Related Research Articles

Molybdenum is a chemical element with the symbol Mo and atomic number 42. The name is from Neo-Latin molybdaenum, which is based on Ancient Greek Μόλυβδος molybdos, meaning lead, since its ores were confused with lead ores. Molybdenum minerals have been known throughout history, but the element was discovered in 1778 by Carl Wilhelm Scheele. The metal was first isolated in 1781 by Peter Jacob Hjelm.
Molybdenum trioxide is chemical compound with the formula MoO3. This compound is produced on the largest scale of any molybdenum compound. It is an intermediate in the production of molybdenum metal. It is also an important industrial catalyst. Molybdenum trioxide occurs as the rare mineral molybdite.
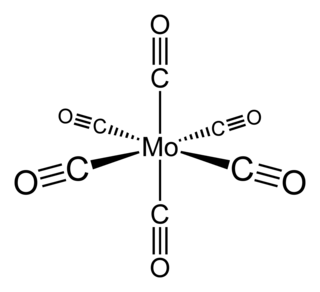
Molybdenum hexacarbonyl (also called molybdenum carbonyl) is the chemical compound with the formula Mo(CO)6. This colorless solid, like its chromium and tungsten analogues, is noteworthy as a volatile, air-stable derivative of a metal in its zero oxidation state.

Molybdic acid refers to hydrated forms of molybdenum trioxide and related species. The monohydrate (MoO3·H2O) and the dihydrate (MoO3·2H2O) are well characterized. They are yellow diamagnetic solids.

Chromium(III) oxide is an inorganic compound with the formula Cr
2O
3. It is one of the principal oxides of chromium and is used as a pigment. In nature, it occurs as the rare mineral eskolaite.

Sodium molybdate, Na2MoO4, is useful as a source of molybdenum. It is often found as the dihydrate, Na2MoO4·2H2O.
Nano-thermite or super-thermite is a metastable intermolecular composite (MICs) characterized by a particle size of its main constituents, a metal and a metal oxide, under 100 nanometers. This allows for high and customizable reaction rates. Nano-thermites contain an oxidizer and a reducing agent, which are intimately mixed on the nanometer scale. MICs, including nano-thermitic materials, are a type of reactive materials investigated for military use, as well as for general applications involving propellants, explosives, and pyrotechnics.

Molybdenum disilicide (MoSi2, or molybdenum silicide), an intermetallic compound, a silicide of molybdenum, is a refractory ceramic with primary use in heating elements. It has moderate density, melting point 2030 °C, and is electrically conductive. At high temperatures it forms a passivation layer of silicon dioxide, protecting it from further oxidation. The thermal stability of MoSi2 alongside its high emissivity make this material, alongside WSi2 attractive for applications as a high emissivity coatings in heat shields for atmospheric entry. MoSi2 is a gray metallic-looking material with tetragonal crystal structure (alpha-modification); its beta-modification is hexagonal and unstable. It is insoluble in most acids but soluble in nitric acid and hydrofluoric acid.

Molybdenum(V) chloride is the inorganic compound with the formula [MoCl5]2. This dark volatile solid is used in research to prepare other molybdenum compounds. It is moisture-sensitive and soluble in chlorinated solvents. Usually called molybdenum pentachloride, it is in fact a dimer with the formula Mo2Cl10.

Tungsten(IV) oxide is the chemical compound with the formula WO2. The bronze-colored solid crystallizes in a monoclinic cell. The rutile-like structure features distorted octahedral WO6 centers with alternate short W–W bonds (248 pm). Each tungsten center has the d2 configuration, which gives the material a high electrical conductivity.

Molybdenum dichloride describes chemical compounds with the empirical formula MoCl2. At least two forms are known, and both have attracted much attention from academic researchers because of the unexpected structures seen for these compounds and the fact that they give rise to hundreds of derivatives. The form discussed here is Mo6Cl12. The other molybdenum(II) chloride is potassium octachlorodimolybdate.
Molybdenum dioxide is the chemical compound with the formula MoO2. It is a violet-colored solid and is a metallic conductor. The mineralogical form of this compound is called tugarinovite, and is only very rarely found.

Molybdenum blue is a term applied to:
Ferro molybdenum is an important iron-molybdenum metal alloy, with a molybdenum content of 60-75% It is the main source for molybdenum alloying of HSLA steel.

Organomolybdenum chemistry is the chemistry of chemical compounds with Mo-C bonds. The heavier group 6 elements molybdenum and tungsten form organometallic compounds similar to those in organochromium chemistry but higher oxidation states tend to be more common.

Kamiokite is an iron-molybdenum oxide mineral with the chemical formula Fe2Mo3O8. The name kamiokite is derived from the locality, the Kamioka mine in Gifu Prefecture, Japan, where this mineral was first discovered in 1975.
In chemistry a molybdate is a compound containing an oxoanion with molybdenum in its highest oxidation state of 6. Molybdenum can form a very large range of such oxoanions which can be discrete structures or polymeric extended structures, although the latter are only found in the solid state.The larger oxoanions are members of group of compounds termed polyoxometalates, and because they contain only one type of metal atom are often called isopolymetalates. The discrete molybdenum oxoanions range in size from the simplest MoO2−
4, found in potassium molybdate up to extremely large structures found in isopoly-molybdenum blues that contain for example 154 Mo atoms. The behaviour of molybdenum is different from the other elements in group 6. Chromium only forms the chromates, CrO2−
4, Cr
2O2−
7, Cr
3O2−
10 and Cr
4O2−
13 ions which are all based on tetrahedral chromium. Tungsten is similar to molybdenum and forms many tungstates containing 6 coordinate tungsten.
In chemistry, molybdenum bronze is a generic name for certain mixed oxides of molybdenum with the generic formula A
xMo
yO
z where A may be hydrogen, an alkali metal cation (such as Li+, Na+, K+), and Tl+. These compounds form deeply coloured plate-like crystals with a metallic sheen, hence their name. These bronzes derive their metallic character from partially occupied 4d bands. The oxidation states in K0.28MoO3 are K+1, O2−, and Mo+5.72. MoO3 is an insulator, with an unfilled 4d band.

Ferrimolybdite is a hydrous iron molybdate mineral with formula: Fe3+2(MoO4)3·8(H2O) or Fe3+2(MoO4)3·n(H2O). It forms coatings and radial aggregates of soft yellow needles which crystallize in the orthorhombic system.

Potassium octacyanomolybdate(IV) is the inorganic salt with the formula K4[Mo(CN)8]. A yellow solid, it is the potassium salt of the homoleptic cyanometalate with the coordination number eight. The complex anion consists of a molybdenum cation in the oxidation state IV and eight anionic cyanide ligands resulting in an overall charge of −4, which is balanced with four potassium cations. K4[Mo(CN)8] is often prepared as its dihydrate.
References
| This set index page lists chemical compounds articles associated with the same name. If an internal link led you here, you may wish to change the link to point directly to the intended article. |