| |
| Clinical data | |
|---|---|
| Other names | Mikamycin A; Virginiamycin M1 |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C28H35N3O7 |
| Molar mass | 525.602 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
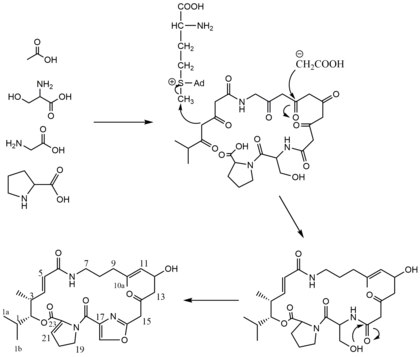
Pristinamycin IIA is a macrolide antibiotic. It is a member of the streptogramin A group of antibiotics and one component of pristinamycin (the other being pristinamycin IA). [1] Pristinamycin IIA was first isolated from the Streptomyces virginiae, but has been isolated from other microorganisms and thus has been given a variety of other names such as Virginiamycin M1, Mikamycin A, and Streptogramin A. [2] Pristinamycin IIA structure was determined by chemical and instrumental techniques, including X-ray crystallography. [2] [3] Pristinamycin IIA is of interest from a biosynthetic viewpoint because it contains the unusual dehydroproline and oxazole ring systems. [2] The only experimental evidence bearing on the formation of the oxazole ring is found in work on the biosynthesis of the alkaloid annuloline. [2] [4]