
Tumor necrosis factor is an adipokine and a cytokine. TNF is a member of the TNF superfamily, which consists of various transmembrane proteins with a homologous TNF domain.
Granzyme B (GrB) is a serine protease most commonly found in the granules of natural killer cells and cytotoxic T cells. It is secreted by these cells along with the pore forming protein perforin to mediate apoptosis in target cells.

Cathepsins are proteases found in all animals as well as other organisms. There are approximately a dozen members of this family, which are distinguished by their structure, catalytic mechanism, and which proteins they cleave. Most of the members become activated at the low pH found in lysosomes. Thus, the activity of this family lies almost entirely within those organelles. There are, however, exceptions such as cathepsin K, which works extracellularly after secretion by osteoclasts in bone resorption. Cathepsins have a vital role in mammalian cellular turnover.

The death-effector domain (DED) is a protein interaction domain found only in eukaryotes that regulates a variety of cellular signalling pathways. The DED domain is found in inactive procaspases and proteins that regulate caspase activation in the apoptosis cascade such as FAS-associating death domain-containing protein (FADD). FADD recruits procaspase 8 and procaspase 10 into a death induced signaling complex (DISC). This recruitment is mediated by a homotypic interaction between the procaspase DED and a second DED that is death effector domain in an adaptor protein that is directly associated with activated TNF receptors. Complex formation allows proteolytic activation of procaspase into the active caspase form which results in the initiation of apoptosis. Structurally the DED domain are a subclass of protein motif known as the death fold and contains 6 alpha helices, that closely resemble the structure of the Death domain (DD).

Caspase-1/Interleukin-1 converting enzyme (ICE) is an evolutionarily conserved enzyme that proteolytically cleaves other proteins, such as the precursors of the inflammatory cytokines interleukin 1β and interleukin 18 as well as the pyroptosis inducer Gasdermin D, into active mature peptides. It plays a central role in cell immunity as an inflammatory response initiator. Once activated through formation of an inflammasome complex, it initiates a proinflammatory response through the cleavage and thus activation of the two inflammatory cytokines, interleukin 1β (IL-1β) and interleukin 18 (IL-18) as well as pyroptosis, a programmed lytic cell death pathway, through cleavage of Gasdermin D. The two inflammatory cytokines activated by Caspase-1 are excreted from the cell to further induce the inflammatory response in neighboring cells.

Cathepsin S is a protein that in humans is encoded by the CTSS gene. Transcript variants utilizing alternative polyadenylation signals exist for this gene.
Casein kinase 2 (CK2/CSNK2) is a serine/threonine-selective protein kinase that has been implicated in cell cycle control, DNA repair, regulation of the circadian rhythm, and other cellular processes. De-regulation of CK2 has been linked to tumorigenesis as a potential protection mechanism for mutated cells. Proper CK2 function is necessary for survival of cells as no knockout models have been successfully generated.

Cathepsin G is a protein that in humans is encoded by the CTSG gene. It is one of the three serine proteases of the chymotrypsin family that are stored in the azurophil granules, and also a member of the peptidase S1 protein family. Cathepsin G plays an important role in eliminating intracellular pathogens and breaking down tissues at inflammatory sites, as well as in anti-inflammatory response.
Pyroptosis is a highly inflammatory form of lytic programmed cell death that occurs most frequently upon infection with intracellular pathogens and is likely to form part of the antimicrobial response. This process promotes the rapid clearance of various bacterial, viral, fungal and protozoan infections by removing intracellular replication niches and enhancing the host's defensive responses. Pyroptosis can take place in immune cells and is also reported to occur in keratinocytes and some epithelial cells.

Cathepsin B belongs to a family of lysosomal cysteine proteases known as the cysteine cathepsins and plays an important role in intracellular proteolysis. In humans, cathepsin B is encoded by the CTSB gene. Cathepsin B is upregulated in certain cancers, in pre-malignant lesions, and in various other pathological conditions.

Cathepsin L2,, is a protein encoded in humans by the CTSV gene.
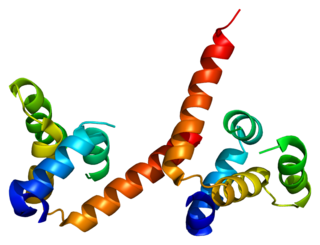
Mucosa-associated lymphoid tissue lymphoma translocation protein 1 is a protein that in humans is encoded by the MALT1 gene. It's the human paracaspase.

Inflammasomes are cytosolic multiprotein oligomers of the innate immune system responsible for the activation of inflammatory responses. Activation and assembly of the inflammasome promotes proteolytic cleavage, maturation and secretion of pro-inflammatory cytokines interleukin 1β (IL-1β) and interleukin 18 (IL-18), as well as cleavage of Gasdermin-D. The N-terminal fragment resulting from this cleavage induces a pro-inflammatory form of programmed cell death distinct from apoptosis, referred to as pyroptosis, and is responsible for secretion of the mature cytokines, presumably through the formation of pores in the plasma membrane.

A pyrin domain is a protein domain and a subclass of protein motif known as the death fold, the 4th and most recently discovered member of the death domain superfamily (DDF). It was originally discovered in the pyrin protein, or marenostrin, encoded by MEFV. The mutation of the MEFV gene is the cause of the disease known as Familial Mediterranean Fever. The domain is encoded in 23 human proteins and at least 31 mouse genes.

In molecular biology mir-210 microRNA is a short RNA molecule. MicroRNAs function to regulate the expression levels of other genes by several mechanisms.

The Interleukin-1 family is a group of 11 cytokines that plays a central role in the regulation of immune and inflammatory responses to infections or sterile insults.

Immunomodulatory imide drugs (IMiDs) are a class of immunomodulatory drugs containing an imide group. The IMiD class includes thalidomide and its analogues (lenalidomide, pomalidomide, and iberdomide. These drugs may also be referred to as 'Cereblon modulators'. Cereblon is the protein targeted by this class of drugs.

Paraptosis is a type of programmed cell death, morphologically distinct from apoptosis and necrosis. The defining features of paraptosis are cytoplasmic vacuolation, independent of caspase activation and inhibition, and lack of apoptotic morphology. Paraptosis lacks several of the hallmark characteristics of apoptosis, such as membrane blebbing, chromatin condensation, and nuclear fragmentation. Like apoptosis and other types of programmed cell death, the cell is involved in causing its own death, and gene expression is required. This is in contrast to necrosis, which is non-programmed cell death that results from injury to the cell.

Dicycloplatin is a chemotherapy medication used to treat a number of cancers which includes the non-small-cell lung carcinoma and prostate cancer.
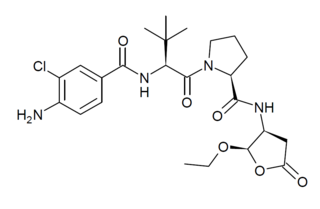
Belnacasan (VX-765) is a drug developed by Vertex Pharmaceuticals which acts as a potent and selective inhibitor of the enzyme caspase 1. This enzyme is involved in inflammation and cell death, and consequently blocking its action may be useful for various medical applications, including treatment of epilepsy, arthritis, aiding recovery from heart attack and slowing the progression of Alzheimer's disease. Belnacasan is an orally active prodrug, being converted in the body to the active drug VRT-043198 (O-desethyl-belnacasan). However while belnacasan has proved well tolerated in human clinical trials, it has not shown sufficient efficacy to be approved for use for any of the applications suggested to date, though research continues into possible future uses of this or similar drugs.

















