Related Research Articles
In chemistry, amines are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia, wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group. Important amines include amino acids, biogenic amines, trimethylamine, and aniline. Inorganic derivatives of ammonia are also called amines, such as monochloramine.

The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals the pressure surrounding the liquid and the liquid changes into a vapor.
A lubricant is a substance that helps to reduce friction between surfaces in mutual contact, which ultimately reduces the heat generated when the surfaces move. It may also have the function of transmitting forces, transporting foreign particles, or heating or cooling the surfaces. The property of reducing friction is known as lubricity.

Vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases at a given temperature in a closed system. The equilibrium vapor pressure is an indication of a liquid's thermodynamic tendency to evaporate. It relates to the balance of particles escaping from the liquid in equilibrium with those in a coexisting vapor phase. A substance with a high vapor pressure at normal temperatures is often referred to as volatile. The pressure exhibited by vapor present above a liquid surface is known as vapor pressure. As the temperature of a liquid increases, the attractive interactions between liquid molecules become less significant in comparison to the entropy of those molecules in the gas phase, increasing the vapor pressure. Thus, liquids with strong intermolecular interactions are likely to have smaller vapor pressures, with the reverse true for weaker interactions.

Diesel fuel, also called diesel oil or historically heavy oil, is any liquid fuel specifically designed for use in a diesel engine, a type of internal combustion engine in which fuel ignition takes place without a spark as a result of compression of the inlet air and then injection of fuel. Therefore, diesel fuel needs good compression ignition characteristics.

Kerogen is solid, insoluble organic matter in sedimentary rocks. It consists of a variety of organic materials, including dead plants, algae, and other microorganisms, that have been compressed and heated by geological processes. Altogether kerogen is estimated to contain 1016 tons of carbon. This makes it the most abundant source of organic compounds on earth, exceeding the total organic content of living matter 10,000-fold.

Aniline is an organic compound with the formula C6H5NH2. Consisting of a phenyl group attached to an amino group, aniline is the simplest aromatic amine. It is an industrially significant commodity chemical, as well as a versatile starting material for fine chemical synthesis. Its main use is in the manufacture of precursors to polyurethane, dyes, and other industrial chemicals. Like most volatile amines, it has the odor of rotten fish. It ignites readily, burning with a smoky flame characteristic of aromatic compounds. It is toxic to humans.
Tribology is the science and engineering of interacting surfaces in relative motion. It includes the study and application of the principles of friction, lubrication and wear. Tribology is highly interdisciplinary, drawing on many academic fields, including physics, chemistry, materials science, mathematics, biology and engineering. People who work in the field of tribology are referred to as tribologists.

An O-ring, also known as a packing or a toric joint, is a mechanical gasket in the shape of a torus; it is a loop of elastomer with a round cross-section, designed to be seated in a groove and compressed during assembly between two or more parts, forming a seal at the interface.

In organic chemistry, nitro compounds are organic compounds that contain one or more nitro functional groups. The nitro group is one of the most common explosophores used globally. The nitro group is also strongly electron-withdrawing. Because of this property, C−H bonds alpha (adjacent) to the nitro group can be acidic. For similar reasons, the presence of nitro groups in aromatic compounds retards electrophilic aromatic substitution but facilitates nucleophilic aromatic substitution. Nitro groups are rarely found in nature. They are almost invariably produced by nitration reactions starting with nitric acid.
Well logging, also known as borehole logging is the practice of making a detailed record of the geologic formations penetrated by a borehole. The log may be based either on visual inspection of samples brought to the surface or on physical measurements made by instruments lowered into the hole. Some types of geophysical well logs can be done during any phase of a well's history: drilling, completing, producing, or abandoning. Well logging is performed in boreholes drilled for the oil and gas, groundwater, mineral and geothermal exploration, as well as part of environmental and geotechnical studies.
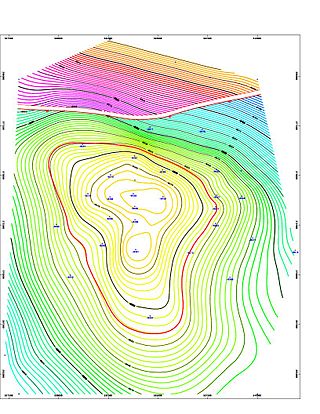
A petroleum reservoir or oil and gas reservoir is a subsurface accumulation of hydrocarbons contained in porous or fractured rock formations.

A wellhead is the component at the surface of an oil or gas well that provides the structural and pressure-containing interface for the drilling and production equipment.
Enhanced oil recovery, also called tertiary recovery, is the extraction of crude oil from an oil field that cannot be extracted otherwise. EOR can extract 30% to 60% or more of a reservoir's oil, compared to 20% to 40% using primary and secondary recovery. According to the US Department of Energy, carbon dioxide and water are injected along with one of three EOR techniques: thermal injection, gas injection, and chemical injection. More advanced, speculative EOR techniques are sometimes called quaternary recovery.
In chemistry, a regular solution is a solution whose entropy of mixing is equal to that of an ideal solution with the same composition, but is non-ideal due to a nonzero enthalpy of mixing. Such a solution is formed by random mixing of components of similar molar volume and without strong specific interactions, and its behavior diverges from that of an ideal solution by showing phase separation at intermediate compositions and temperatures. Its entropy of mixing is equal to that of an ideal solution with the same composition, due to random mixing without strong specific interactions. For two components

Phenyl ether polymers are a class of polymers that contain a phenoxy or a thiophenoxy group as the repeating group in ether linkages. Commercial phenyl ether polymers belong to two chemical classes: polyphenyl ethers (PPEs) and polyphenylene oxides (PPOs). The phenoxy groups in the former class of polymers do not contain any substituents whereas those in the latter class contain 2 to 4 alkyl groups on the phenyl ring. The structure of an oxygen-containing PPE is provided in Figure 1 and that of a 2, 6-xylenol derived PPO is shown in Figure 2. Either class can have the oxygen atoms attached at various positions around the rings.
This glossary of chemistry terms is a list of terms and definitions relevant to chemistry, including chemical laws, diagrams and formulae, laboratory tools, glassware, and equipment. Chemistry is a physical science concerned with the composition, structure, and properties of matter, as well as the changes it undergoes during chemical reactions; it features an extensive vocabulary and a significant amount of jargon.
Crude oil is extracted from the bedrock before being processed in several stages, removing natural contaminants and undesirable hydrocarbons. This separation process produces mineral oil, which can in turn be denoted as paraffinic, naphthenic or aromatic. The differences between these different types of oils are not clear-cut, but mainly depend on the predominant hydrocarbon types in the oil. Paraffinic oil, for example, contains primarily higher alkanes, whereas naphthenic oils have a high share of cyclic alkanes in the mixture.
Oilfield terminology refers to the jargon used by those working in fields within and related to the upstream segment of the petroleum industry. It includes words and phrases describing professions, equipment, and procedures specific to the industry. It may also include slang terms used by oilfield workers to describe the same.
Aniline is an organic compound with the formula C
6H
5NH
2.
References
- ↑ Steven A., Treese; Peter R., Pujado; David S. J., Jones (2015). Handbook of Petroleum Processing (2 ed.). Springer. p. 1693. ISBN 978-3-319-14528-0.
- 1 2 3 "Aniline point test | Schlumberger Oilfield glossary". Schlumberger Oilfield glossary.
{{cite web}}: CS1 maint: url-status (link)
- Gupta, O.P. Fuels, Furnaces, Refractories.