Related Research Articles

The prostaglandins (PG) are a group of physiologically active lipid compounds called eicosanoids having diverse hormone-like effects in animals. Prostaglandins have been found in almost every tissue in humans and other animals. They are derived enzymatically from the fatty acid arachidonic acid. Every prostaglandin contains 20 carbon atoms, including a 5-carbon ring. They are a subclass of eicosanoids and of the prostanoid class of fatty acid derivatives.
Essential fatty acids, or EFAs, are fatty acids that humans and other animals must ingest because the body requires them for good health but cannot synthesize them.

Arachidonic acid is a polyunsaturated omega-6 fatty acid 20:4(ω-6), or 20:4(5,8,11,14). It is structurally related to the saturated arachidic acid found in cupuaçu butter. Its name derives from the New Latin word arachis (peanut), but it is important to note that peanut oil does not contain any arachidonic acid.

Eicosanoids are signaling molecules made by the enzymatic or non-enzymatic oxidation of arachidonic acid or other polyunsaturated fatty acids (PUFAs) that are, similar to arachidonic acid, 20 carbon units in length. Eicosanoids are a sub-category of oxylipins, i.e. oxidized fatty acids of diverse carbon units in length, and are distinguished from other oxylipins by their overwhelming importance as cell signaling molecules. Eicosanoids function in diverse physiological systems and pathological processes such as: mounting or inhibiting inflammation, allergy, fever and other immune responses; regulating the abortion of pregnancy and normal childbirth; contributing to the perception of pain; regulating cell growth; controlling blood pressure; and modulating the regional flow of blood to tissues. In performing these roles, eicosanoids most often act as autocrine signaling agents to impact their cells of origin or as paracrine signaling agents to impact cells in the proximity of their cells of origin. Eicosanoids may also act as endocrine agents to control the function of distant cells.

A lipoxin (LX or Lx), an acronym for lipoxygenase interaction product, is a bioactive autacoid metabolite of arachidonic acid made by various cell types. They are categorized as nonclassic eicosanoids and members of the specialized pro-resolving mediators (SPMs) family of polyunsaturated fatty acid (PUFA) metabolites. Like other SPMs, LXs form during, and then act to resolve, inflammatory responses. Initially, two lipoxins were identified, lipoxin A4 (LXA4) and LXB4, but more recent studies have identified epimers of these two LXs: the epi-lipoxins, 15-epi-LXA4 and 15-epi-LXB4 respectively.

Lipoxygenases are a family of (non-heme) iron-containing enzymes most of which catalyze the dioxygenation of polyunsaturated fatty acids in lipids containing a cis,cis-1,4- pentadiene into cell signaling agents that serve diverse roles as autocrine signals that regulate the function of their parent cells, paracrine signals that regulate the function of nearby cells, and endocrine signals that regulate the function of distant cells.
Prostanoids are a subclass of eicosanoids consisting of the prostaglandins, the thromboxanes, and the prostacyclins
Dihomo-γ-linolenic acid (DGLA) is a 20-carbon ω−6 fatty acid. In physiological literature, it is given the name 20:3 (ω−6). DGLA is a carboxylic acid with a 20-carbon chain and three cis double bonds; the first double bond is located at the sixth carbon from the omega end. DGLA is the elongation product of γ-linolenic acid. GLA, in turn, is a desaturation product of linoleic acid. DGLA is made in the body by the elongation of GLA, by an efficient enzyme which does not appear to suffer any form of (dietary) inhibition. DGLA is an extremely uncommon fatty acid, found only in trace amounts in animal products.
The epoxyeicosatrienoic acids or EETs are signaling molecules formed within various types of cells by the metabolism of arachidonic acid by a specific subset of Cytochrome P450 enzymes termed cytochrome P450 epoxygenases. These nonclassic eicosanoids are generally short-lived, being rapidly converted from epoxides to less active or inactive dihydroxy-eicosatrienoic acids (diHETrEs) by a widely distributed cellular enzyme, Soluble epoxide hydrolase (sEH), also termed Epoxide hydrolase 2. The EETs consequently function as transiently acting, short-range hormones; that is, they work locally to regulate the function of the cells that produce them or of nearby cells. The EETs have been most studied in animal models where they show the ability to lower blood pressure possibly by a) stimulating arterial vasorelaxation and b) inhibiting the kidney's retention of salts and water to decrease intravascular blood volume. In these models, EETs prevent arterial occlusive diseases such as heart attacks and brain strokes not only by their anti-hypertension action but possibly also by their anti-inflammatory effects on blood vessels, their inhibition of platelet activation and thereby blood clotting, and/or their promotion of pro-fibrinolytic removal of blood clots. With respect to their effects on the heart, the EETs are often termed cardio-protective. Beyond these cardiovascular actions that may prevent various cardiovascular diseases, studies have implicated the EETs in the pathological growth of certain types of cancer and in the physiological and possibly pathological perception of neuropathic pain. While studies to date imply that the EETs, EET-forming epoxygenases, and EET-inactivating sEH can be manipulated to control a wide range of human diseases, clinical studies have yet to prove this. Determination of the role of the EETS in human diseases is made particularly difficult because of the large number of EET-forming epoxygenases, large number of epoxygenase substrates other than arachidonic acid, and the large number of activities, some of which may be pathological or injurious, that the EETs possess.
The effects on humans of the ω-3 (omega-3) and ω-6 (omega-6) essential fatty acids (EFAs) are best characterized by their interactions.
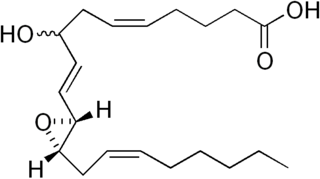
Hepoxilins (Hx) are a set of epoxyalcohol metabolites of polyunsaturated fatty acids (PUFA), i.e. they possess both an epoxide and an alcohol residue. HxA3, HxB3, and their non-enzymatically formed isomers are nonclassic eicosanoid derived from acid the (PUFA), arachidonic acid. A second group of less well studied hepoxilins, HxA4, HxB4, and their non-enzymatically formed isomers are nonclassical eicosanoids derived from the PUFA, eicosapentaenoic acid. Recently, 14,15-HxA3 and 14,15-HxB3 have been defined as arachidonic acid derivatives that are produced by a different metabolic pathway than HxA3, HxB3, HxA4, or HxB4 and differ from the aforementioned hepoxilins in the positions of their hydroxyl and epoxide residues. Finally, hepoxilin-like products of two other PUFAs, docosahexaenoic acid and linoleic acid, have been described. All of these epoxyalcohol metabolites are at least somewhat unstable and are readily enzymatically or non-enzymatically to their corresponding trihydroxy counterparts, the trioxilins (TrX). HxA3 and HxB3, in particular, are being rapidly metabolized to TrXA3, TrXB3, and TrXC3. Hepoxilins have various biological activities in animal models and/or cultured mammalian tissues and cells. The TrX metabolites of HxA3 and HxB3 have less or no activity in most of the systems studied but in some systems retain the activity of their precursor hepoxilins. Based on these studies, it has been proposed that the hepoxilins and trioxilins function in human physiology and pathology by, for example, promoting inflammation responses and dilating arteries to regulate regional blood flow and blood pressure.
The isoprostanes are prostaglandin-like compounds formed in vivo from the free radical-catalyzed peroxidation of essential fatty acids without the direct action of cyclooxygenase (COX) enzymes. The compounds were discovered in 1990 by L. Jackson Roberts and Jason D. Morrow in the Division of Clinical Pharmacology at Vanderbilt University. These nonclassical eicosanoids possess potent biological activity as inflammatory mediators that augment the perception of pain. These compounds are accurate markers of lipid peroxidation in both animal and human models of oxidative stress.
Most of the eicosanoid receptors are integral membrane protein G protein-coupled receptors (GPCRs) that bind and respond to eicosanoid signaling molecules. Eicosanoids are rapidly metabolized to inactive products and therefore are short-lived. Accordingly, the eicosanoid-receptor interaction is typically limited to a local interaction: cells, upon stimulation, metabolize arachidonic acid to an eicosanoid which then binds cognate receptors on either its parent cell or on nearby cells to trigger functional responses within a restricted tissue area, e.g. an inflammatory response to an invading pathogen. In some cases, however, the synthesized eicosanoid travels through the blood to trigger systemic or coordinated tissue responses, e.g. prostaglandin (PG) E2 released locally travels to the hypothalamus to trigger a febrile reaction. An example of a non-GPCR receptor that binds many eicosanoids is the PPAR-γ nuclear receptor.
Prostaglandin receptors or prostanoid receptors represent a sub-class of cell surface membrane receptors that are regarded as the primary receptors for one or more of the classical, naturally occurring prostanoids viz., prostaglandin D2,, PGE2, PGF2alpha, prostacyclin (PGI2), thromboxane A2 (TXA2), and PGH2. They are named based on the prostanoid to which they preferentially bind and respond, e.g. the receptor responsive to PGI2 at lower concentrations than any other prostanoid is named the Prostacyclin receptor (IP). One exception to this rule is the receptor for thromboxane A2 (TP) which binds and responds to PGH2 and TXA2 equally well.

ALOX15 is, like other lipoxygenases, a seminal enzyme in the metabolism of polyunsaturated fatty acids to a wide range of physiologically and pathologically important products. ▼ Gene Function
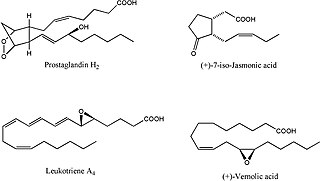
Oxylipins constitute a family of oxygenated natural products which are formed from fatty acids by pathways involving at least one step of dioxygen-dependent oxidation. Oxylipins are derived from polyunsaturated fatty acids (PUFAs) by COX enzymes (cyclooxygenases), by LOX enzymes (lipoxygenases), or by cytochrome P450 epoxygenase.
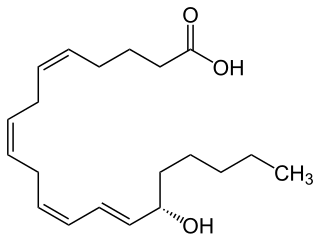
15-Hydroxyeicosatetraenoic acid is an eicosanoid, i.e. a metabolite of arachidonic acid. Various cell types metabolize arachidonic acid to 15(S)-hydroperoxyeicosatetraenoic acid. This initial hydroperoxide product is extremely short-lived in cells: if not otherwise metabolized, it is rapidly reduced to 15(S)-HETE. Both of these metabolites, depending on the cell type which forms them, can be further metabolized to 15-oxo-eicosatetraenoic acid (15-oxo-ETE), 5S,15S-dihydroxy-eicosatetraenoic acid, 5-oxo-15(S)-hydroxyeicosatetraenoic acid (5-oxo-15 -HETE, a subset of specialized pro-resolving mediators viz., the lipoxins, a class of pro-inflammatory mediators, the eoxins, and other products that have less well-defined activities and functions. Thus, 15 -HETE and 15 -HpETE, in addition to having intrinsic biological activities, are key precursors to numerous biologically active derivatives.

Eoxin C4, also known as 14,15-leukotriene C4, is an eoxin. Cells make eoxins by metabolizing arachidonic acid with a 15-lipoxygenase enzyme to form 15(S)-hydroperoxyeicosapentaenoic acid. This product is then converted serially to eoxin A4, EXC4, EXD4, and EXE4 by LTC4 synthase, an unidentified gamma-glutamyltransferase, and an unidentified dipeptidase, respectively, in a pathway which appears similar if not identical to the pathway which forms leukotreines, i.e. LTA4, LTC4, LTD4, and LTE4. This pathway is schematically shown as follows:

Eoxin E4, also known as 14,15-leukotriene E4, is an eoxin. Cells make eoxins by metabolizing arachidonic acid with a 15-lipoxygenase enzyme to form 15(S)-hydroperoxyeicosapentaenoic acid. This product is then converted serially to eoxin A4, EXC4, EXD4, and EXE4 by LTC4 synthase, an unidentified gamma-glutamyltransferase, and an unidentified dipeptidase, respectively, in a pathway which appears similar if not identical to the pathway which forms leukotreines, i.e. LTA4, LTC4, LTD4, and LTE4. This pathway is schematically shown as follows:
Specialized pro-resolving mediators are a large and growing class of cell signaling molecules formed in cells by the metabolism of polyunsaturated fatty acids (PUFA) by one or a combination of lipoxygenase, cyclooxygenase, and cytochrome P450 monooxygenase enzymes. Pre-clinical studies, primarily in animal models and human tissues, implicate SPM in orchestrating the resolution of inflammation. Prominent members include the resolvins and protectins.
References
- ↑ Funk, Colin D. (30 November 2001). "Prostaglandins and Leukotrienes: Advances in Eicosanoid Biology". Science. 294 (5548): 1871–1875. doi:10.1126/science.294.5548.1871. PMID 11729303.
- ↑ Van Dyke TE, Serhan CN (2003). "Resolution of inflammation: a new paradigm for the pathogenesis of periodontal diseases". J. Dent. Res. 82 (2): 82–90. doi:10.1177/154405910308200202. PMID 12562878.
- ↑ Serhan CN, Gotlinger K, Hong S, Arita M (2004). "Resolvins, docosatrienes, and neuroprotectins, novel omega-3-derived mediators, and their aspirin-triggered endogenous epimers: an overview of their protective roles in catabasis". Prostaglandins Other Lipid Mediat. 73 (3–4): 155–72. doi:10.1016/j.prostaglandins.2004.03.005. PMID 15290791.
- ↑ Anderle P, Farmer P, Berger A, Roberts MA (2004). "Nutrigenomic approach to understanding the mechanisms by which dietary long-chain fatty acids induce gene signals and control mechanisms involved in carcinogenesis". Nutrition (Burbank, Los Angeles County, Calif.). 20 (1): 103–8. doi:10.1016/j.nut.2003.09.018. PMID 14698023.
- ↑ Evans AR, Junger H, Southall MD, et al. (2000). "Isoprostanes, novel eicosanoids that produce nociception and sensitize rat sensory neurons". J. Pharmacol. Exp. Ther. 293 (3): 912–20. PMID 10869392.
- ↑ O'Brien WF, Krammer J, O'Leary TD, Mastrogiannis DS (1993). "The effect of acetaminophen on prostacyclin production in pregnant women". Am. J. Obstet. Gynecol. 168 (4): 1164–9. doi:10.1016/0002-9378(93)90362-m. PMID 8475962.
- ↑ Behrendt H, Kasche A, Ebner von Eschenbach C, Risse U, Huss-Marp J, Ring J (2001). "Secretion of proinflammatory eicosanoid-like substances precedes allergen release from pollen grains in the initiation of allergic sensitization". Int. Arch. Allergy Immunol. 124 (1–3): 121–5. doi:10.1159/000053688. PMID 11306946.
- ↑ Sarau HM, Foley JJ, Schmidt DB, et al. (1999). "In vitro and in vivo pharmacological characterization of SB 201993, an eicosanoid-like LTB4 receptor antagonist with anti-inflammatory activity". Prostaglandins Leukot. Essent. Fatty Acids. 61 (1): 55–64. doi:10.1054/plef.1999.0074. PMID 10477044.
- ↑ Cyberlipid Center. "Prostanoids and Related Products". Archived from the original on 2007-02-08. Retrieved 2007-11-02.