
An ester is a chemical compound derived from an acid in which at least one –OH hydroxyl group is replaced by an –O– alkyl (alkoxy) group, as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils.
An emulsion is a mixture of two or more liquids that are normally immiscible owing to liquid-liquid phase separation. Emulsions are part of a more general class of two-phase systems of matter called colloids. Although the terms colloid and emulsion are sometimes used interchangeably, emulsion should be used when both phases, dispersed and continuous, are liquids. In an emulsion, one liquid is dispersed in the other. Examples of emulsions include vinaigrettes, homogenized milk, liquid biomolecular condensates, and some cutting fluids for metal working.
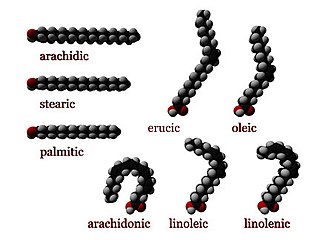
In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, from 4 to 28. Fatty acids are a major component of the lipids in some species such as microalgae but in some other organisms are not found in their standalone form, but instead exist as three main classes of esters: triglycerides, phospholipids, and cholesteryl esters. In any of these forms, fatty acids are both important dietary sources of fuel for animals and important structural components for cells.

Surfactants are compounds that lower the surface tension between two liquids, between a gas and a liquid, or between a liquid and a solid. Surfactants may act as detergents, wetting agents, emulsifiers, foaming agents, or dispersants. The word "surfactant" is a blend of surface-active agent, coined c. 1950.
Sarcosine, also known as N-methylglycine, is the organic compound with the formula CH3N(H)CH2CO2H. It exists at neutral pH as the zwitterion CH3N+(H)2CH2CO2-, which can be obtained as a white, water-soluble powder. Like some other amino acids, sarcosine converts to a cation at low pH and an anion at high pH, with the respective formulas CH3N+(H)2CH2CO2H and CH3N(H)CH2CO2-. Sarcosine is a close relative of glycine, with a secondary amine in place of the primary amine.

Lecithin is a generic term to designate any group of yellow-brownish fatty substances occurring in animal and plant tissues which are amphiphilic – they attract both water and fatty substances, and are used for smoothing food textures, emulsifying, homogenizing liquid mixtures, and repelling sticking materials.

Laundry detergent is a type of detergent used for cleaning dirty laundry (clothes). Laundry detergent is manufactured in powder and liquid form.
Tall oil, also called liquid rosin or tallol, is a viscous yellow-black odorous liquid obtained as a by-product of the kraft process of wood pulp manufacture when pulping mainly coniferous trees. The name originated as an anglicization of the Swedish tallolja. Tall oil is the third largest chemical by-product in a kraft mill after lignin and hemicellulose; the yield of crude tall oil from the process is in the range of 30–50 kg / ton pulp. It may contribute to 1.0–1.5% of the mill's revenue if not used internally.

Sodium formate, HCOONa, is the sodium salt of formic acid, HCOOH. It usually appears as a white deliquescent powder.

Docusate is the common chemical and pharmaceutical name of the anion bis(2-ethylhexyl) sulfosuccinate, also commonly called dioctyl sulfosuccinate (DOSS). It is on the World Health Organization's List of Essential Medicines. Salts of this anion, especially docusate sodium, are widely used in medicine as laxatives and as stool softeners, by mouth or rectally. In 2019, it was the 187th most commonly prescribed medication in the United States, with more than 3 million prescriptions. Some studies claim that docusate is not more effective than a placebo for improving constipation. Other docusate salts with medical use include those of calcium and potassium.

2-Acrylamido-2-methylpropane sulfonic acid (AMPS) was a Trademark name by The Lubrizol Corporation. It is a reactive, hydrophilic, sulfonic acid acrylic monomer used to alter the chemical properties of wide variety of anionic polymers. In the 1970s, the earliest patents using this monomer were filed for acrylic fiber manufacturing. Today, there are over several thousands patents and publications involving use of AMPS in many areas including water treatment, oil field, construction chemicals, hydrogels for medical applications, personal care products, emulsion coatings, adhesives, and rheology modifiers.
Lactylates are organic compounds that are FDA approved for use as food additives and cosmetic ingredients, e.g. as food-grade emulsifiers. These additives are non-toxic, biodegradable, and typically manufactured using biorenewable feedstocks. Owing to their safety and versatile functionality, lactylates are used in a wide variety of food and non-food applications. In the United States, the Food Chemicals Codex specifies the labeling requirements for food ingredients including lactylates. In the European Union, lactylates must be labelled in accordance with the requirements of the applicable EU regulation. Lactylates may be labelled as calcium stearoyl lactylate (CSL), sodium stearoyl lactylate (SSL), or lactylic esters of fatty acids (LEFA).
Copper naphthenate is the copper salt of naphthenic acid. Naphthenic acid is a term commonly used in the petroleum industry to collectively refer to all of the carboxylic acids naturally occurring in crude oil. Naphthenic acids are primarily cycloaliphatic carboxylic acids with 10 to 24 or more carbons, although substantial quantities of non-cyclic, aromatic and heteroatom- containing carboxylic acids are also present. Copper naphthenate is most widely used in wood preservation and for protecting other cellulosic materials such as textiles and cordage from damage by decay fungi and insects. Other metal naphthenates are used as paint driers, rubber adhesion promoters, lubricant additives, and catalysts where oil solubility is required.
Saltwater soap, also called sailors' soap, is a potassium-based soap for use with seawater. Inexpensive common commercial soap will not lather or dissolve in seawater due to high levels of sodium chloride in the water. Similarly, common soap does not work as well as potassium-based soap in hard water where calcium replaces the sodium, making residual insoluble "scum" due to the insolubility of the soap residue. To be an effective cleaning agent, soap must be able to dissolve in water.

Dimethylcarbamoyl chloride (DMCC) is a reagent for transferring a dimethylcarbamoyl group to alcoholic or phenolic hydroxyl groups forming dimethyl carbamates, usually having pharmacological or pesticidal activities. Because of its high toxicity and its carcinogenic properties shown in animal experiments and presumably also in humans, dimethylcarbamoyl chloride can only be used under stringent safety precautions.

Alkenyl succinic anhydrides (ASA) are modified five-membered succinic anhydrides bearing a branched iso-alkenyl chain (C14 to C22). They are colorless, and usually viscous liquids. They are widely used, especially in surface sizing of paper, paperboard, and cardboard, as well as in the hydrophobicization of cellulose fibers. Products treated with it show reduced penetration of aqueous media, such as inks or drinks (like milk or fruit juices).

N,N,N′,N′-Tetramethylformamidinium chloride is the simplest representative of quaternary formamidinium cations of the general formula [R2N−CH=NR2]+ with a chloride as a counterion in which all hydrogen atoms of the protonated formamidine [HC(=NH2)NH2]+ are replaced by methyl groups.
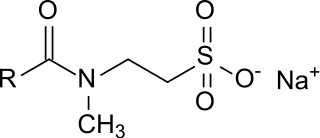
Taurates (or taurides) are a group of mild anionic surfactants. They are composed of a hydrophilic head group, consisting of N-methyltaurine (2-methylaminoethanesulfonic acid) and a lipophilic residue, consisting of a long-chain carboxylic acid (fatty acid), both linked via an amide bond. The fatty acids used could be lauric (C12), myristic (C14), palmitic (C16) or stearic acid (C18), but mainly mixtures of oleic acid (C18:1) and coconut fatty acid (C8 – C18) are used. Besides sodium, no other counterions play a relevant role (these could be e. g. ammonium or other alkali or alkaline earth metals).
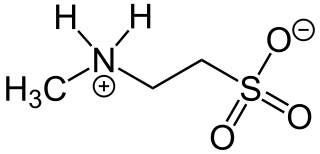
N-Methyltaurine is an aminosulfonic acid which is present as a zwitterion in the crystalline state and in polar solvents. In contrast to the widespread taurine, N-methyltaurine has been found in nature only in red algae, where it is formed by methylation of taurine. It is suitable for esterification with long-chain carboxylic acids to taurides (acylaminoethansulfonaten) because of its high polarity and the relatively good solubility of its alkaline earth metal salts, which are also used as mild anionic surfactants.
Isethionates are esters of long-chain aliphatic carboxylic acids (C8 – C18) with isethionic acid (2-hydroxyethanesulfonic acid) or salts thereof, such as ammonium isethionate or sodium isethionate. They are also referred to as acyl isethionates or acyloxyethanesulfonates.
















