Related Research Articles
In chemical analysis, chromatography is a laboratory technique for the separation of a mixture into its components. The mixture is dissolved in a fluid solvent called the mobile phase, which carries it through a system on which a material called the stationary phase is fixed. Because the different constituents of the mixture tend to have different affinities for the stationary phase and are retained for different lengths of time depending on their interactions with its surface sites, the constituents travel at different apparent velocities in the mobile fluid, causing them to separate. The separation is based on the differential partitioning between the mobile and the stationary phases. Subtle differences in a compound's partition coefficient result in differential retention on the stationary phase and thus affect the separation.

Size-exclusion chromatography (SEC), also known as molecular sieve chromatography, is a chromatographic method in which molecules in solution are separated by their size, and in some cases molecular weight. It is usually applied to large molecules or macromolecular complexes such as proteins and industrial polymers. Typically, when an aqueous solution is used to transport the sample through the column, the technique is known as gel-filtration chromatography, versus the name gel permeation chromatography, which is used when an organic solvent is used as a mobile phase. The chromatography column is packed with fine, porous beads which are composed of dextran polymers (Sephadex), agarose (Sepharose), or polyacrylamide. The pore sizes of these beads are used to estimate the dimensions of macromolecules. SEC is a widely used polymer characterization method because of its ability to provide good molar mass distribution (Mw) results for polymers.

High-performance liquid chromatography (HPLC), formerly referred to as high-pressure liquid chromatography, is a technique in analytical chemistry used to separate, identify, and quantify each component in a mixture. It relies on pumps to pass a pressurized liquid solvent containing the sample mixture through a column filled with a solid adsorbent material. Each component in the sample interacts slightly differently with the adsorbent material, causing different flow rates for the different components and leading to the separation of the components as they flow out of the column.
Gel permeation chromatography (GPC) is a type of size-exclusion chromatography (SEC), that separates analytes on the basis of size, typically in organic solvents. The technique is often used for the analysis of polymers. As a technique, SEC was first developed in 1955 by Lathe and Ruthven. The term gel permeation chromatography can be traced back to J.C. Moore of the Dow Chemical Company who investigated the technique in 1964. The proprietary column technology was licensed to Waters Corporation, who subsequently commercialized this technology in 1964. GPC systems and consumables are now also available from a number of manufacturers. It is often necessary to separate polymers, both to analyze them as well as to purify the desired product.
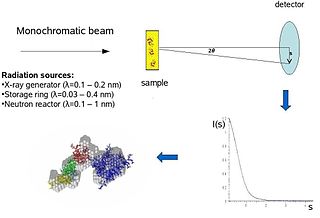
Biological small-angle scattering is a small-angle scattering method for structure analysis of biological materials. Small-angle scattering is used to study the structure of a variety of objects such as solutions of biological macromolecules, nanocomposites, alloys, and synthetic polymers. Small-angle X-ray scattering (SAXS) and small-angle neutron scattering (SANS) are the two complementary techniques known jointly as small-angle scattering (SAS). SAS is an analogous method to X-ray and neutron diffraction, wide angle X-ray scattering, as well as to static light scattering. In contrast to other X-ray and neutron scattering methods, SAS yields information on the sizes and shapes of both crystalline and non-crystalline particles. When used to study biological materials, which are very often in aqueous solution, the scattering pattern is orientation averaged.
The molar mass distribution describes the relationship between the number of moles of each polymer species (Ni) and the molar mass (Mi) of that species. In linear polymers, the individual polymer chains rarely have exactly the same degree of polymerization and molar mass, and there is always a distribution around an average value. The molar mass distribution of a polymer may be modified by polymer fractionation.
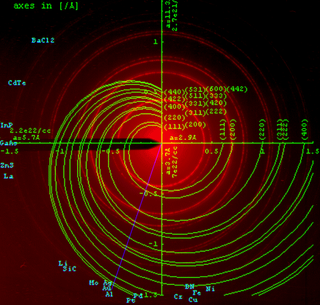
Powder diffraction is a scientific technique using X-ray, neutron, or electron diffraction on powder or microcrystalline samples for structural characterization of materials. An instrument dedicated to performing such powder measurements is called a powder diffractometer.
Elastic recoil detection analysis (ERDA), also referred to as forward recoil scattering, is an ion beam analysis technique in materials science to obtain elemental concentration depth profiles in thin films. This technique is known by several different names. These names are listed below. In the technique of ERDA, an energetic ion beam is directed at a sample to be characterized and there is an elastic nuclear interaction between the ions of beam and the atoms of the target sample. Such interactions are commonly of Coulomb nature. Depending on the kinetics of the ions, cross section area, and the loss of energy of the ions in the matter, ERDA helps determine the quantification of the elemental analysis. It also provides information about the depth profile of the sample.
A particle counter is used for monitoring and diagnosing particle contamination within specific clean media, including air, water and chemicals. Particle counters are used in a variety of applications in support of clean manufacturing practices, industries include: electronic components and assemblies, pharmaceutical drug products and medical devices, and industrial technologies such as oil and gas.

Dynamic light scattering (DLS) is a technique in physics that can be used to determine the size distribution profile of small particles in suspension or polymers in solution. In the scope of DLS, temporal fluctuations are usually analyzed using the intensity or photon auto-correlation function. In the time domain analysis, the autocorrelation function (ACF) usually decays starting from zero delay time, and faster dynamics due to smaller particles lead to faster decorrelation of scattered intensity trace. It has been shown that the intensity ACF is the Fourier transform of the power spectrum, and therefore the DLS measurements can be equally well performed in the spectral domain. DLS can also be used to probe the behavior of complex fluids such as concentrated polymer solutions.
Static light scattering is a technique in physical chemistry that measures the intensity of the scattered light to obtain the average molecular weight Mw of a macromolecule like a polymer or a protein in solution. Measurement of the scattering intensity at many angles allows calculation of the root mean square radius, also called the radius of gyration Rg. By measuring the scattering intensity for many samples of various concentrations, the second virial coefficient, A2, can be calculated.

Particle size analysis, particle size measurement, or simply particle sizing, is the collective name of the technical procedures, or laboratory techniques which determines the size range, and/or the average, or mean size of the particles in a powder or liquid sample.
Absolute molar mass is a process used to determine the characteristics of molecules.

Field-flow fractionation, abbreviated FFF, is a separation technique which does not have a stationary phase. It is similar to liquid chromatography as it works on dilute solutions or suspensions of the solute. Separation is achieved by applying a field perpendicular to the direction of transport of the sample which is pumped through a long and narrow channel. The field exerts a force on the sample components concentrating them towards one of the channel walls, which is called accumulation wall. The force interacts with a property of the sample on which then the separation occurs, in other words on their differing "mobilities" under the force exerted by the field. As an example, for the hydraulic, or cross-flow FFF method, the property driving separation is the translational diffusion coefficient or the hydrodynamic size. For a thermal field, it is the ratio of the thermal and the translational diffusion coefficient.
Polymer characterization is the analytical branch of polymer science.
Multiangle light scattering (MALS) describes a technique for measuring the light scattered by a sample into a plurality of angles. It is used for determining both the absolute molar mass and the average size of molecules in solution, by detecting how they scatter light. A collimated beam from a laser source is most often used, in which case the technique can be referred to as multiangle laser light scattering (MALLS). The insertion of the word laser was intended to reassure those used to making light scattering measurements with conventional light sources, such as Hg-arc lamps that low-angle measurements could now be made. Until the advent of lasers and their associated fine beams of narrow width, the width of conventional light beams used to make such measurements prevented data collection at smaller scattering angles. In recent years, since all commercial light scattering instrumentation use laser sources, this need to mention the light source has been dropped and the term MALS is used throughout.
Brookhaven Instruments Corporation is a Nova Instruments company established in the late 1960s. Brookhaven Instruments designed modern techniques in characterizing nanoparticles, proteins, and polymers using light scattering techniques such as dynamic, static, electrophoretic, and phase analysis for: particle size, zeta potential, molecular mass, and absolute molar mass analysis.

Polymer Char is a company which designs and manufactures instrumentation for polymer analysis.

Asymmetrical flow field-flow fractionation (AF4) is most versatile and most widely used sub-technique within the family of field flow fractionation (FFF) methods. AF4 can be used in aqueous and organic solvents and is able to characterize nanoparticles, polymers and proteins. The theory for AF4 was conceived in 1986 and was established in 1987 and first published by Wahlund and Giddings. AF4 is distinct from symmetrical Flow FFF because it contains only one permeable wall so the cross-flow is caused only by the carrier liquid. The cross-flow is induced by the carrier liquid constantly exiting by way of the semi-permeable wall on the bottom of the channel.

The characterization of nanoparticles is a branch of nanometrology that deals with the characterization, or measurement, of the physical and chemical properties of nanoparticles. Nanoparticles measure less than 100 nanometers in at least one of their external dimensions, and are often engineered for their unique properties. Nanoparticles are unlike conventional chemicals in that their chemical composition and concentration are not sufficient metrics for a complete description, because they vary in other physical properties such as size, shape, surface properties, crystallinity, and dispersion state.