Related Research Articles
In chemistry, a monomer is a molecule that can react together with other monomer molecules to form a larger polymer chain or three-dimensional network in a process called polymerization.
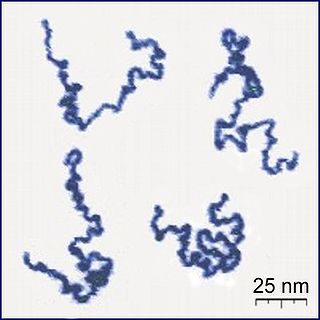
A polymer is a substance or material consisting of very large molecules, or macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compounds, produces unique physical properties including toughness, high elasticity, viscoelasticity, and a tendency to form amorphous and semicrystalline structures rather than crystals.
In polymer chemistry, polymerization, or polymerisation, is a process of reacting monomer molecules together in a chemical reaction to form polymer chains or three-dimensional networks. There are many forms of polymerization and different systems exist to categorize them.

Petrochemicals are the chemical products obtained from petroleum by refining. Some chemical compounds made from petroleum are also obtained from other fossil fuels, such as coal or natural gas, or renewable sources such as maize, palm fruit or sugar cane.

Polyethylene terephthalate, is the most common thermoplastic polymer resin of the polyester family and is used in fibres for clothing, containers for liquids and foods, and thermoforming for manufacturing, and in combination with glass fibre for engineering resins.
A polyamide is a polymer with repeating units linked by amide bonds.
In polymer chemistry, an addition polymer is a polymer that forms by simple linking of monomers without the co-generation of other products. Addition polymerization differs from condensation polymerization, which does co-generate a product, usually water. Addition polymers can be formed by chain polymerization, when the polymer is formed by the sequential addition of monomer units to an active site in a chain reaction, or by polyaddition, when the polymer is formed by addition reactions between species of all degrees of polymerization. Addition polymers are formed by the addition of some simple monomer units repeatedly. Generally polymers are unsaturated compounds like alkenes, alkalines etc. The addition polymerization mainly takes place in free radical mechanism. The free radical mechanism of addition polymerization completed by three steps i.e. Initiation of free radical, Chain propagation, Termination of chain.
A diol is a chemical compound containing two hydroxyl groups. An aliphatic diol is also called a glycol. This pairing of functional groups is pervasive, and many subcategories have been identified.

In polymer chemistry, a copolymer is a polymer derived from more than one species of monomer. The polymerization of monomers into copolymers is called copolymerization. Copolymers obtained by copolymerization of two monomer species are sometimes called bipolymers. Those obtained from three and four monomers are called terpolymers and quaterpolymers, respectively.

Step-growth polymerization refers to a type of polymerization mechanism in which bi-functional or multifunctional monomers react to form first dimers, then trimers, longer oligomers and eventually long chain polymers. Many naturally occurring and some synthetic polymers are produced by step-growth polymerization, e.g. polyesters, polyamides, polyurethanes, etc. Due to the nature of the polymerization mechanism, a high extent of reaction is required to achieve high molecular weight. The easiest way to visualize the mechanism of a step-growth polymerization is a group of people reaching out to hold their hands to form a human chain—each person has two hands. There also is the possibility to have more than two reactive sites on a monomer: In this case branched polymers production take place.
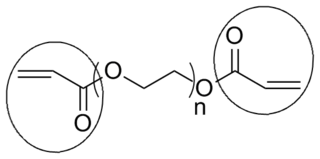
End groups are an important aspect of polymer synthesis and characterization. In polymer chemistry, end groups are functionalities or constitutional units that are at the extremity of a macromolecule or oligomer (IUPAC). In polymer synthesis, like condensation polymerization and free-radical types of polymerization, end-groups are commonly used and can be analyzed for example by nuclear magnetic resonance (NMR) to determine the average length of the polymer. Other methods for characterization of polymers where end-groups are used are mass spectrometry and vibrational spectrometry, like infrared and Raman spectrometry. Not only are these groups important for the analysis of the polymer, but they are also useful for grafting to and from a polymer chain to create a new copolymer. One example of an end group is in the polymer poly(ethylene glycol) diacrylate where the end-groups are circled.
The degree of polymerization, or DP, is the number of monomeric units in a macromolecule or polymer or oligomer molecule.

Polyester is a category of polymers that contain the ester functional group in every repeat unit of their main chain. As a specific material, it most commonly refers to a type called polyethylene terephthalate (PET). Polyesters include naturally occurring chemicals, such as in plants and insects, as well as synthetics such as polybutyrate. Natural polyesters and a few synthetic ones are biodegradable, but most synthetic polyesters are not. Synthetic polyesters are used extensively in clothing.
A glucan is a polysaccharide derived from D-glucose, linked by glycosidic bonds. Many beta-glucans are medically important. They represent a drug target for antifungal medications of the echinocandin class.
Polyester resins are synthetic resins formed by the reaction of dibasic organic acids and polyhydric alcohols. Maleic anhydride is a commonly used raw material with diacid functionality in unsaturated polyester resins. Unsaturated polyester resins are used in sheet moulding compound, bulk moulding compound and the toner of laser printers. Wall panels fabricated from polyester resins reinforced with fiberglass—so-called fiberglass reinforced plastic (FRP)—are typically used in restaurants, kitchens, restrooms and other areas that require washable low-maintenance walls. They are also used extensively in cured-in-place pipe applications. Departments of Transportation in the USA also specify them for use as overlays on roads and bridges. In this application they are known as PCO Polyester Concrete Overlays. These are usually based on isophthalic acid and cut with styrene at high levels—usually up to 50%. Polyesters are also used in anchor bolt adhesives though epoxy based materials are also used. Many companies have and continue to introduce styrene free systems mainly due to odor issues, but also over concerns that styrene is a potential carcinogen. Potable water applications also prefer styrene free. Most polyester resins are viscous, pale coloured liquids consisting of a solution of a polyester in a reactive diluent which is usually styrene, but can also include vinyl toluene and various acrylates.
In polymer chemistry, a repeat unit or repeating unit is a part of a polymer whose repetition would produce the complete polymer chain by linking the repeat units together successively along the chain, like the beads of a necklace.
Polyanhydrides are a class of biodegradable polymers characterized by anhydride bonds that connect repeat units of the polymer backbone chain. Their main application is in the medical device and pharmaceutical industry. In vivo, polyanhydrides degrade into non-toxic diacid monomers that can be metabolized and eliminated from the body. Owing to their safe degradation products, polyanhydrides are considered to be biocompatible.

Hydroxyethylmethacrylate or HEMA (also known as glycol methyacrylate, GMA) is the organic compound with the formula H2C=C(CH3)CO2CH2CH2OH. It is a colorless viscous liquid that readily polymerizes. HEMA is a monomer that is used to make various polymers.
Elastin-like polypeptides (ELPs) are synthetic biopolymers with potential applications in the fields of cancer therapy, tissue scaffolding, and protein purification. For cancer therapy, the addition of functional groups to ELPs can enable them to conjugate with cytotoxic drugs. Also, ELPs may be able to function as polymeric scaffolds, which promote tissue regeneration. This capacity of ELPs has been studied particularly in the context of bone growth. ELPs can also be engineered to recognize specific proteins in solution. The ability of ELPs to undergo morphological changes at certain temperatures enables specific proteins that are bound to the ELPs to be separated out from the rest of the solution via experimental techniques such as centrifugation.

A polyrotaxane is a type of mechanically interlocked molecule consisting of strings and rings, in which multiple rings are threaded onto a molecular axle and prevented from dethreading by two bulky end groups. As oligomeric or polymeric species of rotaxanes, polyrotaxanes are also capable of converting energy input to molecular movements because the ring motions can be controlled by external stimulus. Polyrotaxanes have attracted much attention for decades, because they can help build functional molecular machines with complicated molecular structure.