Mercury hydride may refer to:
- Mercury(I) hydride (HgH or Hg2H2), an extremely unstable gas
- Mercury(II) hydride (HgH2), a volatile but relatively stable white solid
Mercury hydride may refer to:
Hg is the symbol of chemical element mercury.
In organic chemistry, the oxymercuration reaction is an electrophilic addition reaction that transforms an alkene into a neutral alcohol. In oxymercuration, the alkene reacts with mercuric acetate in aqueous solution to yield the addition of an acetoxymercury group and a hydroxy group across the double bond. Carbocations are not formed in this process and thus rearrangements are not observed. The reaction follows Markovnikov's rule. The oxymercuration part of the reaction involves anti addition of OH group but the demercuration part of the reaction involves free radical mechanism and is not stereospecific, i.e. H and OH may be syn or anti to each other.
Mercury chloride can refer to:

Mercury(II) oxide, also called mercuric oxide or simply mercury oxide, is the inorganic compound with the formula HgO. It has a red or orange color. Mercury(II) oxide is a solid at room temperature and pressure. The mineral form montroydite is very rarely found.
Mercury oxide can refer to:
Aluminium can form an amalgam in solution with mercury. Aluminium amalgam may be prepared by either grinding aluminium pellets or wire in mercury, or by allowing aluminium wire to react with a solution of mercury(II) chloride in water.
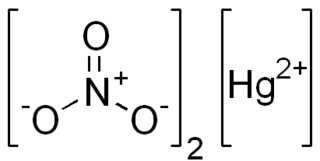
Mercury(II) nitrate is an inorganic compound with the chemical formula Hg(NO3)2. It is the mercury(II) salt of nitric acid HNO3. It contains mercury(II) cations Hg2+ and nitrate anions NO−3, and water of crystallization H2O in the case of a hydrous salt. Mercury(II) nitrate forms hydrates Hg(NO3)2·xH2O. Anhydrous and hydrous salts are colorless or white soluble crystalline solids that are occasionally used as a reagents. Mercury(II) nitrate is made by treating mercury with hot concentrated nitric acid. Neither anhydrous nor monohydrate has been confirmed by X-ray crystallography. The anhydrous material is more widely used.

Mercury(II) thiocyanate is an inorganic chemical compound, the coordination complex of Hg2+ and the thiocyanate anion. It is a white powder. It will produce a large, winding "snake" when ignited, an effect known as the Pharaoh's serpent.

Aluminium hydride is an inorganic compound with the formula AlH3. Alane and its derivatives are part of a family of common reducing reagents in organic synthesis based around group 13 hydrides. In solution—typically in ethereal solvents such tetrahydrofuran or diethyl ether—aluminium hydride forms complexes with Lewis bases, and reacts selectively with particular organic functional groups, and although it is not a reagent of choice, it can react with carbon-carbon multiple bonds. Given its density, and with hydrogen content on the order of 10% by weight, some forms of alane are, as of 2016, active candidates for storing hydrogen and so for power generation in fuel cell applications, including electric vehicles. As of 2006 it was noted that further research was required to identify an efficient, economical way to reverse the process, regenerating alane from spent aluminium product.
Mercury iodide may refer to the following chemical compounds:
Mercury sulfate may refer to:
HGF may refer to:
Mercury fluoride can refer to:
HGH stands for human growth hormone.

Mercury is a chemical element; it has symbol Hg and atomic number 80. It is also known as quicksilver and was formerly named hydrargyrum from the Greek words hydor'water' and argyros'silver', from which its chemical symbol is derived. A heavy, silvery d-block element, mercury is the only metallic element that is known to be liquid at standard temperature and pressure; the only other element that is liquid under these conditions is the halogen bromine, though metals such as caesium, gallium, and rubidium melt just above room temperature.

An amalgam is an alloy of mercury with another metal. It may be a liquid, a soft paste or a solid, depending upon the proportion of mercury. These alloys are formed through metallic bonding, with the electrostatic attractive force of the conduction electrons working to bind all the positively charged metal ions together into a crystal lattice structure. Almost all metals can form amalgams with mercury, the notable exceptions being iron, platinum, tungsten, and tantalum. Silver-mercury amalgams are important in dentistry, and gold-mercury amalgam is used in the extraction of gold from ore. Dentistry has used alloys of mercury with metals such as silver, copper, indium, tin and zinc.
Mercury bromide can refer to:
Binary compounds of hydrogen are binary chemical compounds containing just hydrogen and one other chemical element. By convention all binary hydrogen compounds are called hydrides even when the hydrogen atom in it is not an anion. These hydrogen compounds can be grouped into several types.
Mercury(I) hydride is an inorganic compound with the chemical formula HgH. It has not yet been obtained in bulk, hence its bulk properties remain unknown. However, molecular mercury(I) hydrides with the formulae HgH and Hg
2H
2 have been isolated in solid gas matrices. The molecular hydrides are very unstable toward thermal decomposition. As such the compound is not well characterised, although many of its properties have been calculated via computational chemistry.

Mercury(II) hydride is an inorganic compound with the chemical formula HgH
2. It is both thermodynamically and kinetically unstable at ambient temperature, and as such, little is known about its bulk properties. However, it can also be a white, crystalline solid, which is kinetically stable at temperatures below −125 °C (−193 °F), which was synthesized for the first time in 1951.