
In molecular biology and genetics, transformation is the genetic alteration of a cell resulting from the direct uptake and incorporation of exogenous genetic material from its surroundings through the cell membrane(s). For transformation to take place, the recipient bacterium must be in a state of competence, which might occur in nature as a time-limited response to environmental conditions such as starvation and cell density, and may also be induced in a laboratory.

Amphotericin B is an antifungal medication used for serious fungal infections and leishmaniasis. The fungal infections it is used to treat include mucormycosis, aspergillosis, blastomycosis, candidiasis, coccidioidomycosis, and cryptococcosis. For certain infections it is given with flucytosine. It is typically given intravenously.
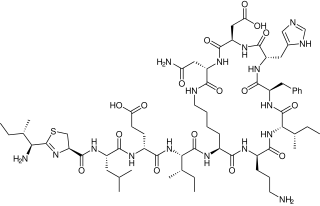
Bacitracin is a polypeptide antibiotic. It is a mixture of related cyclic peptides produced by Bacillus licheniformis bacteria, that was first isolated from the variety "Tracy I" in 1945. These peptides disrupt Gram-positive bacteria by interfering with cell wall and peptidoglycan synthesis.
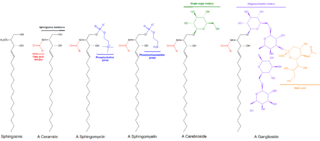
Sphingolipids are a class of lipids containing a backbone of sphingoid bases, which are a set of aliphatic amino alcohols that includes sphingosine. They were discovered in brain extracts in the 1870s and were named after the mythological sphinx because of their enigmatic nature. These compounds play important roles in signal transduction and cell recognition. Sphingolipidoses, or disorders of sphingolipid metabolism, have particular impact on neural tissue. A sphingolipid with a terminal hydroxyl group is a ceramide. Other common groups bonded to the terminal oxygen atom include phosphocholine, yielding a sphingomyelin, and various sugar monomers or dimers, yielding cerebrosides and globosides, respectively. Cerebrosides and globosides are collectively known as glycosphingolipids.

Gramicidin, also called gramicidin D, is a mix of ionophoric antibiotics, gramicidin A, B and C, which make up about 80%, 5%, and 15% of the mix, respectively. Each has 2 isoforms, so the mix has 6 different types of gramicidin molecules. They can be extracted from Brevibacillus brevis soil bacteria. Gramicidins are linear peptides with 15 amino acids. This is in contrast to unrelated gramicidin S, which is a cyclic peptide.

Fungemia is the presence of fungi or yeast in the blood. The most common type, also known as candidemia, candedemia, or systemic candidiasis, is caused by Candida species. Candidemia is also among the most common bloodstream infections of any kind. Infections by other fungi, including Saccharomyces, Aspergillus and Cryptococcus, are also called fungemia. It is most commonly seen in immunosuppressed or immunocompromised patients with severe neutropenia, cancer patients, or in patients with intravenous catheters. It has been suggested that otherwise immunocompetent patients taking infliximab may also be at a higher risk.
Paenibacillus polymyxa, also known as Bacillus polymyxa, is a Gram-positive bacterium capable of fixing nitrogen. It is found in soil, plant tissues, marine sediments and hot springs. It may have a role in forest ecosystems and potential future applications as a biofertilizer and biocontrol agent in agriculture.

Sphingomyelin phosphodiesterase is a hydrolase enzyme that is involved in sphingolipid metabolism reactions. SMase is a member of the DNase I superfamily of enzymes and is responsible for breaking sphingomyelin (SM) down into phosphocholine and ceramide. The activation of SMase has been suggested as a major route for the production of ceramide in response to cellular stresses.

Surfactin is a cyclic lipopeptide, commonly used as an antibiotic for its capacity as a surfactant. It is an amphiphile capable of withstanding hydrophilic and hydrophobic environments. The Gram-positive bacterial species Bacillus subtilis produces surfactin for its antibiotic effects against competitors. Surfactin showcases antibacterial, antiviral, antifungal, and hemolytic effects.

Echinocandins are a class of antifungal drugs that inhibit the synthesis of β-glucan in the fungal cell wall via noncompetitive inhibition of the enzyme 1,3-β glucan synthase. The class has been termed the "penicillin of antifungals," along with the related papulacandins, as their mechanism of action resembles that of penicillin in bacteria. β-glucans are carbohydrate polymers that are cross-linked with other fungal cell wall components, the fungal equivalent to bacterial peptidoglycan. Caspofungin, micafungin, and anidulafungin are semisynthetic echinocandin derivatives with limited clinical use due to their solubility, antifungal spectrum, and pharmacokinetic properties.

Microbial art, agar art, or germ art is artwork created by culturing microorganisms in certain patterns. The microbes used can be bacteria, yeast, fungi, or less commonly, protists. The microbes can be chosen for their natural colours or engineered to express fluorescent proteins and viewed under ultraviolet light to make them fluoresce in colour.
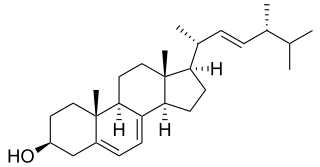
In enzymology, a sterol 14-demethylase (EC 1.14.13.70) is an enzyme of the cytochrome P450 (CYP) superfamily. It is any member of the CYP51 family. It catalyzes a chemical reaction such as:
A lipopeptide is a molecule consisting of a lipid connected to a peptide. They are able to self-assemble into different structures. Many bacteria produce these molecules as a part of their metabolism, especially those of the genus Bacillus, Pseudomonas and Streptomyces. Certain lipopeptides are used as antibiotics. Due to the structural and molecular properties such as the fatty acid chain, it poses the effect of weakening the cell function or destroying the cell. Other lipopeptides are toll-like receptor agonists. Certain lipopeptides can have strong antifungal and hemolytic activities. It has been demonstrated that their activity is generally linked to interactions with the plasma membrane, and sterol components of the plasma membrane could play a major role in this interaction. It is a general trend that adding a lipid group of a certain length to a lipopeptide will increase its bactericidal activity. Lipopeptides with a higher amount of carbon atoms, for example 14 or 16, in its lipid tail will typically have antibacterial activity as well as anti-fungal activity. Therefore, an increase in the alkyl chain can make lipopeptides soluble in water. As well, it opens the cell membrane of the bacteria, so antimicrobial activity can take place.
A killer yeast is a yeast, such as Saccharomyces cerevisiae, which is able to secrete one of a number of toxic proteins which are lethal to susceptible cells. These "killer toxins" are polypeptides that kill sensitive cells of the same or related species, often functioning by creating pores in target cell membranes. These yeast cells are immune to the toxic effects of the protein due to an intrinsic immunity. Killer yeast strains can be a problem in commercial processing because they can kill desirable strains. The killer yeast system was first described in 1963. Study of killer toxins helped to better understand the secretion pathway of yeast, which is similar to those of more complex eukaryotes. It also can be used in treatment of some diseases, mainly those caused by fungi.
Histatins are histidine-rich (cationic) antimicrobial proteins found in saliva. Histatin's involvement in antimicrobial activities makes histatin part of the innate immune system.
Protegrins are small peptides containing 16-18 amino acid residues. Protegrins were first discovered in porcine leukocytes and were found to have antimicrobial activity against bacteria, fungi, and some enveloped viruses. The amino acid composition of protegrins contains six positively charged arginine residues and four cysteine residues. Their secondary structure is classified as cysteine-rich β-sheet antimicrobial peptides, AMPs, that display limited sequence similarity to certain defensins and tachyplesins. In solution, the peptides fold to form an anti-parallel β-strand with the structure stabilized by two cysteine bridges formed among the four cysteine residues. Recent studies suggest that protegrins can bind to lipopolysaccharide, a property that may help them to insert into the membranes of gram-negative bacteria and permeabilize them.

Bacillomycins are a group of antifungal polypeptide antibiotics isolated from Bacillus subtilis.

Bacillus subtilis is a rod-shaped, Gram-positive bacteria that is naturally found in soil and vegetation, and is known for its ability to form a small, tough, protective and metabolically dormant endospore. B. subtilis can divide symmetrically to make two daughter cells, or asymmetrically, producing a single endospore that is resistant to environmental factors such as heat, desiccation, radiation and chemical insult which can persist in the environment for long periods of time. The endospore is formed at times of nutritional stress, allowing the organism to persist in the environment until conditions become favourable. The process of endospore formation has profound morphological and physiological consequences: radical post-replicative remodelling of two progeny cells, accompanied eventually by cessation of metabolic activity in one daughter cell and death by lysis of the other.

C-5 sterol desaturase is an enzyme that is highly conserved among eukaryotes and catalyzes the dehydrogenation of a C-5(6) bond in a sterol intermediate compound as a step in the biosynthesis of major sterols. The precise structure of the enzyme's substrate varies by species. For example, the human C-5 sterol desaturase oxidizes lathosterol, while its ortholog ERG3 in the yeast Saccharomyces cerevisiae oxidizes episterol.
A proteolipid is a protein covalently linked to lipid molecules, which can be fatty acids, isoprenoids or sterols. The process of such a linkage is known as protein lipidation, and falls into the wider category of acylation and post-translational modification. Proteolipids are abundant in brain tissue, and are also present in many other animal and plant tissues. They include ghrelin, a peptide hormone associated with feeding. Many proteolipids are composed of proteins covalenently bound to fatty acid chains, often granting them an interface for interacting with biological membranes. They are not to be confused with lipoproteins, a kind of spherical assembly made up of many molecules of lipids and some apolipoproteins.














