
The National Institutes of Health, commonly referred to as NIH, is the primary agency of the United States government responsible for biomedical and public health research. It was founded in the late 1880s and is now part of the United States Department of Health and Human Services. Many NIH facilities are located in Bethesda, Maryland, and other nearby suburbs of the Washington metropolitan area, with other primary facilities in the Research Triangle Park in North Carolina and smaller satellite facilities located around the United States. The NIH conducts its own scientific research through the NIH Intramural Research Program (IRP) and provides major biomedical research funding to non-NIH research facilities through its Extramural Research Program.

In biology and other experimental sciences, an in silico experiment is one performed on a computer or via computer simulation software. The phrase is pseudo-Latin for 'in silicon', referring to silicon in computer chips. It was coined in 1987 as an allusion to the Latin phrases in vivo, in vitro, and in situ, which are commonly used in biology. The latter phrases refer, respectively, to experiments done in living organisms, outside living organisms, and where they are found in nature.

l-Kynurenine is a metabolite of the amino acid l-tryptophan used in the production of niacin.

Indoleamine-pyrrole 2,3-dioxygenase (IDO or INDO EC 1.13.11.52) is a heme-containing enzyme physiologically expressed in a number of tissues and cells, such as the small intestine, lungs, female genital tract or placenta. In humans is encoded by the IDO1 gene. IDO is involved in tryptophan metabolism. It is one of three enzymes that catalyze the first and rate-limiting step in the kynurenine pathway, the O2-dependent oxidation of L-tryptophan to N-formylkynurenine, the others being indolamine-2,3-dioxygenase 2 (IDO2) and tryptophan 2,3-dioxygenase (TDO). IDO is an important part of the immune system and plays a part in natural defense against various pathogens. It is produced by the cells in response to inflammation and has an immunosuppressive function because of its ability to limit T-cell function and engage mechanisms of immune tolerance. Emerging evidence suggests that IDO becomes activated during tumor development, helping malignant cells escape eradication by the immune system. Expression of IDO has been described in a number of types of cancer, such as acute myeloid leukemia, ovarian cancer or colorectal cancer. IDO is part of the malignant transformation process and plays a key role in suppressing the anti-tumor immune response in the body, so inhibiting it could increase the effect of chemotherapy as well as other immunotherapeutic protocols. Furthermore, there is data implicating a role for IDO1 in the modulation of vascular tone in conditions of inflammation via a novel pathway involving singlet oxygen.
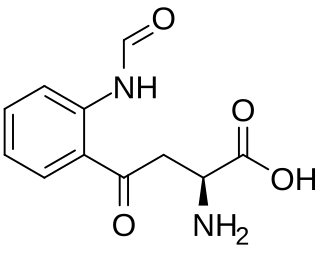
N′-Formylkynurenine is an intermediate in the catabolism of tryptophan. It is a formylated derivative of kynurenine. The formation of N′-formylkynurenine is catalyzed by heme dioxygenases.
In enzymology, tryptophan 2,3-dioxygenase (EC 1.13.11.11) is a heme enzyme that catalyzes the oxidation of L-tryptophan (L-Trp) to N-formyl-L-kynurenine, as the first and rate-limiting step of the kynurenine pathway.
Dioxygenases are oxidoreductase enzymes. Aerobic life, from simple single-celled bacteria species to complex eukaryotic organisms, has evolved to depend on the oxidizing power of dioxygen in various metabolic pathways. From energetic adenosine triphosphate (ATP) generation to xenobiotic degradation, the use of dioxygen as a biological oxidant is widespread and varied in the exact mechanism of its use. Enzymes employ many different schemes to use dioxygen, and this largely depends on the substrate and reaction at hand.

Kynurenine 3-monooxygenase is an enzyme that in humans is encoded by the KMO gene.
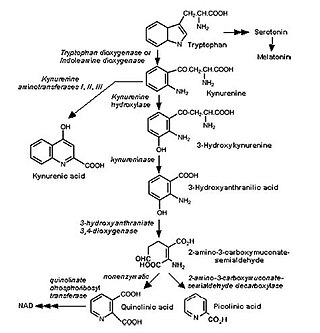
The kynurenine pathway is a metabolic pathway leading to the production of nicotinamide adenine dinucleotide (NAD+). Metabolites involved in the kynurenine pathway include tryptophan, kynurenine, kynurenic acid, xanthurenic acid, quinolinic acid, and 3-hydroxykynurenine. The kynurenine pathway is responsible for total catabolization of tryptophan about 95%. Disruption in the pathway is associated with certain genetic and psychiatric disorders.
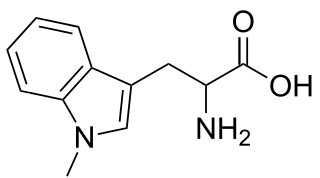
1-Methyltryptophan is a chemical compound that is an inhibitor of the tryptophan catabolic enzyme indoleamine 2,3-dioxygenase. It is a chiral compound that can exist as both D- and L-enantiomers.
Roland Stocker is a Swiss Australian biochemist who discovered the antioxidant activity of bilirubin. He is a former Olympic rower and has represented Switzerland at the 1980 Summer Olympics.

Incyte Corporation is an American multinational pharmaceutical company with headquarters in Wilmington, Delaware, and Morges, Switzerland. The company was created in 2002 through the merger of Incyte Pharmaceuticals, founded in Palo Alto, California in 1991 and Incyte Genomics, Inc. of Delaware. The company currently operates manufacturing and R&D locations in North America, Europe, and Asia.

George C. Prendergast is an American biomedical scientist. His research has focused on cancer pathobiology and immunology. Since 2004, he has been the President and CEO of Lankenau Institute for Medical Research, a cancer-focused research center in the U.S. He is also the co-director of the Program in Cancer Cell Biology & Signaling at the Sidney Kimmel Cancer Center, Thomas Jefferson University.

Immune checkpoints are regulators of the immune system. These pathways are crucial for self-tolerance, which prevents the immune system from attacking cells indiscriminately. However, some cancers can protect themselves from attack by stimulating immune checkpoint targets.

Charles Antzelevitch is an American cardiovascular research scientist in the fields of cardiac electrophysiology and cardiac arrhythmia syndromes.

Epacadostat is an investigational drug for cancer. Epacadostat is an inhibitor of indoleamine 2,3-dioxygenase-1 (IDO1). Epacadostat inhibits IDO1 by competitively blocking it, without interfering with IDO2 or tryptophan 2,3-dioxygenase (TDO). It has antitumor activity in some models, though is most effective when combined with other immunotherapy agents.

Scott K. Dessain is an American oncologist, research scientist, who is a professor at Lankenau Institute for Medical Research and an attending medical oncologist at Lankenau Medical Center, both in Wynnewood, Pennsylvania. He also is co-founder and chief technology officer of Immunome, Inc., a biotechnology company in Philadelphia specializing in development of native human cancer antibodies targeted against cancer antigens. Dessain developed a technology that caused cells to glow, which had been licensed for use by others.

Indoleamine 2,3-dioxygenase 2 (IDO2) is a protein that in humans is encoded by the IDO2 gene.

Gary S. Firestein is an American rheumatologist, professor, and founding director of the Altman Clinical and Translational Research Institute (ACTRI) at the University of California San Diego and Senior Associate Vice Chancellor for Health Sciences at University of California, San Diego.

Linrodostat is an experimental drug being studied for its immunomodulating and antineoplastic activities.



















