
Epoxy is the family of basic components or cured end products of epoxy resins. Epoxy resins, also known as polyepoxides, are a class of reactive prepolymers and polymers which contain epoxide groups. The epoxide functional group is also collectively called epoxy. The IUPAC name for an epoxide group is an oxirane.

Tetrahydrofuran (THF), or oxolane, is an organic compound with the formula (CH2)4O. The compound is classified as heterocyclic compound, specifically a cyclic ether. It is a colorless, water-miscible organic liquid with low viscosity. It is mainly used as a precursor to polymers. Being polar and having a wide liquid range, THF is a versatile solvent.
Furfural is an organic compound with the formula C4H3OCHO. It is a colorless liquid, although commercial samples are often brown. It has an aldehyde group attached to the 2-position of furan. It is a product of the dehydration of sugars, as occurs in a variety of agricultural byproducts, including corncobs, oat, wheat bran, and sawdust. The name furfural comes from the Latin word furfur, meaning bran, referring to its usual source. Furfural is only derived from dryed biomass, In addition to ethanol, acetic acid, and sugar, furfural is one of the oldest organic chemicals available readily purified from natural precursors.
Furan is a heterocyclic organic compound, consisting of a five-membered aromatic ring with four carbon atoms and one oxygen atom. Chemical compounds containing such rings are also referred to as furans.

1,4-Butanediol, also called Butane-1,4-diol, is a primary alcohol and an organic compound with the formula HOCH2CH2CH2CH2OH. It is a colorless viscous liquid first synthesized in 1890 via acidic hydrolysis of N,N'-dinitro-1,4-butanediamine by Dutch chemist Pieter Johannes Dekkers, who called it "tetramethylene glycol".

Xylose is a sugar first isolated from wood, and named for it. Xylose is classified as a monosaccharide of the aldopentose type, which means that it contains five carbon atoms and includes an aldehyde functional group. It is derived from hemicellulose, one of the main constituents of biomass. Like most sugars, it can adopt several structures depending on conditions. With its free aldehyde group, it is a reducing sugar.

Furfuryl alcohol is an organic compound containing a furan substituted with a hydroxymethyl group. It is a colorless liquid, but aged samples appear amber. It possesses a faint odor of burning and a bitter taste. It is miscible with but unstable in water. It is soluble in common organic solvents.
Wood glue is an adhesive used to tightly bond pieces of wood together. Many substances have been used as glues. Traditionally animal proteins like casein from milk or collagen from animal hides and bones were boiled down to make early glues. They worked by solidifying as they dried. Later, glues were made from plant starches like flour or potato starch. When combined with water and heated, the starch gelatinizes and forms a sticky paste as it dries. Plant-based glues were common for books and paper products, though they can break down more easily over time compared to animal-based glues. Examples of modern wood glues include polyvinyl acetate (PVA) and epoxy resins. Some resins used in producing composite wood products may contain formaldehyde. As of 2021, “the wood panel industry uses almost 95% of synthetic petroleum-derived thermosetting adhesives, mainly based on urea, phenol, and melamine, among others”.
1,5-Pentanediol is the organic compound with the formula HO(CH2)5OH. Like other diols, this viscous colourless liquid is used as plasticizer and also forms polyesters that are used as emulsifying agents and resin intermediates.
A thermoset polymer matrix is a synthetic polymer reinforcement where polymers act as binder or matrix to secure in place incorporated particulates, fibres or other reinforcements. They were first developed for structural applications, such as glass-reinforced plastic radar domes on aircraft and graphite-epoxy payload bay doors on the Space Shuttle.

Bisphenol A diglycidyl ether is an organic compound and is a liquid epoxy resin. The compound is a colorless viscous liquid. It is a key component of many epoxy resin formulations. Addition of further Bisphenol A and a catalyst and heat can produce Bisphenol A glycidyl ether epoxy resins of higher molecular weight that are solid.

2-Methyltetrahydrofuran (2-MeTHF) is an organic compound with the molecular formula C5H10O. It is a highly flammable, mobile liquid. It is mainly used as a replacement for Tetrahydrofuran (THF) in specialized applications for its better performance, such as to obtain higher reaction temperatures, or easier separations (as, unlike THF, it is not miscible with water). It is derived from sugars via furfural and is occasionally touted as a biofuel.
3,4-Dihydropyran (DHP) is a heterocyclic compound with the formula C5H8O. The six-membered C5O ring has the unsaturation adjacent to oxygen. The isomeric 3,6-dihydropyran has a methylene separating the double bond and oxygen. DHP is used for protecting group for alcohols. It is a colorless liquid.
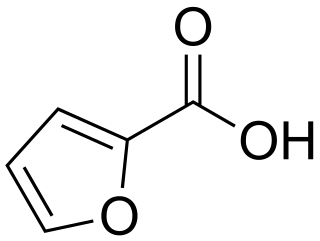
2-Furoic acid is an organic compound, consisting of a furan ring and a carboxylic acid side-group. Along with other furans, its name is derived from the Latin word furfur, meaning bran, from which these compounds were first produced. The salts and esters of furoic acids are known as furoates. 2-Furoic acid is most widely encountered in food products as a preservative and a flavouring agent, where it imparts a sweet, earthy flavour.

5-Hydroxy-2(5H)-furanone is a furanone derived from oxidation of furfural using singlet oxygen. This oxidation is carried out generally in methanol or ethanol with a sensitizer like methylene blue or Rose bengal. The mechanism of this reaction is depicted as below.
n-Butyl glycidyl ether is an industrial chemical used in adhesives, sealants, and as a paint or coating additive. It is principally used to reduce the viscosity of epoxy resin systems.

Furan resin refers to polymers produced from various furan compounds, of which the most common starting materials are furfuryl alcohol and furfural. In the resin and in the cured polyfurfurol, the furan rings are not connected by conjugation. The resins are generally used as binders for sand castings. The furan monomer is typically converted to a free-flowing resin with mild acid catalysis. Curing is achieved using strong acid.
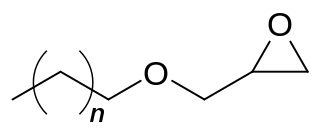
C12-C14 alcohol glycidyl ether (AGE) is an organic chemical in the glycidyl ether family. It is a mixture of mainly 12 and 14 carbon chain alcohols, also called fatty alcohols that have been glycidated. It is an industrial chemical used as a surfactant but primarily for epoxy resin viscosity reduction. It has the CAS number 68609-97-2 but the IUPAC name is more complex as it is a mixture and is 2-(dodecoxymethyl)oxirane;2-(tetradecoxymethyl)oxirane;2-(tridecoxymethyl)oxirane. Other names include dodecyl and tetradecyl glycidyl ethers and alkyl (C12-C14) glycidyl ether.

C12-C13 alcohol glycidyl ether is a mixture of organic chemicals in the glycidyl ether family. It is a mixture of mainly 12 and 13 carbon chain alcohols, also called fatty alcohols that have been glycidated. It is an industrial chemical used as a surfactant but primarily for epoxy resin viscosity reduction. It has the CAS number 120547-52-6.











