Lactate may refer to:
- Lactation, the secretion of milk from the mammary glands
- Lactate, the conjugate base of lactic acid
Lactate may refer to:
Pyruvic acid (IUPAC name: 2-oxopropanoic acid, also called acetoic acid) (CH3COCOOH) is the simplest of the alpha-keto acids, with a carboxylic acid and a ketone functional group. Pyruvate, the conjugate base, CH3COCOO−, is an intermediate in several metabolic pathways throughout the cell.

Long-distance running, or endurance running, is a form of continuous running over distances of at least 3 km (1.9 mi). Physiologically, it is largely aerobic in nature and requires stamina as well as mental strength.
Anaerobic means "living, active, occurring, or existing in the absence of free oxygen", as opposed to aerobic which means "living, active, or occurring only in the presence of oxygen." Anaerobic may also refer to:
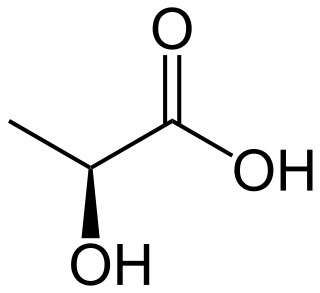
Lactic acid is an organic acid. It has the molecular formula C3H6O3. It is white in the solid state and it is miscible with water. When in the dissolved state, it forms a colorless solution. Production includes both artificial synthesis as well as natural sources. Lactic acid is an alpha-hydroxy acid (AHA) due to the presence of a hydroxyl group adjacent to the carboxyl group. It is used as a synthetic intermediate in many organic synthesis industries and in various biochemical industries. The conjugate base of lactic acid is called lactate (or the lactate anion). The name of the derived acyl group is lactoyl.

Lactic acidosis is a medical condition characterized by a build-up of lactate in the body, with formation of an excessively low pH in the bloodstream. It is a form of metabolic acidosis, in which excessive acid accumulates due to a problem with the body's oxidative metabolism.
The Dietary Reference Intake (DRI) is a system of nutrition recommendations from the National Academy of Medicine (NAM) of the National Academies. It was introduced in 1997 in order to broaden the existing guidelines known as Recommended Dietary Allowances. The DRI values differ from those used in nutrition labeling on food and dietary supplement products in the U.S. and Canada, which uses Reference Daily Intakes (RDIs) and Daily Values (%DV) which were based on outdated RDAs from 1968 but were updated as of 2016.

Anaerobic exercise is a type of exercise that breaks down glucose in the body without using oxygen; anaerobic means "without oxygen". In practical terms, this means that anaerobic exercise is more intense, but shorter in duration than aerobic exercise. This type of exercise leads to a buildup of lactic acid.

Ringer's lactate solution (RL), also known as sodium lactate solution,Lactated Ringer's, and Hartmann's solution, is a mixture of sodium chloride, sodium lactate, potassium chloride, and calcium chloride in water. It is used for replacing fluids and electrolytes in those who have low blood volume or low blood pressure. It may also be used to treat metabolic acidosis and to wash the eye following a chemical burn. It is given by intravenous infusion or applied to the affected area.

Calcium lactate is a white crystalline salt with formula C
6H
10CaO
6, consisting of two lactate anions H
3C(CHOH)CO−
2 for each calcium cation Ca2+
. It forms several hydrates, the most common being the pentahydrate C
6H
10CaO
6·5H
2O.
Lactate inflection point (LIP) is the exercise intensity at which the blood concentration of lactate and/or lactic acid begins to increase rapidly. It is often expressed as 85% of maximum heart rate or 75% of maximum oxygen intake. When exercising at or below the lactate threshold, any lactate produced by the muscles is removed by the body without it building up.
In enzymology, a D-lactate dehydrogenase (cytochrome) is an enzyme that catalyzes the chemical reaction
In enzymology, a D-lactate dehydrogenase is an enzyme that catalyzes the chemical reaction

Lactate dehydrogenase A (LDHA) is an enzyme which in humans is encoded by the LDHA gene. It is a monomer of Lactate dehydrogenase, which exists as a tetramer. The other main subunit is lactate dehydrogenase B (LDHB).

Sodium lactate is the sodium salt of lactic acid, and has a mild saline taste. It is produced by fermentation of a sugar source, such as corn or beets, and then, by neutralizing the resulting lactic acid to create a compound having the formula NaC3H5O3.

Hydroxycarboxylic acid receptor 1 (HCA1), formerly known as G protein-coupled receptor 81 (GPR81), is a protein that in humans is encoded by the HCAR1 gene. HCA1, like the other hydroxycarboxylic acid receptors HCA2 and HCA3, is a Gi/o-coupled G protein-coupled receptor (GPCR). The primary endogenous agonist of HCA1 is lactic acid (and its conjugate base, lactate). More recently, 3,5-dihydroxybenzoic acid has been reported to activate HCA1.

Lactate dehydrogenase (LDH or LD) is an enzyme found in nearly all living cells. LDH catalyzes the conversion of pyruvate to lactate and back, as it converts NAD+ to NADH and back. A dehydrogenase is an enzyme that transfers a hydride from one molecule to another.

Monocarboxylate transporter 1 is a ubiquitous protein that in humans is encoded by the SLC16A1 gene. It is a proton coupled monocarboxylate transporter.
Let Down may refer to:
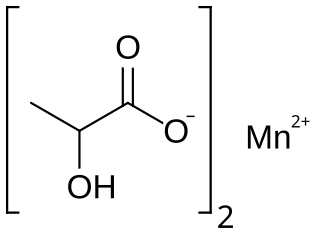
Manganese lactate is an organic chemical compound, a salt of manganese and lactic acid with the formula Mn(C3H5O3)2. The compound forms light pink crystals, soluble in water, forming crystalline hydrates.

Aluminium lactate is a chemical compound, a salt of aluminium and lactic acid with the formula Al(C3H5O3)3.