
Surface tension is the tendency of liquid surfaces at rest to shrink into the minimum surface area possible. Surface tension is what allows objects with a higher density than water such as razor blades and insects to float on a water surface without becoming even partly submerged.

In surface science, surface free energy quantifies the disruption of intermolecular bonds that occurs when a surface is created. In solid-state physics, surfaces must be intrinsically less energetically favorable than the bulk of the material, otherwise there would be a driving force for surfaces to be created, removing the bulk of the material. The surface energy may therefore be defined as the excess energy at the surface of a material compared to the bulk, or it is the work required to build an area of a particular surface. Another way to view the surface energy is to relate it to the work required to cut a bulk sample, creating two surfaces. There is "excess energy" as a result of the now-incomplete, unrealized bonding between the two created surfaces.

Adhesion is the tendency of dissimilar particles or surfaces to cling to one another.
An artificial membrane, or synthetic membrane, is a synthetically created membrane which is usually intended for separation purposes in laboratory or in industry. Synthetic membranes have been successfully used for small and large-scale industrial processes since the middle of twentieth century. A wide variety of synthetic membranes is known. They can be produced from organic materials such as polymers and liquids, as well as inorganic materials. The most of commercially utilized synthetic membranes in separation industry are made of polymeric structures. They can be classified based on their surface chemistry, bulk structure, morphology, and production method. The chemical and physical properties of synthetic membranes and separated particles as well as a choice of driving force define a particular membrane separation process. The most commonly used driving forces of a membrane process in industry are pressure and concentration gradients. The respective membrane process is therefore known as filtration. Synthetic membranes utilized in a separation process can be of different geometry and of respective flow configuration. They can also be categorized based on their application and separation regime. The best known synthetic membrane separation processes include water purification, reverse osmosis, dehydrogenation of natural gas, removal of cell particles by microfiltration and ultrafiltration, removal of microorganisms from dairy products, and Dialysis.

Wetting is the ability of a liquid to maintain contact with a solid surface, resulting from intermolecular interactions when the two are brought together. This happens in presence of a gaseous phase or another liquid phase not miscible with the first one. The degree of wetting (wettability) is determined by a force balance between adhesive and cohesive forces.
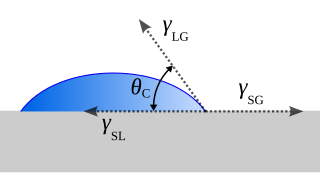
The contact angle is the angle between a liquid surface and a solid surface where they meet. More specifically, it is the angle between the surface tangent on the liquid–vapor interface and the tangent on the solid–liquid interface at their intersection. It quantifies the wettability of a solid surface by a liquid via the Young equation.
Inner sphere complex is a type of surface complex that refers to the surface chemistry changing a water-surface interface to one without water molecules bridging a ligand to the metal ion. Formation of inner sphere complexes occurs when ions bind directly to the surface with no intervening water molecules. These types of surface complexes are restricted to ions that have a high affinity for surface sites and include specifically adsorbed ions that can bind to the surface through covalent bonding.
Imbibition is a special type of diffusion that takes place when liquid is absorbed by solids-colloids causing an increase in volume. Water surface potential movement takes place along a concentration gradient; some dry materials absorb water. A gradient between the absorbent and the liquid is essential for imbibition. For a substance to imbibe a liquid, there must first be some attraction between them. Imbibition occurs when a wetting fluid displaces a non-wetting fluid, the opposite of drainage in which a non-wetting phase displaces the wetting fluid. The two processes are governed by different mechanisms. Imbibition is also a type of diffusion since water movement is along the concentration gradient. Seeds and other such materials have almost no water hence they absorb water easily. Water potential gradient between the absorbent and liquid imbibed is essential for imbibition.

Firefighting foam is a foam used for fire suppression. Its role is to cool the fire and to coat the fuel, preventing its contact with oxygen, thus achieving suppression of the combustion. Firefighting foam was invented by the Russian engineer and chemist Aleksandr Loran in 1902.
In fluid statics, capillary pressure is the pressure between two immiscible fluids in a thin tube, resulting from the interactions of forces between the fluids and solid walls of the tube. Capillary pressure can serve as both an opposing or driving force for fluid transport and is a significant property for research and industrial purposes. It is also observed in natural phenomena.

A Wilhelmy plate is a thin plate that is used to measure equilibrium surface or interfacial tension at an air–liquid or liquid–liquid interface. In this method, the plate is oriented perpendicular to the interface, and the force exerted on it is measured. Based on the work of Ludwig Wilhelmy, this method finds wide use in the preparation and monitoring of Langmuir films.
A cuticle, or cuticula, is any of a variety of tough but flexible, non-mineral outer coverings of an organism, or parts of an organism, that provide protection. Various types of "cuticle" are non-homologous, differing in their origin, structure, function, and chemical composition.
A wetting transition may occur during the process of wetting of a solid surface with a liquid. The transition corresponds to a certain change in contact angle, the macroscopic parameter characterizing wetting. Various contact angles can co-exist on the same solid substrate. Wetting transitions may occur in a different way depending on whether the surface is flat or rough.
The wet strength of paper and paperboard is a measure of how well the web of fibers holding the paper together can resist a force of rupture when the paper is wet. Wet strength is routinely expressed as the ratio of wet to dry tensile force at break.

A hydrophile is a molecule or other molecular entity that is attracted to water molecules and tends to be dissolved by water.

A liquid metal is a metal or a metal alloy which is liquid at or near room temperature.
Capillary flow porometry, also known as porometry, is a characterization technique based on the displacement of a wetting liquid from the sample pores by applying a gas at increasing pressure. It is widely used to measure minimum, maximum and mean flow pore sizes, and pore size distribution of the through pores in membranes nonwovens, paper, filtration and ultrafiltration media, hollow fibers, ceramics, etc.
The liquid entry pressure (LEP) of a hydrophobic membrane is the pressure that must be applied to a dry membrane so that the liquid penetrates inside the membrane. LEP with the application in membrane distillation or pervaporation can be calculated as a first parameter to indicate how wettable a membrane is toward different liquid solutions.

Wetting solutions are liquids containing active chemical compounds that minimise the distance between two immiscible phases by lowering the surface tension to induce optimal spreading. The two phases, known as an interface, can be classified into five categories, namely, solid-solid, solid-liquid, solid-gas, liquid-liquid and liquid-gas.









