Biosynthetic precursor to the tambjamines | |
| Names | |
|---|---|
| IUPAC name 4-Methoxy-2,2'-bipyrrole-5-carboxaldehyde | |
| Other names MBC | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
PubChem CID | |
| |
| |
| Properties | |
| C10H10N2O2 | |
| Molar mass | 190.202 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
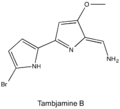
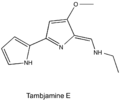
Tambjamines are a group of natural products that are structurally related to the prodiginines. They are enamine derivatives of 4-methoxy-2,2'-bipyrrole-5-carboxaldehyde (MBC). [1]