Related Research Articles

In chemistry, a salt is a solid chemical compound consisting of an ionic assembly of cations and anions. Salts are composed of related numbers of cations and anions so that the product is electrically neutral. These component ions can be inorganic, such as chloride (Cl−), or organic, such as acetate ; and can be monatomic, such as fluoride (F−) or polyatomic, such as sulfate.
A half reaction is either the oxidation or reduction reaction component of a redox reaction. A half reaction is obtained by considering the change in oxidation states of individual substances involved in the redox reaction.
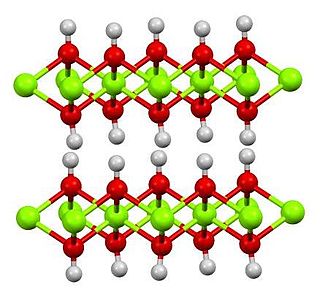
Magnesium hydroxide is the inorganic compound with the chemical formula Mg(OH)2. It occurs in nature as the mineral brucite. It is a white solid with low solubility in water (Ksp = 5.61×10−12). Magnesium hydroxide is a common component of antacids, such as milk of magnesia, as well as laxatives.

The lead–acid battery was invented in 1859 by French physicist Gaston Planté and is the earliest type of rechargeable battery. Despite having a very low energy-to-weight ratio and a low energy-to-volume ratio, its ability to supply high surge currents means that the cells have a relatively large power-to-weight ratio. These features, along with their low cost, make them attractive for use in motor vehicles to provide the high current required by automobile starter motors.
Lead(II) nitrate is an inorganic compound with the chemical formula Pb(NO3)2. It commonly occurs as a colourless crystal or white powder and, unlike most other lead(II) salts, is soluble in water.

Manganese(IV) oxide is the inorganic compound with the formula MnO
2. This blackish or brown solid occurs naturally as the mineral pyrolusite, which is the main ore of manganese and a component of manganese nodules. The principal use for MnO
2 is for dry-cell batteries, such as the alkaline battery and the zinc-carbon battery. MnO
2 is also used as a pigment and as a precursor to other manganese compounds, such as KMnO
4. It is used as a reagent in organic synthesis, for example, for the oxidation of allylic alcohols. MnO
2 in the α polymorph can incorporate a variety of atoms in the "tunnels" or "channels" between the manganese oxide octahedra. There is considerable interest in α-MnO
2 as a possible cathode for lithium ion batteries.

Lead(II) sulfate (PbSO4) is a white solid, which appears white in microcrystalline form. It is also known as fast white, milk white, sulfuric acid lead salt or anglesite.

Lead(II) chloride (PbCl2) is an inorganic compound which is a white solid under ambient conditions. It is poorly soluble in water. Lead(II) chloride is one of the most important lead-based reagents. It also occurs naturally in the form of the mineral cotunnite.

Copper(II) oxide or cupric oxide is the inorganic compound with the formula CuO. A black solid, it is one of the two stable oxides of copper, the other being Cu2O or cuprous oxide. As a mineral, it is known as tenorite. It is a product of copper mining and the precursor to many other copper-containing products and chemical compounds.

Lead(II,IV) oxide, also called red lead or minium, is the inorganic compound with the formula Pb3O4. A bright red or orange solid, it is used as pigment, in the manufacture of batteries, and rustproof primer paints. It is an example of a mixed valence compound, being composed of both Pb(II) and Pb(IV) in the ratio of two to one.

Lead(IV) oxide is the inorganic compound with the formula PbO2. It is an oxide where lead is in an oxidation state of +4. It is a dark-brown solid which is insoluble in water. It exists in two crystalline forms. It has several important applications in electrochemistry, in particular as the positive plate of lead acid batteries.
Lead(II) hydroxide, Pb(OH)2, is a hydroxide of lead, with lead in oxidation state +2. It is doubtful that such a simple compound exists. Lead basic carbonate (PbCO3·2Pb(OH)2) or lead(II) oxide (PbO) is encountered in practice where lead hydroxide is expected. This has been a subject of considerable confusion in the past.
Calcium hypochlorite is an inorganic compound with formula Ca(ClO)2. It is the main active ingredient of commercial products called bleaching powder, chlorine powder, or chlorinated lime, used for water treatment and as a bleaching agent. This compound is relatively stable and has greater available chlorine than sodium hypochlorite (liquid bleach). It is a white solid, although commercial samples appear yellow. It strongly smells of chlorine, owing to its slow decomposition in moist air. It is not highly soluble in hard water, and is more preferably used in soft to medium-hard water. It has two forms: dry (anhydrous); and hydrated (hydrous).
Iron(III) oxide-hydroxide or ferric oxyhydroxide is the chemical compound of iron, oxygen, and hydrogen with formula FeO(OH).

Antimony oxychloride, known since the 15th century, has been known by a plethora of alchemical names. Since the compound functions as both an emetic and a laxative, it was originally used as a purgative.

Lead(II) chromate (PbCrO4) is a chemical compound, a chromate of lead. It has a vivid yellow color and is insoluble in water, and as a result, is used in paints under the name chrome yellow.

Dicopper chloride trihydroxide is the chemical compound with the formula Cu2(OH)3Cl. It is often referred to as tribasic copper chloride (TBCC), copper trihydroxyl chloride or copper hydroxychloride. It is a greenish crystalline solid encountered in mineral deposits, metal corrosion products, industrial products, art and archeological objects, and some living systems. It was originally manufactured on an industrial scale as a precipitated material used as either a chemical intermediate or a fungicide. Since 1994, a purified, crystallized product has been produced at the scale of thousands of tons per year, and used extensively as a nutritional supplement for animals.
Compounds of lead exist in two main oxidation states: +2 and +4. The former is more common. Inorganic lead(IV) compounds are typically strong oxidants or exist only in highly acidic solutions.
Wiklundite is a rare and complex arsenite-silicate mineral with the formula Pb2(Mn2+,Zn)3(Fe3+,Mn2+)2(Mn2+,Mg)19(As3+O3)2(Si,As5+O4)6(OH)18Cl6. The mineral characterizes in a large c unit cell parameter. It was found in Långban, Sweden - a home for many rare and exotic minerals.