Deoxycorticosterone (DOC), or desoxycorticosterone, may refer to:

Progestogens, also sometimes written progestagens or gestagens, are a class of natural or synthetic steroid hormones that bind to and activate the progesterone receptors (PR). Progesterone is the major and most important progestogen in the body. The progestogens are named for their function in maintaining pregnancy, although they are also present at other phases of the estrous and menstrual cycles.
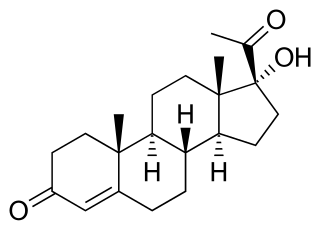
17α-Hydroxyprogesterone (17α-OHP), also known as 17-OH progesterone (17-OHP), or hydroxyprogesterone (OHP), is an endogenous progestogen steroid hormone related to progesterone. It is also a chemical intermediate in the biosynthesis of many other endogenous steroids, including androgens, estrogens, glucocorticoids, and mineralocorticoids, as well as neurosteroids.

Apparent mineralocorticoid excess is an autosomal recessive disorder causing hypertension, hypernatremia and hypokalemia. It results from mutations in the HSD11B2 gene, which encodes the kidney isozyme of 11β-hydroxysteroid dehydrogenase type 2. In an unaffected individual, this isozyme inactivates circulating cortisol to the less active metabolite cortisone. The inactivating mutation leads to elevated local concentrations of cortisol in the aldosterone sensitive tissues like the kidney. Cortisol at high concentrations can cross-react and activate the mineralocorticoid receptor due to the non-selectivity of the receptor, leading to aldosterone-like effects in the kidney. This is what causes the hypokalemia, hypertension, and hypernatremia associated with the syndrome. Patients often present with severe hypertension and end-organ changes associated with it like left ventricular hypertrophy, retinal, renal and neurological vascular changes along with growth retardation and failure to thrive. In serum both aldosterone and renin levels are low.

11-Deoxycorticosterone (DOC), or simply deoxycorticosterone, also known as 21-hydroxyprogesterone, as well as desoxycortone (INN), deoxycortone, and cortexone, is a steroid hormone produced by the adrenal gland that possesses mineralocorticoid activity and acts as a precursor to aldosterone. It is an active (Na+-retaining) mineralocorticoid. As its names indicate, 11-deoxycorticosterone can be understood as the 21-hydroxy-variant of progesterone or as the 11-deoxy-variant of corticosterone.

Aldosterone synthase, also called steroid 18-hydroxylase, corticosterone 18-monooxygenase or P450C18, is a steroid hydroxylase cytochrome P450 enzyme involved in the biosynthesis of the mineralocorticoid aldosterone and other steroids. The enzyme catalyzes sequential hydroxylations of the steroid angular methyl group at C18 after initial 11β-hydroxylation. It is encoded by the CYP11B2 gene in humans.
Deoxycortisol may refer to:

Steroid 11β-hydroxylase, also known as steroid 11β-monooxygenase, is a steroid hydroxylase found in the zona glomerulosa and zona fasciculata of the adrenal cortex. Named officially the cytochrome P450 11B1, mitochondrial, it is a protein that in humans is encoded by the CYP11B1 gene. The enzyme is involved in the biosynthesis of adrenal corticosteroids by catalyzing the addition of hydroxyl groups during oxidation reactions.

Formebolone, also known as formyldienolone, as well as 2-formyl-11α-hydroxy-17α-methyl-δ1-testosterone, is an orally active anabolic-androgenic steroid (AAS) described as an anticatabolic and anabolic drug that is or has been marketed in Spain and Italy. As an AAS, it shows some anabolic activity, though it is inferior to testosterone in terms of potency, but is said to have virtually no androgenic activity. Formebolone counteracts the catabolic effects of potent glucocorticoids like dexamethasone phosphate. A close analogue, roxibolone, shows similar antiglucocorticoid activity to formebolone but, in contrast, is devoid of activity as an AAS.

Roxibolone (INN), also known as 11β,17β-dihydroxy-17α-methyl-3-oxoandrosta-1,4-diene-2-carboxylic acid, is a steroidal antiglucocorticoid described as an anticholesterolemic (cholesterol-lowering) and anabolic drug which was never marketed. Roxibolone is closely related to formebolone, which shows antiglucocorticoid activity similarly and, with the exception of having a carboxaldehyde group at the C2 position instead of a carboxylic acid group, roxibolone is structurally almost identical to. The 2-decyl ester of roxibolone, decylroxibolone, is a long-acting prodrug of roxibolone with similar activity.
The molecular formula C21H30O3 (molar mass: 330.46 g/mol, exact mass: 330.2195 u) may refer to:
Dihydroprogesterone may refer to:
Hydroxyprogesterone (OHP) may refer to:

20α-Dihydroprogesterone (20α-DHP), also known as 20α-hydroxyprogesterone (20α-OHP), is a naturally occurring, endogenous progestogen. It is a metabolite of progesterone, formed by the 20α-hydroxysteroid dehydrogenases (20α-HSDs) AKR1C1, AKR1C2, and AKR1C3 and the 17β-hydroxysteroid dehydrogenase (17β-HSD) HSD17B1. 20α-DHP can be transformed back into progesterone by 20α-HSDs and by the 17β-HSD HSD17B2. HSD17B2 is expressed in the human endometrium and cervix among other tissues. In animal studies, 20α-DHP has been found to be selectively taken up into and retained in target tissues such as the uterus, brain, and skeletal muscle.

21-Deoxycortisol, also known as 11β,17α-dihydroxyprogesterone or as 11β,17α-dihydroxypregn-4-ene-3,20-dione, is a naturally occurring, endogenous steroid related to cortisol (11β,17α,21-trihydroxyprogesterone) which is formed as a metabolite from 17α-hydroxyprogesterone via 11β-hydroxylase.

11β-Hydroxyprogesterone (11β-OHP), also known as 21-deoxycorticosterone, as well as 11β-hydroxypregn-4-ene-3,20-dione, is a naturally occurring, endogenous steroid and derivative of progesterone. It is a potent mineralocorticoid. Syntheses of 11β-OHP from progesterone is catalyzed by the steroid 11β-hydroxylase (CYP11B1) enzyme, and, to a lesser extent, by the aldosterone synthase enzyme (CYP11B2).
11-Hydroxyprogesterone (11-OHP) may refer to:

21-Hydroxypregnenolone, also known as prebediolone, as well as 3β,21-dihydroxypregn-5-en-20-one, is a naturally occurring and endogenous pregnane steroid, and an intermediate in the biosynthesis of 11-deoxycorticosterone (21-hydroxyprogesterone), corticosterone (11β,21-dihydroxyprogesterone), and other corticosteroids. It is formed from pregnenolone in the adrenal glands.

16α-Hydroxyprogesterone (16α-OHP), also known as 16α-hydroxypregn-4-ene-3,20-dione, is a minor endogenous progestogen steroid hormone and a metabolite of progesterone that is formed in lower amounts than 17α-hydroxyprogesterone (17α-OHP). It occurs in micromolar concentrations and its physiological relevance hence is questionable. However, it may accumulate in target tissues and could have a physiological role in the reproductive system and mammary gland development as well as the cardiovascular and central nervous systems.

20β-Dihydroprogesterone (20β-DHP), also known as 20β-hydroxyprogesterone (20β-OHP), is an endogenous metabolite of progesterone which is formed by 20β-hydroxysteroid dehydrogenase (20β-HSD). It is a progestogen similarly to progesterone, with about 20 to 50% of the progestogenic activity of progesterone. It can be converted by 20β-HSD into progesterone in the uterus. The effects of 20β-HSD on the uterus, mammary glands, and in maintaining pregnancy have been studied. The progestogenic activity of 20β-HSD has also been characterized in women.