Related Research Articles
In condensed matter physics and materials science, an amorphous solid is a solid that lacks the long-range order that is characteristic of a crystal. The terms "glass" and "glassy solid" are sometimes used synonymously with amorphous solid; however, these terms refer specifically to amorphous materials that undergo a glass transition. Examples of amorphous solids include glasses, metallic glasses, and certain types of plastics and polymers.

Glass is an amorphous (non-crystalline) solid. Because it is often transparent and chemically inert, glass has found widespread practical, technological, and decorative use in window panes, tableware, and optics. Some common objects made of glass like "a glass" of water, "glasses", and "magnifying glass", are named after the material.

Corrosion is a natural process that converts a refined metal into a more chemically stable oxide. It is the gradual deterioration of materials by chemical or electrochemical reaction with their environment. Corrosion engineering is the field dedicated to controlling and preventing corrosion.

An amorphous metal is a solid metallic material, usually an alloy, with disordered atomic-scale structure. Most metals are crystalline in their solid state, which means they have a highly ordered arrangement of atoms. Amorphous metals are non-crystalline, and have a glass-like structure. But unlike common glasses, such as window glass, which are typically electrical insulators, amorphous metals have good electrical conductivity and can show metallic luster.

Liquidmetal and Vitreloy are commercial names of a series of amorphous metal alloys developed by a California Institute of Technology (Caltech) research team and marketed by Liquidmetal Technologies. Liquidmetal alloys combine a number of desirable material features, including high tensile strength, excellent corrosion resistance, very high coefficient of restitution and excellent anti-wearing characteristics, while also being able to be heat-formed in processes similar to thermoplastics. Despite the name, they are not liquid at room temperature.
Chalcogenide glass is a glass containing one or more chalcogens. Polonium is also a chalcogen but is not used because of its strong radioactivity. Chalcogenide materials behave rather differently from oxides, in particular their lower band gaps contribute to very dissimilar optical and electrical properties.

Bioglass 45S5 or calcium sodium phosphosilicate, is a bioactive glass specifically composed of 45 wt% SiO2, 24.5 wt% CaO, 24.5 wt% Na2O, and 6.0 wt% P2O5. Typical applications of Bioglass 45S5 include: bone grafting biomaterials, repair of periodontal defects, cranial and maxillofacial repair, wound care, blood loss control, stimulation of vascular regeneration, and nerve repair.

Bioactive glasses are a group of surface reactive glass-ceramic biomaterials and include the original bioactive glass, Bioglass. The biocompatibility and bioactivity of these glasses has led them to be used as implant devices in the human body to repair and replace diseased or damaged bones. Most bioactive glasses are silicate-based glasses that are degradable in body fluids and can act as a vehicle for delivering ions beneficial for healing. Bioactive glass is differentiated from other synthetic bone grafting biomaterials, in that it is the only one with anti-infective and angiogenic properties.

Nanocomposite is a multiphase solid material where one of the phases has one, two or three dimensions of less than 100 nanometers (nm) or structures having nano-scale repeat distances between the different phases that make up the material.
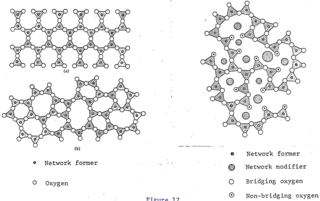
The glass–liquid transition, or glass transition, is the gradual and reversible transition in amorphous materials from a hard and relatively brittle "glassy" state into a viscous or rubbery state as the temperature is increased. An amorphous solid that exhibits a glass transition is called a glass. The reverse transition, achieved by supercooling a viscous liquid into the glass state, is called vitrification.

Bioceramics and bioglasses are ceramic materials that are biocompatible. Bioceramics are an important subset of biomaterials. Bioceramics range in biocompatibility from the ceramic oxides, which are inert in the body, to the other extreme of resorbable materials, which are eventually replaced by the body after they have assisted repair. Bioceramics are used in many types of medical procedures. Bioceramics are typically used as rigid materials in surgical implants, though some bioceramics are flexible. The ceramic materials used are not the same as porcelain type ceramic materials. Rather, bioceramics are closely related to either the body's own materials or are extremely durable metal oxides.

A bioresorbable stent is a tube-like device (stent) that is used to open and widen clogged heart arteries and then dissolves or is absorbed by the body. It is made from a material that can release a drug to prevent scar tissue growth. It can also restore normal vessel function and avoid long-term complications of metal stents.

Friction stir processing (FSP) is a method of changing the properties of a metal through intense, localized plastic deformation. This deformation is produced by forcibly inserting a non-consumable tool into the workpiece, and revolving the tool in a stirring motion as it is pushed laterally through the workpiece. The precursor of this technique, friction stir welding, is used to join multiple pieces of metal without creating the heat affected zone typical of fusion welding.
Splat quenching is a metallurgical, metal morphing technique used for forming metals with a particular crystal structure by means of extremely rapid quenching, or cooling.
Bioresorbablemetals are metals or their alloys that degrade safely within the body. The primary metals in this category are magnesium-based and iron-based alloys, although recently zinc has also been investigated. Currently, the primary uses of bioresorbable metals are as stents for blood vessels and other internal ducts.
Bioactive glasses have been synthesized through methods such as conventional melting, quenching, the sol–gel process, flame synthesis, and microwave irradiation. The synthesis of bioglass has been reviewed by various groups, with sol-gel synthesis being one of the most frequently used methods for producing bioglass composites, particularly for tissue engineering applications. Other methods of bioglass synthesis have been developed, such as flame and microwave synthesis, though they are less prevalent in research.
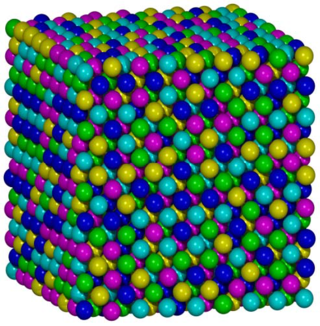
High-entropy alloys (HEAs) are alloys that are formed by mixing equal or relatively large proportions of (usually) five or more elements. Prior to the synthesis of these substances, typical metal alloys comprised one or two major components with smaller amounts of other elements. For example, additional elements can be added to iron to improve its properties, thereby creating an iron-based alloy, but typically in fairly low proportions, such as the proportions of carbon, manganese, and others in various steels. Hence, high-entropy alloys are a novel class of materials. The term "high-entropy alloys" was coined by Taiwanese scientist Jien-Wei Yeh because the entropy increase of mixing is substantially higher when there is a larger number of elements in the mix, and their proportions are more nearly equal. Some alternative names, such as multi-component alloys, compositionally complex alloys and multi-principal-element alloys are also suggested by other researchers.

Alain Reza Yavari was a French scholar in the fields of chemical and physical metallurgy, with a focus on bulk metallic glasses. His transdisciplinary approach across physics and chemistry led to remarkable innovations that contributed to significant breakthroughs in both fundamental and applied material sciences.

A nanostructured film is a film resulting from engineering of nanoscale features, such as dislocations, grain boundaries, defects, or twinning. In contrast to other nanostructures, such as nanoparticles, the film itself may be up to several microns thick, but possesses a large concentration of nanoscale features homogeneously distributed throughout the film. Like other nanomaterials, nanostructured films have sparked much interest as they possess unique properties not found in bulk, non-nanostructured material of the same composition. In particular, nanostructured films have been the subject of recent research due to their superior mechanical properties, including strength, hardness, and corrosion resistance compared to regular films of the same material. Examples of nanostructured films include those produced by grain boundary engineering, such as nano-twinned ultra-fine grain copper, or dual phase nanostructuring, such as crystalline metal and amorphous metallic glass nanocomposites.
Titanium foams exhibit high specific strength, high energy absorption, excellent corrosion resistance and biocompatibility. These materials are ideally suited for applications within the aerospace industry. An inherent resistance to corrosion allows the foam to be a desirable candidate for various filtering applications. Further, titanium's physiological inertness makes its porous form a promising candidate for biomedical implantation devices. The largest advantage in fabricating titanium foams is that the mechanical and functional properties can be adjusted through manufacturing manipulations that vary porosity and cell morphology. The high appeal of titanium foams is directly correlated to a multi-industry demand for advancement in this technology.
References
- 1 2 Mg-Zn-Ca ternary system
- ↑ Ibrahim, H.; Esfahani, S. N.; Poorganji, B.; Dean, D.; Elahinia, M. (January 2017). "Resorbable bone fixation alloys, forming, and post-fabrication treatments". Materials Science and Engineering: C. 70 (1): 870–888. doi: 10.1016/j.msec.2016.09.069 . PMID 27770965.
- ↑ Klement, W.; Willens, R. H.; Duwez, POL (1960). "Non-crystalline structure in solidified gold-silicon Alloys". Nature. 187 (4740): 869–870. Bibcode:1960Natur.187..869K. doi:10.1038/187869b0. S2CID 4203025.
- 1 2 Senkov, O.N.; Scott, J.M. (2005). "Glass forming ability and thermal stability of ternary Ca-Mg-Zn bulk metallic glasses". Journal of Non-Crystalline Solids. 351 (37–39): 3087–3094. Bibcode:2005JNCS..351.3087S. doi:10.1016/j.jnoncrysol.2005.07.022.
- ↑ "Fixing bones with dissolvable glass". PhysicsWorld. Oct 1, 2009.
- 1 2 Wang, Y.B.; et al. (2011). "Biodegradable CaMgZn bulk metallic glass for potential skeletal application". Acta Biomaterialia. 7 (8): 3196–3208. doi:10.1016/j.actbio.2011.04.027. PMID 21571105.
- ↑ Dahlman, J.; Senkov, O.N.; Scott, J.M.; Miracle, D.B. (2007). "Corrosion properties of Ca based bulk metallic glasses" (PDF). Materials Transactions. 48 (7): 1850–1854. doi: 10.2320/matertrans.mj200732 .
- 1 2 Cao, J.D.; et al. (2012). "Ca–Mg–Zn bulk metallic glasses as bioresorbable metals". Acta Biomaterialia. 8 (6): 2375–2383. doi:10.1016/j.actbio.2012.03.009. PMID 22406910.
- ↑ Mills, Georgie. "Mending broken bones with glass". Australia Unlimited. Retrieved 22 April 2013.
- ↑ "BMGs for Electronic, Biomedical and Aerospace Applications". University of New South Wales. Apr 28, 2010. Archived from the original on 2013-01-05.
- ↑ Kirkland, N.T. (2012). "Magnesium biomaterials: Past, present and future". Corrosion Engineering, Science and Technology. 47 (5): 322–328. doi:10.1179/1743278212Y.0000000034. hdl: 10069/29852 . S2CID 135864605.