
Chemical vapor deposition (CVD) is a vacuum deposition method used to produce high-quality, and high-performance, solid materials. The process is often used in the semiconductor industry to produce thin films.

In surface science, surface energy quantifies the disruption of intermolecular bonds that occurs when a surface is created. In solid-state physics, surfaces must be intrinsically less energetically favorable than the bulk of the material, otherwise there would be a driving force for surfaces to be created, removing the bulk of the material. The surface energy may therefore be defined as the excess energy at the surface of a material compared to the bulk, or it is the work required to build an area of a particular surface. Another way to view the surface energy is to relate it to the work required to cut a bulk sample, creating two surfaces. There is "excess energy" as a result of the now-incomplete, unrealized bonding between the two created surfaces.
An artificial membrane, or synthetic membrane, is a synthetically created membrane which is usually intended for separation purposes in laboratory or in industry. Synthetic membranes have been successfully used for small and large-scale industrial processes since the middle of the twentieth century. A wide variety of synthetic membranes is known. They can be produced from organic materials such as polymers and liquids, as well as inorganic materials. Most commercially utilized synthetic membranes in industry are made of polymeric structures. They can be classified based on their surface chemistry, bulk structure, morphology, and production method. The chemical and physical properties of synthetic membranes and separated particles as well as separation driving force define a particular membrane separation process. The most commonly used driving forces of a membrane process in industry are pressure and concentration gradient. The respective membrane process is therefore known as filtration. Synthetic membranes utilized in a separation process can be of different geometry and flow configurations. They can also be categorized based on their application and separation regime. The best known synthetic membrane separation processes include water purification, reverse osmosis, dehydrogenation of natural gas, removal of cell particles by microfiltration and ultrafiltration, removal of microorganisms from dairy products, and dialysis.

Gas mixtures can be effectively separated by synthetic membranes made from polymers such as polyamide or cellulose acetate, or from ceramic materials.

Wetting is the ability of a liquid to maintain contact with a solid surface, resulting from intermolecular interactions when the two are brought together. This happens in presence of a gaseous phase or another liquid phase not miscible with the first one. The degree of wetting (wettability) is determined by a force balance between adhesive and cohesive forces.
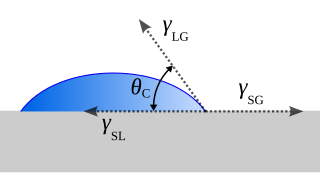
The contact angle is the angle between a liquid surface and a solid surface where they meet. More specifically, it is the angle between the surface tangent on the liquid–vapor interface and the tangent on the solid–liquid interface at their intersection. It quantifies the wettability of a solid surface by a liquid via the Young equation.

In chemistry and materials science, ultrahydrophobic surfaces are highly hydrophobic, i.e., extremely difficult to wet. The contact angles of a water droplet on an ultrahydrophobic material exceed 150°. This is also referred to as the lotus effect, after the superhydrophobic leaves of the lotus plant. A droplet striking these kinds of surfaces can fully rebound like an elastic ball. Interactions of bouncing drops can be further reduced using special superhydrophobic surfaces that promote symmetry breaking, pancake bouncing or waterbowl bouncing.
Thin-film composite membranes are semipermeable membranes manufactured to provide selectivity with high permeability. Most TFC's are used in water purification or water desalination systems. They also have use in chemical applications such as gas separations, dehumidification, batteries and fuel cells. A TFC membrane can be considered a molecular sieve constructed in the form of a film from two or more layered materials. The additional layers provide structural strength and a low-defect surface to support a selective layer that is thin enough to be selective but not so thick that it causes low permeability.
Nanofiltration is a membrane filtration process that uses nanometer sized pores through which particles smaller than about 1–10 nanometers pass through the membrane. Nanofiltration membranes have pore sizes of about 1–10 nanometers, smaller than those used in microfiltration and ultrafiltration, but a slightly bigger than those in reverse osmosis. Membranes used are predominantly polymer thin films. It is used to soften, disinfect, and remove impurities from water, and to purify or separate chemicals such as pharmaceuticals.
Gas diffusion electrodes (GDE) are electrodes with a conjunction of a solid, liquid and gaseous interface, and an electrical conducting catalyst supporting an electrochemical reaction between the liquid and the gaseous phase.

Membrane fouling is a process whereby a solution or a particle is deposited on a membrane surface or in membrane pores in a processes such as in a membrane bioreactor, reverse osmosis, forward osmosis, membrane distillation, ultrafiltration, microfiltration, or nanofiltration so that the membrane's performance is degraded. It is a major obstacle to the widespread use of this technology. Membrane fouling can cause severe flux decline and affect the quality of the water produced. Severe fouling may require intense chemical cleaning or membrane replacement. This increases the operating costs of a treatment plant. There are various types of foulants: colloidal, biological, organic and scaling.

A membrane is a selective barrier; it allows some things to pass through but stops others. Such things may be molecules, ions, or other small particles. Membranes can be generally classified into synthetic membranes and biological membranes. Biological membranes include cell membranes ; nuclear membranes, which cover a cell nucleus; and tissue membranes, such as mucosae and serosae. Synthetic membranes are made by humans for use in laboratories and industry.

A hydrophile is a molecule or other molecular entity that is attracted to water molecules and tends to be dissolved by water.
Membrane distillation (MD) is a thermally driven separation process in which separation is driven by phase change. A hydrophobic membrane presents a barrier for the liquid phase, allowing the vapour phase to pass through the membrane's pores. The driving force of the process is a partial vapour pressure difference commonly triggered by a temperature difference.
Capillary flow porometry, also known as porometry, is a characterization technique based on the displacement of a wetting liquid from the sample pores by applying a gas at increasing pressure. It is widely used to measure minimum, maximum and mean flow pore sizes, and pore size distribution of the through pores in membranes nonwovens, paper, filtration and ultrafiltration media, hollow fibers, ceramics, etc.
A superhydrophobic coating is a thin surface layer that repels water. It is made from superhydrophobic (ultrahydrophobicity) materials. Droplets hitting this kind of coating can fully rebound. Generally speaking, superhydrophobic coatings are made from composite materials where one component provides the roughness and the other provides low surface energy.
Flotation of flexible objects is a phenomenon in which the bending of a flexible material allows an object to displace a greater amount of fluid than if it were completely rigid. This ability to displace more fluid translates directly into an ability to support greater loads, giving the flexible structure an advantage over a similarly rigid one. Inspiration to study the effects of elasticity are taken from nature, where plants, such as black pepper, and animals living at the water surface have evolved to take advantage of the load-bearing benefits elasticity imparts.
Evapoporometry is a method used to determine pore-size in synthetic membranes. Based on the Kelvin equation, this technique is most accurate for detection of pore diameters between 4 nm to 150 nm.

John Henry Lienhard V is the Abdul Latif Jameel Professor of Water and Mechanical Engineering at the Massachusetts Institute of Technology. His research focuses on desalination, heat transfer, and thermodynamics. He has also written several engineering textbooks.

Wetting solutions are liquids containing active chemical compounds that minimise the distance between two immiscible phases by lowering the surface tension to induce optimal spreading. The two phases, known as an interface, can be classified into five categories, namely, solid-solid, solid-liquid, solid-gas, liquid-liquid and liquid-gas.

















