Chemical properties
| Methoxytoluene Isomers | |||
|---|---|---|---|
| Common names | 2-Methoxytoluene 2-Methylanisole Ortho cresyl methyl ether | 3-Methoxytoluene 3-Methylanisole Meta cresyl methyl ether | 4-Methoxytoluene 4-Methylanisole Para cresyl methyl ether |
| Structure | 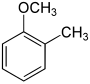 |  |  |
| PubChem number | [33637] | [7530] | [7731] |
| CAS number | [578-58-5] | [100-84-5] | [104-93-8] |
| Melting point | −34.1 °C (−31 °F; 238 K) | −47 °C (−52.6 °F; 226 K) | −23 °C (44.6 °F; 280 K) |
| Boiling point | 171 °C (318.2 °F; 432 K) | 175.5 °C (323.6 °F; 435 K) | 175.5 °C (323.6 °F; 435 K) |
| Density | 0.9798 g/cm3 | 0.9716 g/cm3 | 0.969 g/cm3 |