
Manganese is a chemical element with the symbol Mn and atomic number 25. It is not found as a free element in nature; it is often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy uses, particularly in stainless steels.

The pyroxenes (commonly abbreviated to Px) are a group of important rock-forming inosilicate minerals found in many igneous and metamorphic rocks. Pyroxenes have the general formula XY(Si,Al)2O6, where X represents calcium, sodium, iron (II) or magnesium and more rarely zinc, manganese or lithium, and Y represents ions of smaller size, such as chromium, aluminium, iron (III), magnesium, cobalt, manganese, scandium, titanium, vanadium or even iron (II). Although aluminium substitutes extensively for silicon in silicates such as feldspars and amphiboles, the substitution occurs only to a limited extent in most pyroxenes. They share a common structure consisting of single chains of silica tetrahedra. Pyroxenes that crystallize in the monoclinic system are known as clinopyroxenes and those that crystallize in the orthorhombic system are known as orthopyroxenes.

Axinite is a brown to violet-brown, or reddish-brown bladed group of minerals composed of calcium aluminium boro-silicate, (Ca,Fe,Mn)3Al2BO3Si4O12OH. Axinite is pyroelectric and piezoelectric.

Manganese(IV) oxide is the inorganic compound with the formula MnO
2. This blackish or brown solid occurs naturally as the mineral pyrolusite, which is the main ore of manganese and a component of manganese nodules. The principal use for MnO
2 is for dry-cell batteries, such as the alkaline battery and the zinc-carbon battery. MnO
2 is also used as a pigment and as a precursor to other manganese compounds, such as KMnO
4. It is used as a reagent in organic synthesis, for example, for the oxidation of allylic alcohols. MnO
2 is α polymorph that can incorporate a variety of atoms in the "tunnels" or "channels" between the manganese oxide octahedra. There is considerable interest in α-MnO
2 as a possible cathode for lithium ion batteries.

Rhodonite is a manganese inosilicate, (Mn, Fe, Mg, Ca)SiO3 and member of the pyroxenoid group of minerals, crystallizing in the triclinic system. It commonly occurs as cleavable to compact masses with a rose-red color (the name comes from the Greek ῥόδος rhodos, rosy), often tending to brown because of surface oxidation.

Cummingtonite is a metamorphic amphibole with the chemical composition (Mg,Fe2+
)
2(Mg,Fe2+
)
5Si
8O
22(OH)
2, magnesium iron silicate hydroxide.

Tephroite is the manganese endmember of the olivine group of nesosilicate minerals with the formula Mn2SiO4. A solid solution series exists between tephroite and its analogues, the group endmembers fayalite and forsterite. Divalent iron or magnesium may readily replace manganese in the olivine crystal structure.
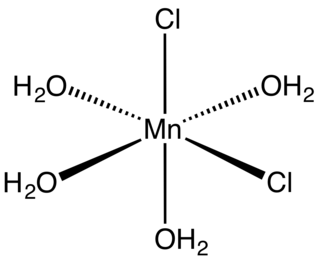
Manganese(II) chloride is the dichloride salt of manganese, MnCl2. This inorganic chemical exists in the anhydrous form, as well as the dihydrate (MnCl2·2H2O) and tetrahydrate (MnCl2·4H2O), with the tetrahydrate being the most common form. Like many Mn(II) species, these salts are pink, with the paleness of the color being characteristic of transition metal complexes with high spin d5 configurations.

Manganese(III) fluoride (also known as Manganese trifluoride) is the inorganic compound with the formula MnF3. This red/purplish solid is useful for converting hydrocarbons into fluorocarbons, i.e., it is a fluorination agent. It forms a hydrate and many derivatives.

Braunite is a silicate mineral containing both di- and tri-valent manganese with the chemical formula: Mn2+Mn3+6[O8|SiO4]. Common impurities include iron, calcium, boron, barium, titanium, aluminium, and magnesium.

Todorokite is a rare complex hydrous manganese oxide mineral with the formula (Na,Ca,K,Ba,Sr)1-x(Mn,Mg,Al)6O12·3-4H2O. It was named in 1934 for the type locality, the Todoroki mine, Hokkaido, Japan. It belongs to the prismatic class 2/m of the monoclinic crystal system, but the angle β between the a and c axes is close to 90°, making it seem orthorhombic. It is a brown to black mineral which occurs in massive or tuberose forms. It is quite soft with a Mohs hardness of 1.5, and a specific gravity of 3.49 - 3.82. It is a component of deep ocean basin manganese nodules.
Alloy steel is steel that is alloyed with a variety of elements in total amounts between 1.0% and 50% by weight to improve its mechanical properties. Alloy steels are broken down into two groups: low alloy steels and high alloy steels. The difference between the two is disputed. Smith and Hashemi define the difference at 4.0%, while Degarmo, et al., define it at 8.0%. Most commonly, the phrase "alloy steel" refers to low-alloy steels.

Manganese tetrafluoride, MnF4, is the highest fluoride of manganese. It is a powerful oxidizing agent and is used as a means of purifying elemental fluorine.

Bustamite is a calcium manganese inosilicate (chain silicate) and a member of the wollastonite group. Magnesium, zinc and iron are common impurities substituting for manganese. Bustamite is the high-temperature polymorph of CaMnSi2O6 and johannsenite is the low temperature polymorph. The inversion takes place at 830 °C (1,530 °F), but may be very slow.
Bustamite could be confused with light-colored rhodonite or pyroxmangite, but both these minerals are biaxial (+) whereas bustamite is biaxial (-).
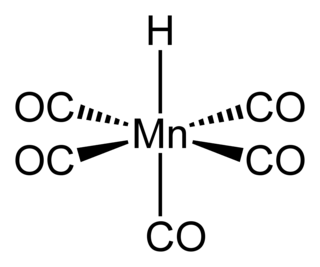
Pentacarbonylhydridomanganese is an organometallic compound with formula HMn(CO)5. This compound is one of the most stable "first-row" transition metal hydrides.

Manganvesuvianite is a rare mineral with formula Ca19Mn3+(Al,Mn3+,Fe3+)10(Mg,Mn2+)2(Si2O7)4(SiO4)10O(OH)9. The mineral is red to nearly black in color. Discovered in South Africa and described in 2002, it was so named for the prevalence of manganese in its composition and its relation to vesuvianite.

Manganese germanide (MnGe) is an intermetallic compound, a germanide of manganese. Its crystals have a cubic symmetry with no inversion center, they are therefore helical, with right-hand and left-handed chiralities.

Manganese disilicide (MnSi2) is an intermetallic compound, a silicide of manganese. It is a non-stoichiometric compound, with a silicon defficiency expressed as MnSi2–x. Crystal structures of many MnSi2–x compounds resemble a chimney ladder and are called Nowotny phases. They include MnSi2 (x=0), Mn4Si7 (x=0.250), Mn11Si19 (x=0.273), Mn15Si26 (x=0.267) and Mn27Si47 (x=0.259). These phases have very similar unit cells whose length varies from 1.75 nm for MnSi2 or Mn4Si7, which have almost the same structures, to 11.8 nm for Mn27Si47.

Manganese monosilicide (MnSi) is an intermetallic compound, a silicide of manganese. It occurs in cosmic dust as the mineral brownleeite. MnSi has a cubic crystal lattice with no inversion center; therefore its crystal structure is helical, with right-hand and left-hand chiralities.

















