Meconium aspiration syndrome (MAS) also known as neonatal aspiration of meconium is a medical condition affecting newborn infants. It describes the spectrum of disorders and pathophysiology of newborns born in meconium-stained amniotic fluid (MSAF) and have meconium within their lungs. Therefore, MAS has a wide range of severity depending on what conditions and complications develop after parturition. Furthermore, the pathophysiology of MAS is multifactorial and extremely complex which is why it is the leading cause of morbidity and mortality in term infants.
Acute respiratory distress syndrome (ARDS) is a type of respiratory failure characterized by rapid onset of widespread inflammation in the lungs. Symptoms include shortness of breath (dyspnea), rapid breathing (tachypnea), and bluish skin coloration (cyanosis). For those who survive, a decreased quality of life is common.

Infantile respiratory distress syndrome (IRDS), also called respiratory distress syndrome of newborn, or increasingly surfactant deficiency disorder (SDD), and previously called hyaline membrane disease (HMD), is a syndrome in premature infants caused by developmental insufficiency of pulmonary surfactant production and structural immaturity in the lungs. It can also be a consequence of neonatal infection and can result from a genetic problem with the production of surfactant-associated proteins.

Liquid breathing is a form of respiration in which a normally air-breathing organism breathes an oxygen-rich liquid (such as a perfluorocarbon), rather than breathing air, by selecting a liquid that can hold a large amount of oxygen and is capable of CO2 gas exchange.

Pulmonary surfactant is a surface-active complex of phospholipids and proteins formed by type II alveolar cells. The proteins and lipids that make up the surfactant have both hydrophilic and hydrophobic regions. By adsorbing to the air-water interface of alveoli, with hydrophilic head groups in the water and the hydrophobic tails facing towards the air, the main lipid component of surfactant, dipalmitoylphosphatidylcholine (DPPC), reduces surface tension.
Antenatal steroids, also known as antenatal corticosteroids, are medications administered to pregnant women expecting a preterm birth. When administered, these steroids accelerate the maturation of the fetus' lungs, which reduces the likelihood of infant respiratory distress syndrome and infant mortality. The effectiveness of this corticosteroid treatment on humans was first demonstrated in 1972 by Sir Graham Liggins and Ross Howie, during a randomized control trial using betamethasone.
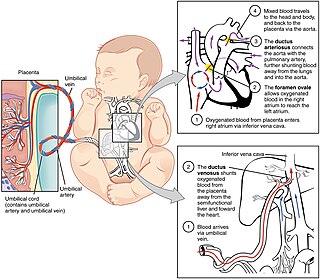
In humans, the circulatory system is different before and after birth. The fetal circulation is composed of the placenta, umbilical blood vessels encapsulated by the umbilical cord, heart and systemic blood vessels. A major difference between the fetal circulation and postnatal circulation is that the lungs are not used during the fetal stage resulting in the presence of shunts to move oxygenated blood and nutrients from the placenta to the fetal tissue. At birth, the start of breathing and the severance of the umbilical cord prompt various changes that quickly transform fetal circulation into postnatal circulation.

Respiratory diseases, or lung diseases, are pathological conditions affecting the organs and tissues that make gas exchange difficult in air-breathing animals. They include conditions of the respiratory tract including the trachea, bronchi, bronchioles, alveoli, pleurae, pleural cavity, the nerves and muscles of respiration. Respiratory diseases range from mild and self-limiting, such as the common cold, influenza, and pharyngitis to life-threatening diseases such as bacterial pneumonia, pulmonary embolism, tuberculosis, acute asthma, lung cancer, and severe acute respiratory syndromes, such as COVID-19. Respiratory diseases can be classified in many different ways, including by the organ or tissue involved, by the type and pattern of associated signs and symptoms, or by the cause of the disease.
Bronchoalveolar lavage (BAL), also known as bronchoalveolar washing, is a diagnostic method of the lower respiratory system in which a bronchoscope is passed through the mouth or nose into an appropriate airway in the lungs, with a measured amount of fluid introduced and then collected for examination. This method is typically performed to diagnose pathogenic infections of the lower respiratory airways, though it also has been shown to have utility in diagnosing interstitial lung disease. Bronchoalveolar lavage can be a more sensitive method of detection than nasal swabs in respiratory molecular diagnostics, as has been the case with SARS-CoV-2 where bronchoalveolar lavage samples detect copies of viral RNA after negative nasal swab testing.

Bronchopulmonary dysplasia is a chronic lung disease which affects premature infants. Premature (preterm) infants who require treatment with supplemental oxygen or require long-term oxygen are at a higher risk. The alveoli that are present tend to not be mature enough to function normally. It is also more common in infants with low birth weight (LBW) and those who receive prolonged mechanical ventilation to treat respiratory distress syndrome. It results in significant morbidity and mortality. The definition of bronchopulmonary dysplasia has continued to evolve primarily due to changes in the population, such as more survivors at earlier gestational ages, and improved neonatal management including surfactant, antenatal glucocorticoid therapy, and less aggressive mechanical ventilation.
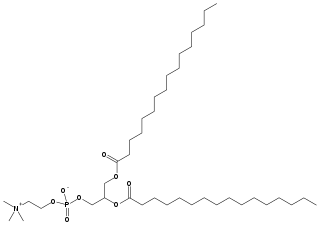
Dipalmitoylphosphatidylcholine (DPPC) is a phospholipid (and a lecithin) consisting of two C16 palmitic acid groups attached to a phosphatidylcholine head-group.

Surfactant protein B is an essential lipid-associated protein found in pulmonary surfactant. Without it, the lung would not be able to inflate after a deep breath out. It rearranges lipid molecules in the fluid lining the lung so that tiny air sacs in the lung, called alveoli, can more easily inflate.

Surfactant protein A1(SP-A1), also known as Pulmonary surfactant-associated protein A1(PSP-A) is a protein that in humans is encoded by the SFTPA1 gene.
Persistent fetal circulation is a condition caused by a failure in the systemic circulation and pulmonary circulation to convert from the antenatal circulation pattern to the "normal" pattern. Infants experience a high mean arterial pulmonary artery pressure and a high afterload at the right ventricle. This means that the heart is working against higher pressures, which makes it more difficult for the heart to pump blood.
Lucinactant is a liquid medication used to treat infant respiratory distress syndrome. It is a pulmonary surfactant for infants who lack enough natural surfactant in their lungs. Whereas earlier medicines of the class, such as beractant, calfactant (Infasurf), and poractant (Curosurf), are derived from animals, lucinactant is synthetic. It was approved for use in the United States by the U.S. Food and Drug Administration (FDA) on March 6, 2012.
Poractant alfa is a pulmonary surfactant sold under the brand name Curosurf by Chiesi Farmaceutici. Poractant alfa is an extract of natural porcine lung surfactant. As with other surfactants, marked improvement on oxygenation may occur within minutes of the administration of poractant alfa. The new generic form of surfactant is Varasurf developed in PersisGen Co. and commercialized by ArnaGen Pharmad. It has fully comparable quality profile with Curosurf.
Beractant, also known by the trade name of Survanta, is a modified bovine pulmonary surfactant containing bovine lung extract, to which synthetic DPPC, tripalmitin and palmitic acid are added. The composition provides 25 mg/mL phospholipids, 0.5 to 1.75 mg/mL triglycerides, 1.4 to 3.5 mg/mL free fatty acids, and <1.0 mg/mL total surfactant proteins. As an intratracheal suspension, it can be used for the prevention and treatment of neonatal respiratory distress syndrome. Survanta is manufactured by Abbvie.
Calfactant, also known as Infasurf, is an intratracheal suspension derived from the natural surfactant in calf lungs. It is used in premature infants with lung surfactant deficiency that causes infant respiratory distress syndrome (IRDS).

Cow lung is a type of offal used in various cuisines and also as a source for pulmonary surfactants. In Peru it is known as bofe, and in Nigeria as Fùkù. In Indonesia, Paru goreng is a popular type of Padang food, and Nasi kuning can be made with cow lung.

Henry Lewis Halliday was a British-Irish peaditrician and neonatologist. In 2021, Halliday was awarded the James Spence Medal for research into neonatology, for coordinating two of the largest neonatal multicentre trials for prevention and treatment of a number of neonatal respiratory illnesses and for a breakthrough in the development of a new lung surfactant that brought relief to very small babies suffering from infant respiratory distress syndrome (RDS).