Related Research Articles
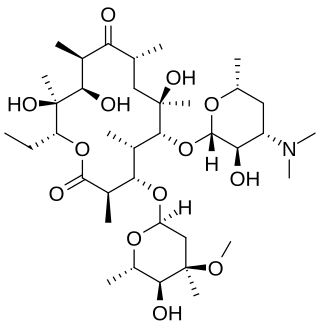
Erythromycin is an antibiotic used for the treatment of a number of bacterial infections. This includes respiratory tract infections, skin infections, chlamydia infections, pelvic inflammatory disease, and syphilis. It may also be used during pregnancy to prevent Group B streptococcal infection in the newborn, as well as to improve delayed stomach emptying. It can be given intravenously and by mouth. An eye ointment is routinely recommended after delivery to prevent eye infections in the newborn.

The Macrolides are a class of natural products that consist of a large macrocyclic lactone ring to which one or more deoxy sugars, usually cladinose and desosamine, may be attached. The lactone rings are usually 14-, 15-, or 16-membered. Macrolides belong to the polyketide class of natural products. Some macrolides have antibiotic or antifungal activity and are used as pharmaceutical drugs. Rapamycin is also a macrolide and was originally developed as an antifungal, but is now used as an immunosuppressant drug and is being investigated as a potential longevity therapeutic.

Treponema pallidum, formerly known as Spirochaeta pallida, is a spirochaete bacterium with various subspecies that cause the diseases syphilis, bejel, and yaws. It is transmitted only among humans. It is a helically coiled microorganism usually 6–15 μm long and 0.1–0.2 μm wide. T. pallidum's lack of either a tricarboxylic acid cycle or oxidative phosphorylation results in minimal metabolic activity. The treponemes have a cytoplasmic and an outer membrane. Using light microscopy, treponemes are visible only by using dark field illumination. Treponema pallidum consists of three subspecies, T. p. pallidum, T. p. endemicum, and T. p. pertenue, each of which has a distinct associated disease.
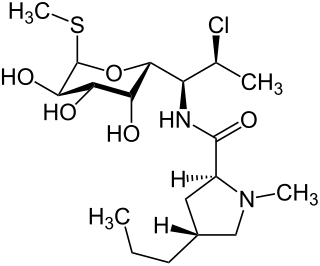
Clindamycin is an antibiotic medication used for the treatment of a number of bacterial infections, including osteomyelitis (bone) or joint infections, pelvic inflammatory disease, strep throat, pneumonia, acute otitis media, and endocarditis. It can also be used to treat acne, and some cases of methicillin-resistant Staphylococcus aureus (MRSA). In combination with quinine, it can be used to treat malaria. It is available by mouth, by injection into a vein, and as a cream or a gel to be applied to the skin or in the vagina.
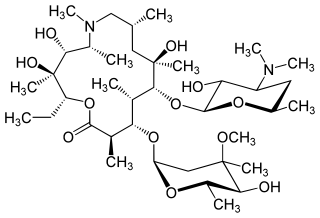
Azithromycin, sold under the brand names Zithromax and Azasite, is an antibiotic medication used for the treatment of a number of bacterial infections. This includes middle ear infections, strep throat, pneumonia, traveler's diarrhea, and certain other intestinal infections. Along with other medications, it may also be used for malaria. It can be taken by mouth or intravenously.

Ketolides are antibiotics belonging to the macrolide group. Ketolides are derived from erythromycin by substituting the cladinose sugar with a keto-group and attaching a cyclic carbamate group in the lactone ring. These modifications give ketolides much broader spectrum than other macrolides. Moreover, ketolides are effective against macrolide-resistant bacteria, due to their ability to bind at two sites at the bacterial ribosome as well as having a structural modification that makes them poor substrates for efflux-pump mediated resistance.
Polyene antimycotics, sometimes referred to as polyene antibiotics, are a class of antimicrobial polyene compounds that target fungi. These polyene antimycotics are typically obtained from some species of Streptomyces bacteria. Previously, polyenes were thought to bind to ergosterol in the fungal cell membrane and thus weakening it and causing leakage of K+ and Na+ ions, which could contribute to fungal cell death. However, more detailed studies of polyene molecular properties have challenged this model suggesting that polyenes instead bind and extract ergosterol directly from the cellular membrane thus disrupting the many cellular functions ergosterols perform. Amphotericin B, nystatin, and natamycin are examples of polyene antimycotics. They are a subgroup of macrolides.

Lincosamides are a class of antibiotics, which include lincomycin, clindamycin, and pirlimycin.

Filipin is a mixture of chemical compounds first isolated by chemists at the Upjohn company in 1955 from the mycelium and culture filtrates of a previously unknown actinomycete, Streptomyces filipinensis. It was discovered in a soil sample collected in the Philippine Islands, hence the name filipin. The isolate possessed potent antifungal activity. It was identified as a polyene macrolide based on its characteristic UV-Vis and IR spectra.

Azalides such as azithromycin are a class of macrolide antibiotics that were originally manufactured in response to the poor acid stability exhibited by original macrolides (erythromycin). Following the clinical overuse of macrolides and azalides, ketolides have been developed to combat surfacing macrolide-azalide resistance among streptococci species. Azalides have several advantages over erythromycin such as more potent gram negative antimicrobial activity, acid stability, and side effect tolerability. Although there are few drug interactions with azithromycin, it weakly inhibits the CYP4A4 enzyme.

Flurithromycin is a second generation macrolide antibiotic. It is a fluorinated derivative of erythromycin A. It is a broad spectrum antibiotic with similar bactericidal action to erythromycin. Unlike erythromycin, flurithromycin is more tolerant of acidic environments, meaning more survives the digestion process, resulting in higher serum levels, and more efficacious elimination of susceptible bacteria, including staphylococcus aureus and streptococcus pyogenes.

Desosamine is a 3-(dimethylamino)-3,4,6-trideoxyhexose found in certain macrolide antibiotics such as the commonly prescribed erythromycin, azithromycin, clarithroymcin, methymycin, narbomycin, oleandomycin, picromycin and roxithromycin. As the name suggests, these macrolide antibiotics contain a macrolide or lactone ring and they are attached to the ring Desosamine which is crucial for bactericidal activity. The biological action of the desosamine-based macrolide antibiotics is to inhibit the bacterial ribosomal protein synthesis. These antibiotics which contain Desosamine are widely used to cure bacterial-causing infections in human respiratory system, skin, muscle tissues, and urethra.

A protein synthesis inhibitor is a compound that stops or slows the growth or proliferation of cells by disrupting the processes that lead directly to the generation of new proteins.

Tylosin is a macrolide antibiotic and bacteriostatic feed additive used in veterinary medicine. It has a broad spectrum of activity against Gram-positive organisms and a limited range of Gram-negative organisms. It is found naturally as a fermentation product of Streptomyces fradiae.
Streptogramin A is a group of antibiotics within the larger family of antibiotics known as streptogramins. They are synthesized by the bacteria Streptomyces virginiae. The streptogramin family of antibiotics consists of two distinct groups: group A antibiotics contain a 23-membered unsaturated ring with lactone and peptide bonds while group B antibiotics are depsipeptides. While structurally different, these two groups of antibiotics act synergistically, providing greater antibiotic activity than the combined activity of the separate components. These antibiotics have until recently been commercially manufactured as feed additives in agriculture, although today there is increased interest in their ability to combat antibiotic-resistant bacteria, particularly vancomycin-resistant bacteria.

Pristinamycin IIA is a macrolide antibiotic. It is a member of the streptogramin A group of antibiotics and one component of pristinamycin. Pristinamycin IIA was first isolated from the Streptomyces virginiae, but has been isolated from other microorganisms and thus has been given a variety of other names such as Virginiamycin M1, Mikamycin A, and Streptogramin A. Pristinamycin IIA structure was determined by chemical and instrumental techniques, including X-ray crystallography. Pristinamycin IIA is of interest from a biosynthetic viewpoint because it contains the unusual dehydroproline and oxazole ring systems. The only experimental evidence bearing on the formation of the oxazole ring is found in work on the biosynthesis of the alkaloid annuloline.
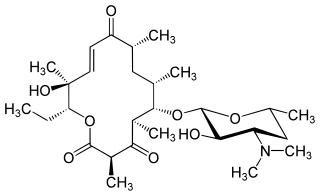
Pikromycin was studied by Brokmann and Hekel in 1951 and was the first antibiotic macrolide to be isolated. Pikromycin is synthesized through a type I polyketide synthase system in Streptomyces venezuelae, a species of Gram-positive bacterium in the genus Streptomyces. Pikromycin is derived from narbonolide, a 14-membered ring macrolide. Along with the narbonolide backbone, pikromycin includes a desosamine sugar and a hydroxyl group. Although Pikromycin is not a clinically useful antibiotic, it can be used as a raw material to synthesize antibiotic ketolide compounds such as ertythromycins and new epothilones.

Carbomycin, also known as magnamycin, is a colorless, optically active crystalline macrolide antibiotic with the molecular formula C42H67NO16. It is derived from the bacterium Streptomyces halstedii and active in inhibiting the growth of Gram-positive bacteria and "certain Mycoplasma strains." Its structure was first proposed by Robert Woodward in 1957 and was subsequently corrected in 1965.
Streptomyces ambofaciens is a bacterium species from the genus Streptomyces which has been isolated from soil from France. Streptomyces ambofaciens produces ambobactin, foromacidin A, foromacidin B, foromacidin C, 18-deoxospiramicin I, 17-methylenespiramycin I and congocidin.
Dactylosporangium aurantiacum is a Gram-positive soil-based actinobacterium in the family Micromonosporaceae. Like all Dactylosporangium species, aurantiacum is aerobic and mesophilic.
References
- 1 2 Hochlowski, J. E.; Swanson, S. J.; Ranfranz, L. M.; Whittern, D. N.; Buko, A. M.; McAlpine, J. B. (1987). "Tiacumicins, a novel complex of 18-membered macrolides. II. Isolation and structure determination". The Journal of Antibiotics. 40 (5): 575–588. doi: 10.7164/antibiotics.40.575 . ISSN 0021-8820. PMID 3610816.