA to daughter cells upon mitosis results in a failure to maintain euploidy leading to aneuploidy. In other words, the daughter cells do not have the same number of chromosomes as the cell they originated from. Chromosomal instability is the most common form of genetic instability and cause of aneuploidy.
Travel outside the Earth's protective atmosphere, magnetosphere, and in free fall can harm human health, and understanding such harm is essential for successful crewed spaceflight. Potential effects on the central nervous system (CNS) are particularly important. A vigorous ground-based cellular and animal model research program will help quantify the risk to the CNS from space radiation exposure on future long distance space missions and promote the development of optimized countermeasures.
Telomeres, the caps on the ends of eukaryotic chromosomes, play critical roles in cellular aging and cancer. An important facet to how telomeres function in these roles is their involvement in cell cycle regulation.

Jan Karlseder is an Austrian molecular biologist, a professor in the Molecular and Cellular Biology Laboratory, the Director of the Paul F. Glenn Center for Biology of Aging Research and the holder of the Donald and Darlene Shiley Chair at the Salk Institute for Biological Studies.










