
Chemotherapy is a type of cancer treatment that uses one or more anti-cancer drugs as part of a standardized chemotherapy regimen. Chemotherapy may be given with a curative intent or it may aim to prolong life or to reduce symptoms. Chemotherapy is one of the major categories of the medical discipline specifically devoted to pharmacotherapy for cancer, which is called medical oncology.

Colorectal cancer (CRC), also known as bowel cancer, colon cancer, or rectal cancer, is the development of cancer from the colon or rectum. Signs and symptoms may include blood in the stool, a change in bowel movements, weight loss, and fatigue. Most colorectal cancers are due to old age and lifestyle factors, with only a small number of cases due to underlying genetic disorders. Risk factors include diet, obesity, smoking, and lack of physical activity. Dietary factors that increase the risk include red meat, processed meat, and alcohol. Another risk factor is inflammatory bowel disease, which includes Crohn's disease and ulcerative colitis. Some of the inherited genetic disorders that can cause colorectal cancer include familial adenomatous polyposis and hereditary non-polyposis colon cancer; however, these represent less than 5% of cases. It typically starts as a benign tumor, often in the form of a polyp, which over time becomes cancerous.

Bortezomib, sold under the brand name Velcade among others, is an anti-cancer medication used to treat multiple myeloma and mantle cell lymphoma. This includes multiple myeloma in those who have and have not previously received treatment. It is generally used together with other medications. It is given by injection.

Oxaliplatin, sold under the brand name Eloxatin among others, is a cancer medication used to treat colorectal cancer. It is given by injection into a vein.
FOLFOX is a chemotherapy regimen for treatment of colorectal cancer, made up of the drugs folinic acid, fluorouracil, and oxaliplatin.

Dihydropyrimidine dehydrogenase deficiency is an autosomal recessive metabolic disorder in which there is absent or significantly decreased activity of dihydropyrimidine dehydrogenase, an enzyme involved in the metabolism of uracil and thymine.
Triplatin tetranitrate is a platinum-based cytotoxic drug that underwent clinical trials for the treatment of human cancer. The drug acts by forming adducts with cellular DNA, preventing DNA transcription and replication, thereby inducing apoptosis. Other platinum-containing anticancer drugs include cisplatin, carboplatin, and oxaliplatin.
Matuzumab is a humanized monoclonal antibody for the treatment of cancer. It binds to the epidermal growth factor receptor (EGFR) with high affinity. The mouse monoclonal antibody (mAb425) from which matuzumab was developed at the Wistar Institute in Philadelphia, Pennsylvania

Chemotherapy-induced acral erythema, also known as palmar-plantar erythrodysesthesia or hand-foot syndrome is reddening, swelling, numbness and desquamation on palms of the hands and soles of the feet that can occur after chemotherapy in patients with cancer. Hand-foot syndrome is also rarely seen in sickle-cell disease. These skin changes usually are well demarcated. Acral erythema typically disappears within a few weeks after discontinuation of the offending drug.
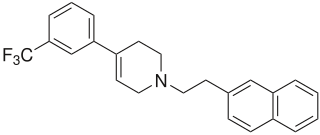
Xaliproden is a drug which acts as a 5HT1A agonist. It has neurotrophic and neuroprotective effects in vitro, and has been proposed for use in the treatment of several neurodegenerative conditions including amyotrophic lateral sclerosis (ALS) and Alzheimer's disease.
Tegafur/uracil is a chemotherapy drug combination used in the treatment of cancer, primarily bowel cancer. It is also called UFT or UFUR.
Ramucirumab is a fully human monoclonal antibody (IgG1) developed for the treatment of solid tumors. This drug was developed by ImClone Systems Inc. It was isolated from a native phage display library from Dyax.

Human papillomavirus-positive oropharyngeal cancer, is a cancer of the throat caused by the human papillomavirus type 16 virus (HPV16). In the past, cancer of the oropharynx (throat) was associated with the use of alcohol or tobacco or both, but the majority of cases are now associated with the HPV virus, acquired by having oral contact with the genitals of a person who has a genital HPV infection. Risk factors include having a large number of sexual partners, a history of oral-genital sex or anal–oral sex, having a female partner with a history of either an abnormal Pap smear or cervical dysplasia, having chronic periodontitis, and, among men, younger age at first intercourse and a history of genital warts. HPV-positive OPC is considered a separate disease from HPV-negative oropharyngeal cancer.
Glembatumumab vedotin is an antibody-drug conjugate (ADC) that targets cancer cells expressing transmembrane glycoprotein NMB (GPNMB).

Trastuzumab emtansine, sold under the brand name Kadcyla, is an antibody-drug conjugate consisting of the humanized monoclonal antibody trastuzumab (Herceptin) covalently linked to the cytotoxic agent DM1. Trastuzumab alone stops growth of cancer cells by binding to the HER2 receptor, whereas trastuzumab emtansine undergoes receptor-mediated internalization into cells, is catabolized in lysosomes where DM1-containing catabolites are released and subsequently bind tubulin to cause mitotic arrest and cell death. Trastuzumab binding to HER2 prevents homodimerization or heterodimerization (HER2/HER3) of the receptor, ultimately inhibiting the activation of MAPK and PI3K/AKT cellular signalling pathways. Because the monoclonal antibody targets HER2, and HER2 is only over-expressed in cancer cells, the conjugate delivers the cytotoxic agent DM1 specifically to tumor cells. The conjugate is abbreviated T-DM1.
Chemotherapy-induced peripheral neuropathy (CIPN) is a nerve-damaging side effect of antineoplastic agents in the common cancer treatment, chemotherapy. CIPN afflicts between 30% and 40% of patients undergoing chemotherapy. Antineoplastic agents in chemotherapy are designed to eliminate rapidly dividing cancer cells, but they can also damage healthy structures, including the peripheral nervous system. CIPN involves various symptoms such as tingling, pain, and numbness in the hands and feet. These symptoms can impair activities of daily living, such as typing or dressing, reduce balance, and increase risk of falls and hospitalizations. They can also give cause to reduce or discontinue chemotherapy. Researchers have conducted clinical trials and studies to uncover the various symptoms, causes, pathogenesis, diagnoses, risk factors, and treatments of CIPN.

Momelotinib (INN, formerly GS-0387, CYT-387) is an inhibitor of Janus kinases JAK1 and JAK2, acting as an ATP competitor with IC50 values of 11 and 18 nM, respectively. The inhibitor is significantly less active towards other kinases, including JAK3 (IC50 = 0.16 μM).
Electrochemical skin conductance (ESC) is an objective, non-invasive and quantitative electrophysiological measure. It is based on reverse iontophoresis and (multiple) steady chronoamperometry.
Bradley J. Monk is an American gynecologic oncologist, academician and researcher. He is a Professor on the Clinical Scholar Track in the Department of Obstetrics and Gynecology at the University of Arizona College of Medicine in Phoenix, Arizona, as well as at the Creighton University School of Medicine in Omaha, Nebraska. He also serves as Director of the Division of Gynecologic Oncology at the St. Joseph's Hospital and Medical Center in Phoenix.
Xavier Pivot is a French oncologist, university professor, breast cancer specialist and general director of the Strasbourg Cancer Institute. He is best known for his research on treatments with trastuzumab in HER2-positive breast cancers.










