Arsenic is a chemical element; it has symbol As and atomic number 33. Arsenic occurs in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. Arsenic is a metalloid. It has various allotropes, but only the grey form, which has a metallic appearance, is important to industry.

Sodium cyanide is a poisonous compound with the formula NaCN. It is a white, water-soluble solid. Cyanide has a high affinity for metals, which leads to the high toxicity of this salt. Its main application, in gold mining, also exploits its high reactivity toward metals. It is a moderately strong base.

Arsenic poisoning is a medical condition that occurs due to elevated levels of arsenic in the body. If arsenic poisoning occurs over a brief period of time, symptoms may include vomiting, abdominal pain, encephalopathy, and watery diarrhea that contains blood. Long-term exposure can result in thickening of the skin, darker skin, abdominal pain, diarrhea, heart disease, numbness, and cancer.

Arsine (IUPAC name: arsane) is an inorganic compound with the formula AsH3. This flammable, pyrophoric, and highly toxic pnictogen hydride gas is one of the simplest compounds of arsenic. Despite its lethality, it finds some applications in the semiconductor industry and for the synthesis of organoarsenic compounds. The term arsine is commonly used to describe a class of organoarsenic compounds of the formula AsH3−xRx, where R = aryl or alkyl. For example, As(C6H5)3, called triphenylarsine, is referred to as "an arsine".

Neurotoxins are toxins that are destructive to nerve tissue. Neurotoxins are an extensive class of exogenous chemical neurological insults that can adversely affect function in both developing and mature nervous tissue. The term can also be used to classify endogenous compounds, which, when abnormally contacted, can prove neurologically toxic. Though neurotoxins are often neurologically destructive, their ability to specifically target neural components is important in the study of nervous systems. Common examples of neurotoxins include lead, ethanol, glutamate, nitric oxide, botulinum toxin, tetanus toxin, and tetrodotoxin. Some substances such as nitric oxide and glutamate are in fact essential for proper function of the body and only exert neurotoxic effects at excessive concentrations.
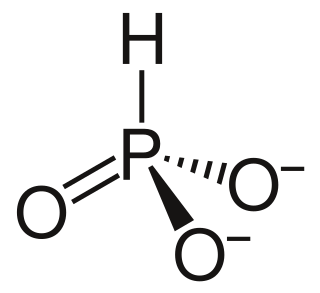
A phosphite anion or phosphite in inorganic chemistry usually refers to [HPO3]2− but includes [H2PO3]− ([HPO2(OH)]−). These anions are the conjugate bases of phosphorous acid (H3PO3). The corresponding salts, e.g. sodium phosphite (Na2HPO3) are reducing in character.

Arsenic trisulfide is the inorganic compound with the formula As2S3. It is a dark yellow solid that is insoluble in water. It also occurs as the mineral orpiment, which has been used as a pigment called King's yellow. It is produced in the analysis of arsenic compounds. It is a group V/VI, intrinsic p-type semiconductor and exhibits photo-induced phase-change properties.
Vanadium(V) oxide (vanadia) is the inorganic compound with the formula V2O5. Commonly known as vanadium pentoxide, it is a brown/yellow solid, although when freshly precipitated from aqueous solution, its colour is deep orange. Because of its high oxidation state, it is both an amphoteric oxide and an oxidizing agent. From the industrial perspective, it is the most important compound of vanadium, being the principal precursor to alloys of vanadium and is a widely used industrial catalyst.

Arsenic trioxide, sold under the brand name Trisenox among others, is an inorganic compound with the formula As
2O
3. As an industrial chemical, its major uses include the manufacture of wood preservatives, pesticides, and glass. It is also used as a medication to treat a type of cancer known as acute promyelocytic leukemia. For this use it is given by injection into a vein.

Antimony(III) oxide is the inorganic compound with the formula Sb2O3. It is the most important commercial compound of antimony. It is found in nature as the minerals valentinite and senarmontite. Like most polymeric oxides, Sb2O3 dissolves in aqueous solutions with hydrolysis. A mixed arsenic-antimony oxide occurs in nature as the very rare mineral stibioclaudetite.
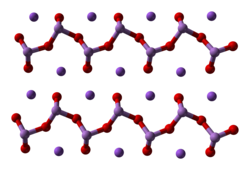
The arsenate is an ion with the chemical formula AsO3−4. Bonding in arsenate consists of a central arsenic atom, with oxidation state +5, double bonded to one oxygen atom and single bonded to a further three oxygen atoms. The four oxygen atoms orient around the arsenic atom in a tetrahedral geometry. Resonance disperses the ion's −3 charge across all four oxygen atoms.

Arsenous acid (or arsenious acid) is the inorganic compound with the formula H3AsO3. It is known to occur in aqueous solutions, but it has not been isolated as a pure material, although this fact does not detract from the significance of As(OH)3.
In chemistry, an arsenite is a chemical compound containing an arsenic oxyanion where arsenic has oxidation state +3. Note that in fields that commonly deal with groundwater chemistry, arsenite is used generically to identify soluble AsIII anions. IUPAC have recommended that arsenite compounds are to be named as arsenate(III), for example ortho-arsenite is called trioxidoarsenate(III). Ortho-arsenite contrasts to the corresponding anions of the lighter members of group 15, phosphite which has the structure HPO2−3 and nitrite, NO−2 which is bent.

Sodium molybdate, Na2MoO4, is useful as a source of molybdenum. This white, crystalline salt is often encountered as the dihydrate, Na2MoO4·2H2O.

Arsenic pentoxide is the inorganic compound with the formula As2O5. This glassy, white, deliquescent solid is relatively unstable, consistent with the rarity of the As(V) oxidation state. More common, and far more important commercially, is arsenic(III) oxide (As2O3). All inorganic arsenic compounds are highly toxic and thus find only limited commercial applications.

Dibromomethane or methylene bromide, or methylene dibromide is a halomethane with the formula CH2Br2. It is slightly soluble in water but very soluble in organic solvents. It is a colorless liquid.
Potassium arsenite (KAsO2) is an inorganic compound that exists in two forms, potassium meta-arsenite (KAsO2) and potassium ortho-arsenite (K3AsO3). It is composed of arsenite ions (AsO33− or AsO2−) with arsenic always existing in the +3 oxidation state. Like many other arsenic containing compounds, potassium arsenite is highly toxic and carcinogenic to humans. Potassium arsenite forms the basis of Fowler’s solution, which was historically used as a medicinal tonic, but due to its toxic nature its use was discontinued. Potassium arsenite is still, however, used as a rodenticide.

Methyldichloroarsine, sometimes abbreviated "MD" and also known as methyl Dick, is an organoarsenic compound with the formula CH3AsCl2. This colourless volatile liquid is a highly toxic vesicant that has been used in chemical warfare.
Arsenic biochemistry refers to biochemical processes that can use arsenic or its compounds, such as arsenate. Arsenic is a moderately abundant element in Earth's crust, and although many arsenic compounds are often considered highly toxic to most life, a wide variety of organoarsenic compounds are produced biologically and various organic and inorganic arsenic compounds are metabolized by numerous organisms. This pattern is general for other related elements, including selenium, which can exhibit both beneficial and deleterious effects. Arsenic biochemistry has become topical since many toxic arsenic compounds are found in some aquifers, potentially affecting many millions of people via biochemical processes.
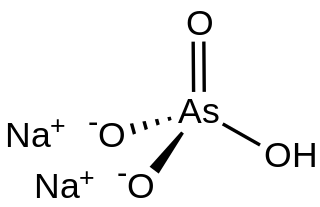
Disodium hydrogen arsenate is the inorganic compound with the formula Na2HAsO4.7H2O. The compound consists of a salt and seven molecules of water of crystallization although for simplicity the formula usually omits the water component. The other sodium arsenates are NaH2AsO4 and Na3AsO4, the latter being called sodium arsenate. Disodium hydrogen arsenate is highly toxic. The salt is the conjugate base of arsenic acid. It is a white, water-soluble solid.