Yttrium oxide may refer to:
- Yttrium(II) oxide, YO, a dark brown solid
- Yttrium(III) oxide, Y2O3, a colorless solid
Yttrium oxide may refer to:

A laser is constructed from three principal parts:

Terbium is a chemical element with the symbol Tb and atomic number 65. It is a silvery-white, rare earth metal that is malleable, ductile, and soft enough to be cut with a knife. The ninth member of the lanthanide series, terbium is a fairly electropositive metal that reacts with water, evolving hydrogen gas. Terbium is never found in nature as a free element, but it is contained in many minerals, including cerite, gadolinite, monazite, xenotime, and euxenite.

The skull crucible process was developed at the Lebedev Physical Institute in Moscow to manufacture cubic zirconia. It was invented to solve the problem of cubic zirconia's melting-point being too high for even platinum crucibles.

Zirconium dioxide, sometimes known as zirconia, is a white crystalline oxide of zirconium. Its most naturally occurring form, with a monoclinic crystalline structure, is the mineral baddeleyite. A dopant stabilized cubic structured zirconia, cubic zirconia, is synthesized in various colours for use as a gemstone and a diamond simulant.

Group 3 is the first group of transition metals in the periodic table. This group is closely related to the rare-earth elements. Although some controversy exists regarding the composition and placement of this group, it is generally agreed among those who study the matter that this group contains the four elements scandium (Sc), yttrium (Y), lutetium (Lu), and lawrencium (Lr). The group is also called the scandium group or scandium family after its lightest member.

An incandescent gas mantle, gas mantle or Welsbach mantle is a device for generating incandescent bright white light when heated by a flame. The name refers to its original heat source in gas lights which illuminated the streets of Europe and North America in the late 19th century. Mantle refers to the way it hangs like a cloak above the flame. Gas mantles were also used in portable camping lanterns, pressure lanterns and some oil lamps.

Thorium dioxide (ThO2), also called thorium(IV) oxide, is a crystalline solid, often white or yellow in color. Also known as thoria, it is produced mainly as a by-product of lanthanide and uranium production. Thorianite is the name of the mineralogical form of thorium dioxide. It is moderately rare and crystallizes in an isometric system. The melting point of thorium oxide is 3300 °C – the highest of all known oxides. Only a few elements (including tungsten and carbon) and a few compounds (including tantalum carbide) have higher melting points. All thorium compounds are radioactive because there are no stable isotopes of thorium.

Yttrium oxide, also known as yttria, is Y2O3. It is an air-stable, white solid substance.
In chemistry, a mixed oxide is a somewhat informal name for an oxide that contains cations of more than one chemical element or cations of a single element in several states of oxidation.

Yttrium iron garnet (YIG) is a kind of synthetic garnet, with chemical composition Y3Fe2(FeO4)3, or Y3Fe5O12. It is a ferrimagnetic material with a Curie temperature of 560 K. YIG may also be known as yttrium ferrite garnet, or as iron yttrium oxide or yttrium iron oxide, the latter two names usually associated with powdered forms.
Lutetium aluminum garnet (commonly abbreviated LuAG, molecular formula Al5Lu3O12) is an inorganic compound with a unique crystal structure primarily known for its use in high-efficiency laser devices. LuAG is also useful in the synthesis of transparent ceramics.
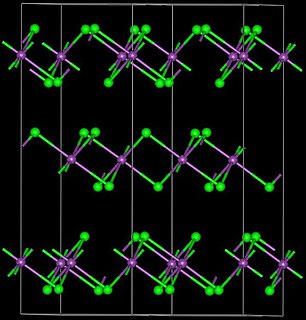
Yttrium(III) bromide is an inorganic compound with the chemical formula YBr3. It is a white solid. Anhydrous yttrium(III) bromide can be produced by reacting yttrium oxide or yttrium(III) bromide hydrate and ammonium bromide. The reaction proceeds via the intermediate (NH4)3YBr6. Another method is to react yttrium carbide (YC2) and elemental bromine. Yttrium(III) bromide can be reduced by yttrium metal to YBr or Y2Br3. It can react with osmium to produce Y4Br4Os.

Yttrium is a chemical element with the symbol Y and atomic number 39. It is a silvery-metallic transition metal chemically similar to the lanthanides and has often been classified as a "rare-earth element". Yttrium is almost always found in combination with lanthanide elements in rare-earth minerals, and is never found in nature as a free element. 89Y is the only stable isotope, and the only isotope found in the Earth's crust.
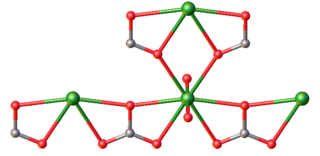
Uranyl carbonate refers to the inorganic compound with the formula UO2CO3. Also known by its mineral name rutherfordine, this material consists of uranyl (UO22+) and carbonate (CO32-). The compound is a polymeric, each uranium(VI) center being bonded to eight O atoms. Hydrolysis products of rutherfordine are also found in both the mineral and organic fractions of coal and its fly ash and is the main component of uranium in mine tailing seepage water.

Europium(III) oxide (Eu2O3), is a chemical compound of europium and oxygen. It is widely used as a red or blue phosphor in television sets and fluorescent lamps, and as an activator for yttrium-based phosphors. It is also an agent for the manufacture of fluorescent glass. Europium fluorescence is used in the anti-counterfeiting phosphors in Euro banknotes.
A dopant, also called a doping agent, is a trace of impurity element that is introduced into a chemical material to alter its original electrical or optical properties. The amount of dopant necessary to cause changes is typically very low. When doped into crystalline substances, the dopant's atoms get incorporated into its crystal lattice. The crystalline materials are frequently either crystals of a semiconductor such as silicon and germanium for use in solid-state electronics, or transparent crystals for use in the production of various laser types; however, in some cases of the latter, noncrystalline substances such as glass can also be doped with impurities.
A complex oxide is a chemical compound that contains oxygen and at least two other elements. Complex oxide materials are notable for their wide range of magnetic and electronic properties, such as ferromagnetism, ferroelectricity, and high-temperature superconductivity. These properties often come from their strongly correlated electrons in d or f orbitals.

Yttrium(III) nitrate is an inorganic compound with the formula Y(NO3)3. The hexahydrate is the most common form commercially available.
An yttrium compound is a chemical compound containing yttrium. Among these compounds, yttrium generally has a +3 valence. The solubility properties of yttrium compounds are similar to those of the lanthanides. For example oxalates and carbonates are hardly soluble in water, but soluble in excess oxalate or carbonate solutions as complexes are formed. Sulfates and double sulfates are generally soluble. They resemble the "yttrium group" of heavy lanathanide elements.

Lutetium(III) fluoride is an inorganic compound with a chemical formula LuF3.