Hydrogen peroxide is a chemical compound with the formula H
2O
2. In its pure form, it is a very pale blue liquid, slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution in water for consumer use, and in higher concentrations for industrial use. Concentrated hydrogen peroxide, or "high-test peroxide", decomposes explosively when heated and has been used as a propellant in rocketry.
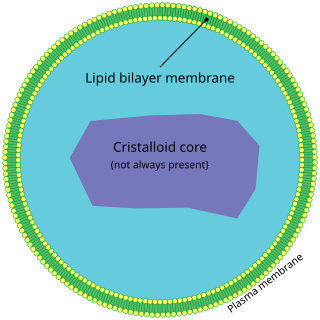
A peroxisome (IPA: [pɛɜˈɹɒksɪˌsoʊm]) is a membrane-bound organelle, a type of microbody, found in the cytoplasm of virtually all eukaryotic cells. Peroxisomes are oxidative organelles. Frequently, molecular oxygen serves as a co-substrate, from which hydrogen peroxide (H2O2) is then formed. Peroxisomes owe their name to hydrogen peroxide generating and scavenging activities. They perform key roles in lipid metabolism and the conversion of reactive oxygen species. Peroxisomes are involved in the catabolism of very long chain fatty acids, branched chain fatty acids, bile acid intermediates (in the liver), D-amino acids, and polyamines, the reduction of reactive oxygen species – specifically hydrogen peroxide – and the biosynthesis of plasmalogens, i.e., ether phospholipids critical for the normal function of mammalian brains and lungs. They also contain approximately 10% of the total activity of two enzymes (Glucose-6-phosphate dehydrogenase and 6-Phosphogluconate dehydrogenase) in the pentose phosphate pathway, which is important for energy metabolism. It is vigorously debated whether peroxisomes are involved in isoprenoid and cholesterol synthesis in animals. Other known peroxisomal functions include the glyoxylate cycle in germinating seeds ("glyoxysomes"), photorespiration in leaves, glycolysis in trypanosomes ("glycosomes"), and methanol and/or amine oxidation and assimilation in some yeasts.
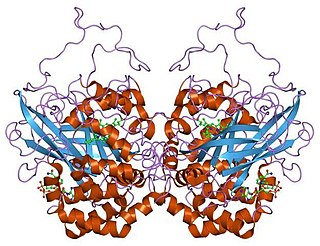
Catalase is a common enzyme found in nearly all living organisms exposed to oxygen which catalyzes the decomposition of hydrogen peroxide to water and oxygen. It is a very important enzyme in protecting the cell from oxidative damage by reactive oxygen species (ROS). Catalase has one of the highest turnover numbers of all enzymes; one catalase molecule can convert millions of hydrogen peroxide molecules to water and oxygen each second.
Rancidification is the process of complete or incomplete oxidation or hydrolysis of fats and oils when exposed to air, light, or moisture or by bacterial action, resulting in unpleasant taste and odor. Specifically, it is the hydrolysis or autoxidation of fats into short-chain aldehydes, ketones and free fatty acids, which are objectionable in taste and odor. When these processes occur in food, undesirable odors and flavors can result.
A molecular lesion, or a point lesion, is damage to the structure of a biological molecule such as DNA, RNA, or protein. This damage may result in the reduction or absence of normal function, and in rare cases the gain of a new function. Lesions in DNA may consist of breaks or other changes in chemical structure of the helix, ultimately preventing transcription. Meanwhile, lesions in proteins consist of both broken bonds and improper folding of the amino acid chain. While many nucleic acid lesions are general across DNA and RNA, some are specific to one, such as thymine dimers being found exclusively in DNA. Several cellular repair mechanisms exist, ranging from global to specific, in order to prevent lasting damage resulting from lesions.

Lipid peroxidation is the chain of reactions of oxidative degradation of lipids. It is the process in which free radicals "steal" electrons from the lipids in cell membranes, resulting in cell damage. This process proceeds by a free radical chain reaction mechanism. It most often affects polyunsaturated fatty acids, because they contain multiple double bonds in between which lie methylene bridges (-CH2-) that possess especially reactive hydrogen atoms. As with any radical reaction, the reaction consists of three major steps: initiation, propagation, and termination. The chemical products of this oxidation are known as lipid peroxides or lipid oxidation products (LOPs).

Oxidative stress reflects an imbalance between the systemic manifestation of reactive oxygen species (ROS) and a biological system's ability to readily detoxify the reactive intermediates or to repair the resulting damage. Disturbances in the normal redox state of cells can cause toxic effects through the production of peroxides and free radicals that damage all components of the cell, including proteins, lipids, and DNA. Oxidative stress from oxidative metabolism causes base damage, as well as strand breaks in DNA. Base damage is mostly indirect and caused by the ROS generated, e.g., O2− (superoxide radical), OH (hydroxyl radical) and H2O2 (hydrogen peroxide). Further, some reactive oxidative species act as cellular messengers in redox signaling. Thus, oxidative stress can cause disruptions in normal mechanisms of cellular signaling.

Organic peroxides are organic compounds containing the peroxide functional group (ROOR′). If the R′ is hydrogen, the compounds are called hydroperoxides, which are discussed in that article. Peresters are the peroxy analog of esters and have general structure RC(O)OOR. The O−O bond of peroxides easily breaks, producing free radicals of the form RO•. Thus, organic peroxides are useful as initiators for some types of polymerisation, such as the epoxy resins used in glass-reinforced plastics. MEKP and benzoyl peroxide are commonly used for this purpose. However, the same property also means that organic peroxides can either intentionally or unintentionally initiate explosive polymerisation in materials with unsaturated chemical bonds, and this process has been used in explosives. Organic peroxides, like their inorganic counterparts, are powerful bleaching agents.
The FOX reagent, or ferrous oxidation−xylenol orange, is used to measure levels of hydrogen peroxide in biological systems. The reagent is incubated with the sample and absorbance of the product form after a series of oxidation reactions is then measured at a wavelength of 560 nm. The reagent itself is an aqueous solution of ferrous ammonium sulfate, sorbitol, sulfuric acid and xylenol orange.

Malondialdehyde (MDA) is the organic compound with the nominal formula CH2(CHO)2. A colorless liquid, malondialdehyde is a highly reactive compound that occurs as the enol. It occurs naturally and is a marker for oxidative stress.
Autoxidation refers to oxidations brought about by reactions with oxygen at normal temperatures, without the intervention of flame or electric spark. The term is usually used to describe the degradation of organic compounds in air at ambient temperatures. Many common phenomena can be attributed to autoxidation, such as food going rancid, the 'drying' of varnishes and paints, and the perishing of rubber. It is also an important concept in both industrial chemistry and biology. Autoxidation is therefore a fairly broad term and can encompass examples of photooxygenation and catalytic oxidation.
Thiobarbituric acid is an organic compound and a heterocycle. It is used as a reagent in assaying malondialdehyde.
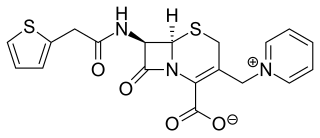
Cephaloridine is a first-generation semisynthetic derivative of antibiotic cephalosporin C. It is a Beta lactam antibiotic, like penicillin. Its chemical structure contains 3 cephems, 4 carboxyl groups and three pyridinium methyl groups.

Cyanide poisoning is poisoning that results from exposure to any of a number of forms of cyanide. Early symptoms include headache, dizziness, fast heart rate, shortness of breath, and vomiting. This phase may then be followed by seizures, slow heart rate, low blood pressure, loss of consciousness, and cardiac arrest. Onset of symptoms usually occurs within a few minutes. Some survivors have long-term neurological problems.
Cytostasis is the inhibition of cell growth and multiplication. Cytostatic refers to a cellular component or medicine that inhibits cell division.

9-Hydroxyoctadecadienoic acid has been used in the literature to designate either or both of two stereoisomer metabolites of the essential fatty acid, linoleic acid: 9(S)-hydroxy-10(E),12(Z)-octadecadienoic acid and 9(R)-hydroxy-10(E),12(Z)-octadecadienoic acid ; these two metabolites differ in having their hydroxy residues in the S or R configurations, respectively. The accompanying figure gives the structure for 9(S)-HETE. Two other 9-hydroxy linoleic acid derivatives occur in nature, the 10E,12E isomers of 9(S)-HODE and 9(R)-HODE viz., 9(S)-hydroxy-10E,12E-octadecadienoic acid and 9(R)-hydroxy-10E,12E-octadecadienoic acid ; these two derivatives have their double bond at carbon 12 in the E or trans configuration as opposed to the Z or cis configuration. The four 9-HODE isomers, particularly under conditions of oxidative stress, may form together in cells and tissues; they have overlapping but not identical biological activities and significances. Because many studies have not distinguished between the S and R stereoisomers and, particularly in identifying tissue levels, the two EE isomers, 9-HODE is used here when the isomer studied is unclear.

Reactive carbonyl species (RCS) are molecules with highly reactive carbonyl groups, and often known for their damaging effects on proteins, nucleic acids, and lipids. They are often generated as metabolic products. Important RCSs include 3-deoxyglucosone, glyoxal, and methylglyoxal. RCSs react with amines and thiol groups leading to advanced glycation endproducts (AGEs). AGE's are indicators of diabetes.

Reactive aldehyde species (RASP), also known as reactive aldehydes, refer to a class of electrophilic organic aldehyde molecules that are generally toxic or facilitate inflammation. RASP covalently react with amine groups and thiol groups, particularly in proteins. Following threshold amounts of binding to the electrophile-responsive proteome, RASP modify protein function, as has been described with MAP kinase, protein kinase C, and other proteins that potentiate cytokine release and other aspects of inflammation. Binding of RASP to proteins can also lead to NF-kB activation, autoantibody formation, inflammasome activation, and activation of Scavenger Receptor A. RASP are formed via a variety of processes, including oxidation of alcohols, polyamine metabolism and lipid peroxidation. In addition to binding to proteins and other amine or thiol-containing molecules such as glutathione, RASP are metabolized by aldehyde dehydrogenases or aldehyde reductases. Due to the toxicity of RASP, only a small number of genetic mutations in aldehyde dehydrogenases allow for viable offspring, resulting in Sjögren-Larsson Syndrome, Succinic Semi-Aldehyde Dehydrogenase Deficiency, and other rare diseases.

Isotope effect is observed when molecules containing heavier isotopes of the same atoms are engaged in a chemical reaction at a slower rate. Deuterium-reinforced lipids can be used for the protection of living cells by slowing the chain reaction of lipid peroxidation. The lipid bilayer of the cell and organelle membranes contain polyunsaturated fatty acids (PUFA) are key components of cell and organelle membranes. Any process that either increases oxidation of PUFAs or hinders their ability to be replaced can lead to serious disease. Correspondingly, drugs that stop the chain reaction of lipid peroxidation have preventive and therapeutic potential.

Reinforced lipids are lipid molecules in which some of the fatty acids contain deuterium instead of hydrogen. They can be used for the protection of living cells by slowing the chain reaction due to isotope effect on lipid peroxidation. The lipid bilayer of the cell and organelle membranes contain polyunsaturated fatty acids (PUFA) are key components of cell and organelle membranes. Any process that either increases oxidation of PUFAs or hinders their ability to be replaced can lead to serious disease. Correspondingly, use of reinforced lipids that stop the chain reaction of lipid peroxidation has preventive and therapeutic potential.













