Name

STAMPEDE is an acronym for "Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation of Drug Efficacy". [1]
Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation of Drug Efficacy (STAMPEDE) is a clinical trial investigating treatments for high risk or terminal prostate cancer. Recruitment started in 2005 and ends in 2022 and in January 2020, over 10,000 participants have joined the trial.

STAMPEDE is an acronym for "Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation of Drug Efficacy". [1]
The trial investigates new approaches for men with metastatic and locally advanced prostate cancer. [1] 117 hospitals in the UK and Switzerland are involved with over 10,000 men are expected to take part in the trial. [2]
The men are randomly assigned to one of a number of "arms". The primary outcome by which the arms were judged is overall survival. [2]
The following arms are documented in the publicly available literature.
Standard of care (SOC) is lifelong androgen deprivation therapy (ADT). [3] After 2015 SOC was expanded to include docetaxel and radiotherapy (RT) at the start of treatment. [2]
SOC plus zoledronic acid. [3]
Standard care plus docetaxel and prednisolone. [1]
SOC plus zoledronic acid and docetaxel. [3]
SOC plus zoledronic acid and celecoxib. [1]
SOC plus abiraterone acetate and prednisolone. [1]
SOC plus RT. [1]
SOC plus abiraterone and enzalutamide and prednisolone. [1]
SOC plus metformin. [1]
SOC with transdermal oestradiol replacing standard ADT. [1]
James, Sydes & Clarke 2016 reported that "the addition of docetaxel to standard of care was associated with improved survival, with an HR of 0·78 and a difference in median survival of 10 months, as well as improvements in prostate-cancer-specific survival, failure-free survival, and skeletal-related events". They also noted that docetaxel plus zoledronic acid "was associated with similar improvements, although the benefit observed was smaller". [4] The overall conclusion was that "standard of care should be updated to include docetaxel chemotherapy in suitable patients with metastatic disease, and docetaxel may be considered for men with high-risk non-metastatic prostate cancer with or without radiotherapy". [5]
Parker, James & Brawley 2018 reported on radiotherapy. For patients with a high metastatic burden the radiotherapy did not improve survival. No improvement was noted for unselected patients, but for men with a low burden overall survival did improve. There is some discussion as to how the burden is identified (CT and bone scans versus PET). The findings may also be applicable to other cancers where there is a small volume. [2]
Cancer Research UK 2019 presents the main findings as:

Prostate cancer is cancer of the prostate. The prostate is a gland in the male reproductive system that surrounds the urethra just below the bladder. Most prostate cancers are slow growing. Cancerous cells may spread to other areas of the body, particularly the bones and lymph nodes. It may initially cause no symptoms. In later stages, symptoms include pain or difficulty urinating, blood in the urine, or pain in the pelvis or back. Benign prostatic hyperplasia may produce similar symptoms. Other late symptoms include fatigue, due to low levels of red blood cells.
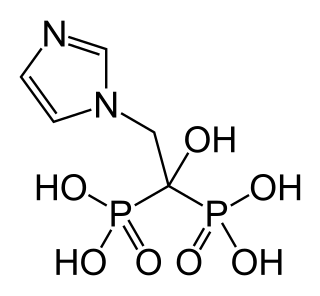
Zoledronic acid, also known as zoledronate, is a medication used to treat a number of bone diseases. These include osteoporosis, high blood calcium due to cancer, bone breakdown due to cancer, and Paget’s disease of bone. It is given by injection into a vein.

Docetaxel, sold under the brand name Taxotere among others, is a chemotherapy medication used to treat a number of types of cancer. This includes breast cancer, head and neck cancer, stomach cancer, prostate cancer and non-small-cell lung cancer. It may be used by itself or along with other chemotherapy medication. It is given by slow injection into a vein.
Nicholas J. Vogelzang is a medical oncologist with Comprehensive Cancer Centers of Nevada (CCCN). He serves as Medical Director of the Research Executive Committee and Associate Chair of the Developmental Therapeutics and Genitourinary Committees for US Oncology Research. His research interests include clinical trials for genitourinary malignancies and mesothelioma.
Adjuvant therapy, also known as adjunct therapy, and adjuvant care, is therapy that is given in addition to the primary or initial therapy to maximize its effectiveness. The surgeries and complex treatment regimens used in cancer therapy have led the term to be used mainly to describe adjuvant cancer treatments. An example of such adjuvant therapy is the additional treatment usually given after surgery where all detectable disease has been removed, but where there remains a statistical risk of relapse due to the presence of undetected disease. If known disease is left behind following surgery, then further treatment is not technically adjuvant.
Receptor activator of nuclear factor kappa-Β ligand (RANKL), also known as tumor necrosis factor ligand superfamily member 11 (TNFSF11), TNF-related activation-induced cytokine (TRANCE), osteoprotegerin ligand (OPGL), and osteoclast differentiation factor (ODF), is a protein that in humans is encoded by the TNFSF11 gene.

Abiraterone acetate, sold under the brand name Zytiga among others, is a medication used to treat prostate cancer. Specifically it is used together with a corticosteroid for metastatic castration-resistant prostate cancer (mCRPC) and metastatic high-risk castration-sensitive prostate cancer (mCSPC). It should either be used following removal of the testicles or along with a gonadotropin-releasing hormone (GnRH) analog. It is taken by mouth.

Enzalutamide, sold under the brand name Xtandi, is a nonsteroidal antiandrogen (NSAA) medication which is used in the treatment of prostate cancer. It is indicated for use in conjunction with castration in the treatment of metastatic castration-resistant prostate cancer (mCRPC), nonmetastatic castration-resistant prostate cancer, and metastatic castration-sensitive prostate cancer (mCSPC). It is taken by mouth.
Ramucirumab is a fully human monoclonal antibody (IgG1) developed for the treatment of solid tumors. This drug was developed by ImClone Systems Inc. It was isolated from a native phage display library from Dyax.
Radium-223 is an isotope of radium with an 11.4-day half-life. It was discovered in 1905 by T. Godlewski, a Polish chemist from Kraków, and was historically known as actinium X (AcX). Radium-223 dichloride is an alpha particle-emitting radiotherapy drug that mimics calcium and forms complexes with hydroxyapatite at areas of increased bone turnover. The principal use of radium-223, as a radiopharmaceutical to treat metastatic cancers in bone, takes advantage of its chemical similarity to calcium, and the short range of the alpha radiation it emits.
James L. Gulley is an American cancer researcher and the Director of the Medical Oncology Service at National Cancer Institute.
Treatment for prostate cancer may involve active surveillance, surgery, radiation therapy – including brachytherapy and external-beam radiation therapy, proton therapy, high-intensity focused ultrasound (HIFU), cryosurgery, hormonal therapy, chemotherapy, or some combination. Treatments also extend to survivorship based interventions. These interventions are focused on five domains including: physical symptoms, psychological symptoms, surveillance, health promotion and care coordination. However, a published review has found only high levels of evidence for interventions that target physical and psychological symptom management and health promotion, with no reviews of interventions for either care coordination or surveillance. The favored treatment option depends on the stage of the disease, the Gleason score, and the PSA level. Other important factors include the man's age, his general health, and his feelings about potential treatments and their possible side-effects. Because all treatments can have significant side-effects, such as erectile dysfunction and urinary incontinence, treatment discussions often focus on balancing the goals of therapy with the risks of lifestyle alterations.
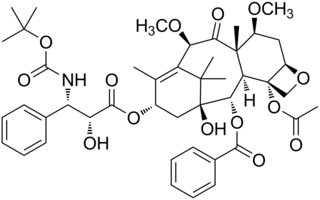
Cabazitaxel, sold under the brand name Jevtana, is a semi-synthetic derivative of a natural taxoid. It was developed by Sanofi-Aventis and was approved by the U.S. FDA for the treatment of hormone-refractory prostate cancer on June 17, 2010. It is a microtubule inhibitor, and the fourth taxane to be approved as a cancer therapy.
Sipuleucel-T, developed by Dendreon Pharmaceuticals, LLC, is a cell-based cancer immunotherapy for prostate cancer (CaP). It is a personalized treatment that works by programming each patient's immune system to seek out cancer and attack it as if it were foreign.

Trastuzumab emtansine also known as ado-trastuzumab emtansine and sold under the trade name Kadcyla, is an antibody-drug conjugate consisting of the humanized monoclonal antibody trastuzumab (Herceptin) covalently linked to the cytotoxic agent DM1. Trastuzumab alone stops growth of cancer cells by binding to the HER2 receptor, whereas trastuzumab emtansine undergoes receptor-mediated internalization into cells, is catabolized in lysosomes where DM1-containing catabolites are released and subsequently bind tubulin to cause mitotic arrest and cell death. Trastuzumab binding to HER2 prevents homodimerization or heterodimerization (HER2/HER3) of the receptor, ultimately inhibiting the activation of MAPK and PI3K/AKT cellular signalling pathways. Because the monoclonal antibody targets HER2, and HER2 is only over-expressed in cancer cells, the conjugate delivers the cytotoxic agent DM1 specifically to tumor cells. The conjugate is abbreviated T-DM1.

Nanobiotix is a biotechnology company that uses nanomedicine to develop new radiotherapy techniques for cancer patients. The company is headquartered in Paris, with additional corporate offices in New York and Massachusetts.
PROSTVAC is a cancer immunotherapy candidate in clinical development by Bavarian Nordic for the treatment of all prostate cancer although clinical trials are focusing on more advanced cases of metastatic castration-resistant prostate cancer (mCRPC). PROSTVAC is a vaccine designed to enable the immune system to recognize and attack prostate cancer cells by triggering a specific and targeted T cell immune response to cancer cells that express the tumor-associated antigen prostate-specific antigen (PSA).

Seviteronel is an experimental cancer medication which is under development by Viamet Pharmaceuticals and Innocrin Pharmaceuticals for the treatment of prostate cancer and breast cancer. It is a nonsteroidal CYP17A1 inhibitor and works by inhibiting the production of androgens and estrogens in the body. As of July 2017, seviteronel is in phase II clinical trials for both prostate cancer and breast cancer. In January 2016, it was designated fast-track status by the United States Food and Drug Administration for prostate cancer. In April 2017, seviteronel received fast-track designation for breast cancer as well.
Abituzumab is a humanized IgG2 monoclonal antibody (mAb) targeted at CD51 currently in development by Merck KGaA Darmstadt, Germany in an attempt to prevent bone lesion metastases in castration-resistant prostate cancer.
The Prostate Adenocarcinoma: TransCutaneous Hormones (PATCH) study is a large randomized controlled trial in the United Kingdom of high-dose transdermal estradiol patches versus gonadotropin-releasing hormone agonist therapy in the treatment of prostate cancer in men. It is specifically comparing three to four 100 μg/day estradiol patches against goserelin implants in approximately 2,200 men with prostate cancer. The study was started in March 2006 and is estimated for completion in August 2021, with additional reports expected in 2023 and 2024. Its objectives include comparison of survival, cardiovascular mortality and morbidity, pharmacological activity, other side effects and toxicities, and quality of life. A report on long-term cardiovascular outcomes was published in February 2021, and other reports for the study have also been published.