Related Research Articles
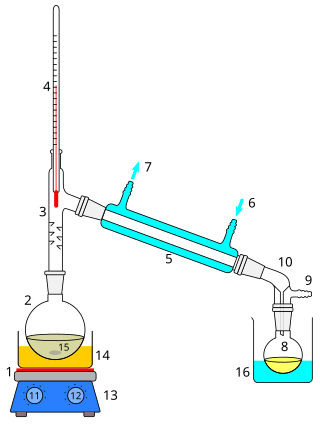
Distillation, or classical distillation, is the process of separating the components or substances from a liquid mixture by using selective boiling and condensation, usually inside an apparatus known as a still. Dry distillation is the heating of solid materials to produce gaseous products ; this may involve chemical changes such as destructive distillation or cracking. Distillation may result in essentially complete separation, or it may be a partial separation that increases the concentration of selected components; in either case, the process exploits differences in the relative volatility of the mixture's components. In industrial applications, distillation is a unit operation of practically universal importance, but is a physical separation process, not a chemical reaction. An installation used for distillation, especially of distilled beverages, is a distillery. Distillation includes the following applications:

Laboratory glassware refers to a variety of equipment used in scientific work, and traditionally made of glass. Glass can be blown, bent, cut, molded, and formed into many sizes and shapes, and is therefore common in chemistry, biology, and analytical laboratories. Many laboratories have training programs to demonstrate how glassware is used and to alert first–time users to the safety hazards involved with using glassware.

Boiling is the rapid phase transition from liquid to gas or vapor; the reverse of boiling is condensation. Boiling occurs when a liquid is heated to its boiling point, so that the vapour pressure of the liquid is equal to the pressure exerted on the liquid by the surrounding atmosphere. Boiling and evaporation are the two main forms of liquid vapourization.

Liquid hydrogen (H2(l)) is the liquid state of the element hydrogen. Hydrogen is found naturally in the molecular H2 form.

A Florence flask/boiling flask is a type of flask used as an item of laboratory glassware and is named after the city Florence. It is used as a container to hold liquids. A Florence flask has a round body, a long neck, and often a flat bottom. It is designed for uniform heating, boiling, distillation and ease of swirling; it is produced in a number of different glass thicknesses to stand different types of use. They are often made of borosilicate glass for heat and chemical resistance. Traditional Florence flasks typically do not have a ground glass joint on their rather longer necks, but typically have a slight lip or flange around the tip of the neck. The common volume for a Florence flask is 1 litre.

A test tube, also known as a culture tube or sample tube, is a common piece of laboratory glassware consisting of a finger-like length of glass or clear plastic tubing, open at the top and closed at the bottom.

A magnetic stirrer or magnetic mixer is a laboratory device that employs a rotating magnetic field to cause a stir bar immersed in a liquid to spin very quickly, thus stirring it. The rotating field may be created either by a rotating magnet or a set of stationary electromagnets, placed beneath the vessel with the liquid. It is used in chemistry and biology as a convenient way to stir small volumes and where other forms of stirring, such as overhead stirrers and stirring rods, may not be viable.
Fractional distillation is the separation of a mixture into its component parts, or fractions. Chemical compounds are separated by heating them to a temperature at which one or more fractions of the mixture will vaporize. It uses distillation to fractionate. Generally the component parts have boiling points that differ by less than 25 °C (45 °F) from each other under a pressure of one atmosphere. If the difference in boiling points is greater than 25 °C, a simple distillation is typically used. It is used to refine crude oil.
In thermodynamics, superheating is the phenomenon in which a liquid is heated to a temperature higher than its boiling point, without boiling. This is a so-called metastable state or metastate, where boiling might occur at any time, induced by external or internal effects. Superheating is achieved by heating a homogeneous substance in a clean container, free of nucleation sites, while taking care not to disturb the liquid.

A stopper is a cylindrical or conical closure used to seal a container, such as a bottle, tube or barrel. Unlike a lid or bottle cap, which encloses a container from the outside without displacing the inner volume, a bung is partially or wholly inserted inside the container to act as a seal. Synonyms are bung and cork. A bung can be defined as "a plug or closure used to close an opening in a drum or barrel. It is called a plug when referring to a steel drum closure."

A rotary evaporator (rotavap) is a device used in chemical laboratories for the efficient and gentle removal of solvents from samples by evaporation. When referenced in the chemistry research literature, description of the use of this technique and equipment may include the phrase "rotary evaporator", though use is often rather signaled by other language.

A hot plate or hotplate is a portable self-contained tabletop small appliance cooktop that features one or more electric heating elements or gas burners. A hot plate can be used as a stand-alone appliance, but is often used as a substitute for one of the burners from an oven range or a kitchen stove. Hot plates are often used for food preparation, generally in locations where a full kitchen stove would not be convenient or practical. They can also be used as a heat source in laboratories. A hot plate can have a flat surface or round surface. Hot plates can be used for traveling or in areas without electricity.

A boiling chip, boiling stone, porous bitanti-bumping granule is a tiny, unevenly shaped piece of substance added to liquids to make them boil more calmly. Boiling chips are frequently employed in distillation and heating. When a liquid becomes superheated, a speck of dust or a stirring rod can cause violent flash boiling. Boiling chips provide nucleation sites so the liquid boils smoothly without becoming superheated or bumping.
Degassing, also known as degasification, is the removal of dissolved gases from liquids, especially water or aqueous solutions. There are numerous methods for removing gases from liquids.

In thermodynamics, nucleation is the first step in the formation of either a new thermodynamic phase or structure via self-assembly or self-organization within a substance or mixture. Nucleation is typically defined to be the process that determines how long an observer has to wait before the new phase or self-organized structure appears. For example, if a volume of water is cooled below 0 °C, it will tend to freeze into ice, but volumes of water cooled only a few degrees below 0 °C often stay completely free of ice for long periods (supercooling). At these conditions, nucleation of ice is either slow or does not occur at all. However, at lower temperatures nucleation is fast, and ice crystals appear after little or no delay.
The Marcusson apparatus, Dean-Stark apparatus, Dean–Stark receiver, distilling trap, or Dean–Stark Head is a piece of laboratory glassware used in synthetic chemistry to collect water from a reactor. It is used in combination with a reflux condenser and a distillation flask for the separation of water from liquids. This may be a continuous removal of the water that is produced during a chemical reaction performed at reflux temperature, such as in esterification reactions. The original setup by Julius Marcusson was refined by the American chemists Ernest Woodward Dean (1888–1959) and David Dewey Stark (1893–1979) in 1920 for determination of the water content in petroleum.

Round-bottom flasks are types of flasks having spherical bottoms used as laboratory glassware, mostly for chemical or biochemical work. They are typically made of glass for chemical inertness; and in modern days, they are usually made of heat-resistant borosilicate glass. There is at least one tubular section known as the neck with an opening at the tip. Two- or three-necked flasks are common as well. Round bottom flasks come in many sizes, from 5 mL to 20 L, with the sizes usually inscribed on the glass. In pilot plants even larger flasks are encountered.

An eye dropper, also called Pasteur pipette or simply dropper, is a device used to transfer small quantities of liquids. They are used in the laboratory and also to dispense small amounts of liquid medicines. A very common use was to dispense eye drops into the eye. The commonly recognized form is a glass tube tapered to a narrow point and fitted with a rubber bulb at the top, although many styles of both plastic and glass droppers exist. The combination of the pipette and rubber bulb has also been referred to as a teat pipette. The Pasteur pipette name is from the French scientist Louis Pasteur, who used a variant of them extensively during his research. In the past, there was no equipment to transfer a chemical solution without exposing it to the external environment. The hygiene and purity of chemical compounds is necessary for the expected result of each experiment. The eye dropper, both glass and plastic types, can be sterilized and plugged with a rubber bulb at the open end of the pipette preventing any contamination from the atmosphere. Generally, they are considered cheap enough to be disposable, however, so long as the glass point is not chipped, the eye dropper may be washed and reused indefinitely.
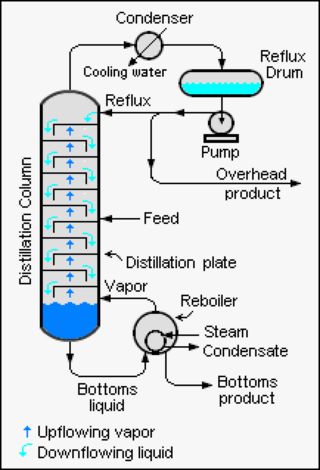
Reflux is a technique involving the condensation of vapors and the return of this condensate to the system from which it originated. It is used in industrial and laboratory distillations. It is also used in chemistry to supply energy to reactions over a long period of time.
A test tube holder is used to hold test tubes. It is used for holding a test tube in place when the tube is hot or should not be touched. For example, a test tube holder can be used to hold a test tube while it is being heated. Moreover, when heating the tube with liquid or solid contained inside, the holder ought to tightly hold a test tube in order for the tube to be safely held while heating.
References
- ↑ "Proper Heating of Test Tubes". CR Scientific LLC. Retrieved 30 October 2010.
- ↑ Moya-Barrios, Reinaldo (2015). CHEM 2401 Lab Manual. Dalhousie University Chemistry Department.
- ↑ Beran, Jo (1 November 2010). Laboratory Manual for Principles of General Chemistry. John Wiley and Sons. p. 25. ISBN 978-0-470-64789-9.