
Tuberculosis (TB), also known colloquially as the "white death", or historically as consumption, is an infectious disease usually caused by Mycobacterium tuberculosis (MTB) bacteria. Tuberculosis generally affects the lungs, but it can also affect other parts of the body. Most infections show no symptoms, in which case it is known as latent tuberculosis. Around 10% of latent infections progress to active disease which, if left untreated, kill about half of those affected. Typical symptoms of active TB are chronic cough with blood-containing mucus, fever, night sweats, and weight loss. Infection of other organs can cause a wide range of symptoms.

The Mantoux test or Mendel–Mantoux test is a tool for screening for tuberculosis (TB) and for tuberculosis diagnosis. It is one of the major tuberculin skin tests used around the world, largely replacing multiple-puncture tests such as the tine test. The Heaf test, a form of tine test, was used until 2005 in the UK, when it was replaced by the Mantoux test. The Mantoux test is endorsed by the American Thoracic Society and Centers for Disease Control and Prevention. It was also used in the USSR and is now prevalent in most of the post-Soviet states, although Soviet mantoux produced many false positives due to children's allergic reaction.

HIV tests are used to detect the presence of the human immunodeficiency virus (HIV), the virus that causes acquired immunodeficiency syndrome (AIDS), in serum, saliva, or urine. Such tests may detect antibodies, antigens, or RNA.

The Venereal Disease Research Laboratory test (VDRL) is a blood test for syphilis and related non-venereal treponematoses that was developed by the eponymous US laboratory. The VDRL test is used to screen for syphilis, whereas other, more specific tests are used to diagnose the disease.

Feline leukemia virus (FeLV) is a retrovirus that infects cats. FeLV can be transmitted from infected cats when the transfer of saliva or nasal secretions is involved. If not defeated by the animal's immune system, the virus weakens the cat's immune system, which can lead to diseases which can be lethal. Because FeLV is cat-to-cat contagious, FeLV+ cats should only live with other FeLV+ cats.

Hybridoma technology is a method for producing large numbers of identical antibodies. This process starts by injecting a mouse with an antigen that provokes an immune response. A type of white blood cell, the B cell, produces antibodies that bind to the injected antigen. These antibody producing B-cells are then harvested from the mouse and, in turn, fused with immortal myeloma cancer cells, to produce a hybrid cell line called a hybridoma, which has both the antibody-producing ability of the B-cell and the longevity and reproductivity of the myeloma. The hybridomas can be grown in culture, each culture starting with one viable hybridoma cell, producing cultures each of which consists of genetically identical hybridomas which produce one antibody per culture (monoclonal) rather than mixtures of different antibodies (polyclonal). The myeloma cell line that is used in this process is selected for its ability to grow in tissue culture and for an absence of antibody synthesis. In contrast to polyclonal antibodies, which are mixtures of many different antibody molecules, the monoclonal antibodies produced by each hybridoma line are all chemically identical.

Tuberculosis is diagnosed by finding Mycobacterium tuberculosis bacteria in a clinical specimen taken from the patient. While other investigations may strongly suggest tuberculosis as the diagnosis, they cannot confirm it.
This is a list of AIDS-related topics, many of which were originally taken from the public domain U.S. Department of Health Glossary of HIV/AIDS-Related Terms, 4th Edition.

Tuberculous meningitis, also known as TB meningitis or tubercular meningitis, is a specific type of bacterial meningitis caused by the Mycobacterium tuberculosis infection of the meninges—the system of membranes which envelop the central nervous system.

'Interferon-γ release assays (IGRA) are medical tests used in the diagnosis of some infectious diseases, especially tuberculosis. Interferon-γ (IFN-γ) release assays rely on the fact that T-lymphocytes will release IFN-γ when exposed to specific antigens. These tests are mostly developed for the field of tuberculosis diagnosis, but in theory, may be used in the diagnosis of other diseases that rely on cell-mediated immunity, e.g. cytomegalovirus and leishmaniasis and COVID-19. For example, in patients with cutaneous adverse drug reactions, the challenge of peripheral blood lymphocytes with the drug causing the reaction produced a positive test result for half of the drugs tested.
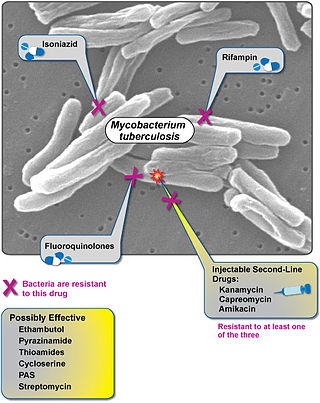
Extensively drug-resistant tuberculosis (XDR-TB) is a form of tuberculosis caused by bacteria that are resistant to some of the most effective anti-TB drugs. XDR-TB strains have arisen after the mismanagement of individuals with multidrug-resistant TB (MDR-TB).
Interferon-gamma release assays (IGRAs) are diagnostic tools for latent tuberculosis infection (LTBI). They are surrogate markers of Mycobacterium tuberculosis infection and indicate a cellular immune response to M. tuberculosis if the latter is present.
T-SPOT.TB is a type of ELISpot assay used for tuberculosis diagnosis, which belongs to the group of interferon gamma release assays. The test is manufactured by Oxford Immunotec in the UK. It is available in most European countries, the United States as well as various other countries. It was developed by researchers at the University of Oxford in England.
Anti-streptolysin O is the antibody made against streptolysin O, an immunogenic, oxygen-labile streptococcal hemolytic exotoxin produced by most strains of group A and many strains of groups C and G Streptococcus bacteria. The "O" in the name stands for oxygen-labile; the other related toxin being oxygen-stable streptolysin-S. The main function of streptolysin O is to cause hemolysis —in particular, beta-hemolysis.
In the diagnostic laboratory, virus infections can be confirmed by a myriad of methods. Diagnostic virology has changed rapidly due to the advent of molecular techniques and increased clinical sensitivity of serological assays.
The Xpert MTB/RIF is a cartridge-based nucleic acid amplification test (NAAT) for simultaneous rapid tuberculosis diagnosis and rapid antibiotic sensitivity test. It is an automated diagnostic test that can identify Mycobacterium tuberculosis (MTB) DNA and resistance to rifampicin (RIF). It was co-developed by the laboratory of Professor David Alland at the University of Medicine and Dentistry of New Jersey (UMDNJ), Cepheid Inc. and Foundation for Innovative New Diagnostics, with additional financial support from the US National Institutes of Health (NIH).
mChip is a portable blood test device which is capable of diagnosing an infection of HIV or Syphilis within 15 minutes and could be used effectively against HIV/AIDS in developing countries. The mChip costs about US$ 1 and the entire diagnostic kit costs about US$ 100. mChip was developed so that people in regions with poor health facilities can access portable diagnosis for HIV/AIDS rather than travelling long distances to go to clinics for diagnosis.

Surround optical-fiber immunoassay (SOFIA) is an ultrasensitive, in vitro diagnostic platform incorporating a surround optical-fiber assembly that captures fluorescence emissions from an entire sample. The technology's defining characteristics are its extremely high limit of detection, sensitivity, and dynamic range. SOFIA's sensitivity is measured at the attogram level (10−18 g), making it about one billion times more sensitive than conventional diagnostic techniques. Based on its enhanced dynamic range, SOFIA is able to discriminate levels of analyte in a sample over 10 orders of magnitude, facilitating accurate titering.
The mainstay of malaria diagnosis has been the microscopic examination of blood, utilizing blood films. Although blood is the sample most frequently used to make a diagnosis, both saliva and urine have been investigated as alternative, less invasive specimens. More recently, modern techniques utilizing antigen tests or polymerase chain reaction have been discovered, though these are not widely implemented in malaria endemic regions. Areas that cannot afford laboratory diagnostic tests often use only a history of subjective fever as the indication to treat for malaria.
Viral load monitoring for HIV is the regular measurement of the viral load of individual HIV-positive people as part of their personal plan for treatment of HIV/AIDS. A count of the viral load is routine before the start of HIV treatment.










