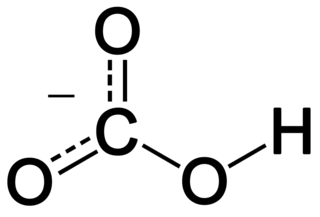
In inorganic chemistry, bicarbonate is an intermediate form in the deprotonation of carbonic acid. It is a polyatomic anion with the chemical formula HCO−
3.
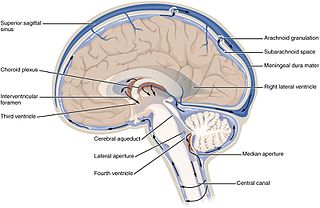
Cerebrospinal fluid (CSF) is a clear, colorless body fluid found in the brain and spinal cord. It is produced by specialised ependymal cells in the choroid plexuses of the ventricles of the brain, and absorbed in the arachnoid granulations. There is about 125 mL of CSF at any one time, and about 500 mL is generated every day. CSF acts as a cushion or buffer, providing basic mechanical and immunological protection to the brain inside the skull. CSF also serves a vital function in the cerebral autoregulation of cerebral blood flow.
The chloride ion is the anion Cl−. It is formed when the element chlorine gains an electron or when a compound such as hydrogen chloride is dissolved in water or other polar solvents. Chloride salts such as sodium chloride are often very soluble in water. It is an essential electrolyte located in all body fluids responsible for maintaining acid/base balance, transmitting nerve impulses and regulating fluid in and out of cells. Less frequently, the word chloride may also form part of the "common" name of chemical compounds in which one or more chlorine atoms are covalently bonded. For example, methyl chloride, with the standard name chloromethane is an organic compound with a covalent C−Cl bond in which the chlorine is not an anion.

Soil pH is a measure of the acidity or basicity (alkalinity) of a soil. pH is defined as the negative logarithm (base 10) of the activity of hydronium ions in a solution. In soils, it is measured in a slurry of soil mixed with water, and normally falls between 3 and 10, with 7 being neutral. Acid soils have a pH below 7 and alkaline soils have a pH above 7. Ultra-acidic soils and very strongly alkaline soils are rare.
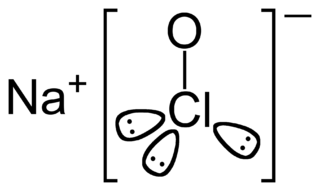
Sodium hypochlorite is a chemical compound with the formula NaOCl or NaClO, comprising a sodium cation and a hypochlorite anion. It may also be viewed as the sodium salt of hypochlorous acid. The anhydrous compound is unstable and may decompose explosively. It can be crystallized as a pentahydrate NaOCl·5H
2O, a pale greenish-yellow solid which is not explosive and is stable if kept refrigerated.
Apnea is the cessation of breathing. During apnea, there is no movement of the muscles of inhalation, and the volume of the lungs initially remains unchanged. Depending on how blocked the airways are (patency), there may or may not be a flow of gas between the lungs and the environment; gas exchange within the lungs and cellular respiration is not affected. Voluntarily doing this is called holding one's breath.

Chlorine dioxide is a chemical compound with the formula ClO2 that exists as yellowish-green gas above 11 °C, a reddish-brown liquid between −59 °C and 11 °C, and as bright orange crystals when colder. It is an oxidizing agent, able to transfer oxygen to a variety of substrates, while gaining one or more electrons via oxidation-reduction (redox). It does not hydrolyze when it enters water, and is usually handled as a dissolved gas in solution in water. Potential hazards with chlorine dioxide include health concerns, explosiveness and fire ignition. It is commonly used as a bleach.
A lysis buffer is a buffer solution used for the purpose of breaking open cells for use in molecular biology experiments that analyze the labile macromolecules of the cells. Most lysis buffers contain buffering salts and ionic salts to regulate the pH and osmolarity of the lysate. Sometimes detergents are added to break up membrane structures. For lysis buffers targeted at protein extraction, protease inhibitors are often included, and in difficult cases may be almost required. Lysis buffers can be used on both animal and plant tissue cells.

Intracranial pressure (ICP) is the pressure exerted by fluids such as cerebrospinal fluid (CSF) inside the skull and on the brain tissue. ICP is measured in millimeters of mercury (mmHg) and, at rest, is normally 7–15 mmHg for a supine adult. The body has various mechanisms by which it keeps the ICP stable, with CSF pressures varying by about 1 mmHg in normal adults through shifts in production and absorption of CSF.

Saline, also known as saline solution, is a mixture of sodium chloride in water and has a number of uses in medicine. Applied to the affected area it is used to clean wounds, help remove contact lenses, and help with dry eyes. By injection into a vein it is used to treat dehydration such as from gastroenteritis and diabetic ketoacidosis. It is also used to dilute other medications to be given by injection.
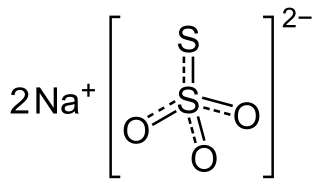
Sodium thiosulfate (sodium thiosulphate) is an inorganic compound with the formula Na2S2O3.xH2O. Typically it is available as the white or colorless pentahydrate, Na2S2O3·5H2O. The solid is an efflorescent (loses water readily) crystalline substance that dissolves well in water.
Phosphate-buffered saline is a buffer solution commonly used in biological research. It is a water-based salt solution containing disodium hydrogen phosphate, sodium chloride and, in some formulations, potassium chloride and potassium dihydrogen phosphate. The buffer helps to maintain a constant pH. The osmolarity and ion concentrations of the solutions match those of the human body (isotonic).
TAE buffer is a buffer solution containing a mixture of Tris base, acetic acid and EDTA.
Carbogen, also called Meduna's Mixture after its inventor Ladislas Meduna, is a mixture of carbon dioxide and oxygen gas. Meduna's original formula was 5% CO2 and 95% oxygen, but the term carbogen can refer to any mixture of these two gases, from 1.5% to 50% CO2.

The Gibbs–Donnan effect is a name for the behaviour of charged particles near a semi-permeable membrane that sometimes fail to distribute evenly across the two sides of the membrane. The usual cause is the presence of a different charged substance that is unable to pass through the membrane and thus creates an uneven electrical charge. For example, the large anionic proteins in blood plasma are not permeable to capillary walls. Because small cations are attracted, but are not bound to the proteins, small anions will cross capillary walls away from the anionic proteins more readily than small cations.
Tris-buffered saline (TBS) is a buffer used in some biochemical techniques to maintain the pH within a relatively narrow range. Tris has a slightly alkaline buffering capacity in the 7–9.2 range. The conjugate acid of Tris has a pKa of 8.07 at 25 °C. The pKa declines approximately 0.03 units per degree Celsius rise in temperature. This can lead to relatively dramatic pH shifts when there are shifts in solution temperature. Sodium chloride concentration may vary from 100 to 200 mM, tris concentration from 5 to 100 mM and pH from 7.2 to 8.0. A common formulation of TBS is 150 mM NaCl, 50 mM Tris-HCl, pH 7.6. TBS can also be prepared by using commercially made TBS buffer tablets or pouches.
Central chemoreceptors of the central nervous system, located on the ventrolateral medullary surface in the vicinity of the exit of the 9th and 10th cranial nerves, are sensitive to the pH of their environment.
Borate buffered saline is a buffer used in some biochemical techniques to maintain the pH within a relatively narrow range. Borate buffers have an alkaline buffering capacity in the 8–10 range. Boric acid has a pKa of 9.14 at 25°C.
A balanced salt solution (BSS) is a solution made to a physiological pH and isotonic salt concentration. Solutions most commonly include sodium, potassium, calcium, magnesium, and chloride. Balanced salt solutions are used for washing tissues and cells and are usually combined with other agents to treat the tissues and cells. They provide the cells with water and inorganic ions, while maintaining a physiological pH and osmotic pressure.

The slice preparation or brain slice is a laboratory technique in electrophysiology that allows the study of a synapse or neural circuit in isolation from the rest of the brain, in controlled physiological conditions. Brain tissue is initially sliced via a tissue slicer then immersed in artificial cerebrospinal fluid (aCSF) for stimulation and/or recording. The technique allows for greater experimental control, through elimination of the effects of the rest of the brain on the circuit of interest, careful control of the physiological conditions through perfusion of substrates through the incubation fluid, to precise manipulation of neurotransmitter activity through perfusion of agonists and antagonists. However, the increase in control comes with a decrease in the ease with which the results can be applied to the whole neural system.