
Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, and transition metals, and sometimes broadened to include metalloids like boron, silicon, and selenium, as well. Aside from bonds to organyl fragments or molecules, bonds to 'inorganic' carbon, like carbon monoxide, cyanide, or carbide, are generally considered to be organometallic as well. Some related compounds such as transition metal hydrides and metal phosphine complexes are often included in discussions of organometallic compounds, though strictly speaking, they are not necessarily organometallic. The related but distinct term "metalorganic compound" refers to metal-containing compounds lacking direct metal-carbon bonds but which contain organic ligands. Metal β-diketonates, alkoxides, dialkylamides, and metal phosphine complexes are representative members of this class. The field of organometallic chemistry combines aspects of traditional inorganic and organic chemistry.

Arsine (IUPAC name: arsane) is an inorganic compound with the formula AsH3. This flammable, pyrophoric, and highly toxic pnictogen hydride gas is one of the simplest compounds of arsenic. Despite its lethality, it finds some applications in the semiconductor industry and for the synthesis of organoarsenic compounds. The term arsine is commonly used to describe a class of organoarsenic compounds of the formula AsH3−xRx, where R = aryl or alkyl. For example, As(C6H5)3, called triphenylarsine, is referred to as "an arsine".

Cacodyl, also known as dicacodyl or tetramethyldiarsine, (CH3)2As–As(CH3)2, is an organoarsenic compound that constitutes a major part of "Cadet's fuming liquid" (named after the French chemist Louis Claude Cadet de Gassicourt). It is a poisonous oily liquid with an extremely unpleasant garlicky odor. Cacodyl undergoes spontaneous combustion in dry air.

Arsenic trioxide, sold under the brand name Trisenox among others, is an inorganic compound with the formula As
2O
3. As an industrial chemical, its major uses include the manufacture of wood preservatives, pesticides, and glass. It is also used as a medication to treat a type of cancer known as acute promyelocytic leukemia. For this use it is given by injection into a vein.

Cacodylic acid is an organoarsenic compound with the formula (CH3)2AsO2H. With the formula R2As(O)OH, it is the simplest of the arsinic acids. It is a colorless solid that is soluble in water.

Cacodyl oxide is a chemical compound of the formula [(CH3)2As]2O. This organoarsenic compound is primarily of historical significance since it is sometimes considered to be the first organometallic compound synthesized in relatively pure form.

Arsenic tribromide is the inorganic compound with the formula AsBr3. This pyramidal molecule is the only known binary arsenic bromide. AsBr3 is noteworthy for its very high refractive index of approximately 2.3. It also has a very high diamagnetic susceptibility. The compound exists as colourless deliquescent crystals that fume in moist air.

Potassium acetate (also called potassium ethanoate), (CH3COOK) is the potassium salt of acetic acid. It is a hygroscopic solid at room temperature.

Rhenium(VII) oxide is the inorganic compound with the formula Re2O7. This yellowish solid is the anhydride of HOReO3. Perrhenic acid, Re2O7·2H2O, is closely related to Re2O7. Re2O7 is the raw material for all rhenium compounds, being the volatile fraction obtained upon roasting the host ore.
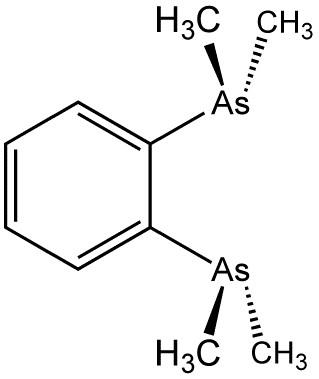
1,2-Bis(dimethylarsino)benzene (diars) is the organoarsenic compound with the formula C6H4(As(CH3)2)2. The molecule consists of two dimethylarsino groups attached to adjacent carbon centers of a benzene ring. It is a chelating ligand in coordination chemistry. This colourless oil is commonly abbreviated "diars."
Organoarsenic chemistry is the chemistry of compounds containing a chemical bond between arsenic and carbon. A few organoarsenic compounds, also called "organoarsenicals," are produced industrially with uses as insecticides, herbicides, and fungicides. In general these applications are declining in step with growing concerns about their impact on the environment and human health. The parent compounds are arsane and arsenic acid. Despite their toxicity, organoarsenic biomolecules are well known.
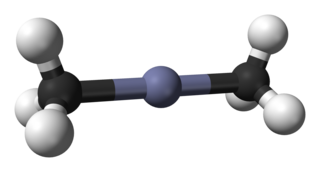
Dimethylzinc, also known as zinc methyl, DMZ, or DMZn, is an organozinc compound with the chemical formula Zn(CH3)2. It belongs to the large series of similar compounds such as diethylzinc.
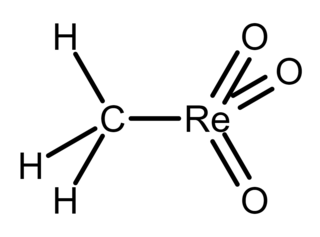
Methylrhenium trioxide, also known as methyltrioxorhenium(VII), is an organometallic compound with the formula CH3−ReO3. It is a volatile, colourless solid that has been used as a catalyst in some laboratory experiments. In this compound, rhenium has a tetrahedral coordination geometry with one methyl and three oxo ligands. The oxidation state of rhenium is +7.
Arsenic pentafluoride is a chemical compound of arsenic and fluorine. It is a toxic, colorless gas. The oxidation state of arsenic is +5.
Louis Claude Cadet de Gassicourt was a French chemist who synthesised the first organometalic compound.

Methyldichloroarsine, sometimes abbreviated "MD" and also known as methyl Dick, is an organoarsenic compound with the formula CH3AsCl2. This colourless volatile liquid is a highly toxic vesicant that has been used in chemical warfare.

Dichlorine hexoxide is the chemical compound with the molecular formula Cl
2O
6, which is correct for its gaseous state. However, in liquid or solid form, this chlorine oxide ionizes into the dark red ionic compound chloryl perchlorate [ClO
2]+
[ClO
4]−
, which may be thought of as the mixed anhydride of chloric and perchloric acids.
Organorhenium chemistry describes the compounds with Re−C bonds. Because rhenium is a rare element, relatively few applications exist, but the area has been a rich source of concepts and a few useful catalysts.
Radical theory is an obsolete scientific theory in chemistry describing the structure of organic compounds. The theory was pioneered by Justus von Liebig, Friedrich Wöhler and Auguste Laurent around 1830 and is not related to the modern understanding of free radicals. In this theory, organic compounds were thought to exist as combinations of radicals that could be exchanged in chemical reactions just as chemical elements could be interchanged in inorganic compounds.

Cacodyl cyanide is a highly toxic organoarsenic compound discovered by Robert Bunsen in the 1840s. It is very volatile and flammable, as it shares the chemical properties of both arsenic and cyanide.
















