The Anatomical Therapeutic Chemical (ATC) Classification System is a drug classification system that classifies the active ingredients of drugs according to the organ or system on which they act and their therapeutic, pharmacological and chemical properties. Its purpose is an aid to monitor drug use and for research to improve quality medication use. It does not imply drug recommendation or efficacy. It is controlled by the World Health Organization Collaborating Centre for Drug Statistics Methodology (WHOCC), and was first published in 1976.

Pharmacology is a science of medical drug and medication, including a substance's origin, composition, pharmacokinetics, therapeutic use, and toxicology. More specifically, it is the study of the interactions that occur between a living organism and chemicals that affect normal or abnormal biochemical function. If substances have medicinal properties, they are considered pharmaceuticals.
A medication is a drug used to diagnose, cure, treat, or prevent disease. Drug therapy (pharmacotherapy) is an important part of the medical field and relies on the science of pharmacology for continual advancement and on pharmacy for appropriate management.

The national drug code (NDC) is a unique product identifier used in the United States for drugs intended for human use. The Drug Listing Act of 1972 requires registered drug establishments to provide the Food and Drug Administration (FDA) with a current list of all drugs manufactured, prepared, propagated, compounded, or processed by it for commercial distribution. Drug products are identified and reported using the NDC.
The pregnancy category of a medication is an assessment of the risk of fetal injury due to the pharmaceutical, if it is used as directed by the mother during pregnancy. It does not include any risks conferred by pharmaceutical agents or their metabolites in breast milk.

Pharmacognosy is the study of crude drugs obtained from medicinal plants, animals, fungi, and other natural sources. The American Society of Pharmacognosy defines pharmacognosy as "the study of the physical, chemical, biochemical, and biological properties of drugs, drug substances, or potential drugs or drug substances of natural origin as well as the search for new drugs from natural sources".
An international nonproprietary name (INN) is an official generic and nonproprietary name given to a pharmaceutical drug or an active ingredient. INNs are intended to make communication more precise by providing a unique standard name for each active ingredient, to avoid prescribing errors. The INN system has been coordinated by the World Health Organization (WHO) since 1953.
A medical classification is used to transform descriptions of medical diagnoses or procedures into standardized statistical code in a process known as clinical coding. Diagnosis classifications list diagnosis codes, which are used to track diseases and other health conditions, inclusive of chronic diseases such as diabetes mellitus and heart disease, and infectious diseases such as norovirus, the flu, and athlete's foot. Procedure classifications list procedure code, which are used to capture interventional data. These diagnosis and procedure codes are used by health care providers, government health programs, private health insurance companies, workers' compensation carriers, software developers, and others for a variety of applications in medicine, public health and medical informatics, including:
Pharmacovigilance, also known as drug safety, is the pharmaceutical science relating to the "collection, detection, assessment, monitoring, and prevention" of adverse effects with pharmaceutical products. The etymological roots for the word "pharmacovigilance" are: pharmakon and vigilare. As such, pharmacovigilance heavily focuses on adverse drug reactions (ADR), which are defined as any response to a drug which is noxious and unintended, including lack of efficacy. Medication errors such as overdose, and misuse and abuse of a drug as well as drug exposure during pregnancy and breastfeeding, are also of interest, even without an adverse event, because they may result in an adverse drug reaction.
Pharmaceutical codes are used in medical classification to uniquely identify medication. They may uniquely identify an active ingredient, drug system in general, or a specific pharmaceutical product from a specific manufacturer.
Pharmacotherapy, also known as pharmacological therapy or drug therapy, is defined as medical treatment that utilizes one or more pharmaceutical drugs to improve ongoing symptoms, treat the underlying condition, or act as a prevention for other diseases (prophylaxis).

Uppsala Monitoring Centre (UMC), located in Uppsala, Sweden, is the field name for the World Health Organization Collaborating Centre for International Drug Monitoring. UMC works by collecting, assessing and communicating information from member countries' national pharmacovigilance centres in regard to the benefits, harm, effectiveness and risks of drugs.

Martindale: The Complete Drug Reference is a reference book published by Pharmaceutical Press listing some 6,000 drugs and medicines used throughout the world, including details of over 125,000 proprietary preparations. It also includes almost 700 disease treatment reviews.
Dosage forms are pharmaceutical drug products in the form in which they are marketed for use, with a specific mixture of active ingredients and inactive components (excipients), in a particular configuration, and apportioned into a particular dose. For example, two products may both be amoxicillin, but one is in 500 mg capsules and another is in 250 mg chewable tablets. The term unit dose can also sometimes encompass non-reusable packaging as well, although the FDA distinguishes that by unit-dose "packaging" or "dispensing". Depending on the context, multi(ple) unit dose can refer to distinct drug products packaged together, or to a single drug product containing multiple drugs and/or doses. The term dosage form can also sometimes refer only to the pharmaceutical formulation of a drug product's constituent drug substance(s) and any blends involved, without considering matters beyond that. Because of the somewhat vague boundaries and unclear overlap of these terms and certain variants and qualifiers within the pharmaceutical industry, caution is often advisable when conversing with someone who may be unfamiliar with another person's use of the term.

A drug is any chemical substance that when consumed causes a change in an organism's physiology, including its psychology, if applicable. Drugs are typically distinguished from food and other substances that provide nutritional support. Consumption of drugs can be via inhalation, injection, smoking, ingestion, absorption via a patch on the skin, suppository, or dissolution under the tongue.
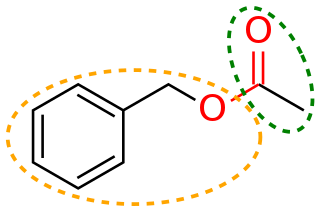
In organic chemistry, a moiety is a part of a molecule that is given a name because it is identified as a part of other molecules as well.
Marketing authorisation is the process of reviewing and assessing the evidence to support a medicinal product, such as a drug, in relation to its marketing, finalised by granting of a licence to be sold.
Drug nomenclature is the systematic naming of drugs, especially pharmaceutical drugs. In the majority of circumstances, drugs have 3 types of names: chemical names, the most important of which is the IUPAC name; generic or nonproprietary names, the most important of which are international nonproprietary names (INNs); and trade names, which are brand names. Under the INN system, generic names for drugs are constructed out of affixes and stems that classify the drugs into useful categories while keeping related names distinguishable. A marketed drug might also have a company code or compound code.
VigiBase is a World Health Organization's (WHO) global Individual Case Safety Report (ICSR) database that contains ICSRs submitted by the participating member states enrolled under WHO's international drug monitoring programme. It is the single largest drug safety data repository in the world. Since 1978, the Uppsala Monitoring Centre on behalf of WHO, have been maintaining VigiBase. Vigibase is used to obtain the information about a safety profile of a medicinal product. These data are used by pharmaceutical industries, academic institutions and regulatory authorities for statistical signal detection, updating periodic reports, ICSR comparisons with company databases and studying the reporting patterns. The data is collected from each of its 110 member states which currently comprises to over 10 million ICSRs. About a hundred thousand ICSRs are added each year.

A botanical drug is defined in the United States Federal Food, Drug, and Cosmetic Act as a botanical product that is marketed as diagnosing, mitigating, treating, or curing a disease; a botanical product in turn, is a finished, labeled product that contains ingredients from plants. Chemicals that are purified from plants, like paclitaxel, and highly purified products of industrial fermentation, like biopharmaceuticals, are not considered to be botanical products.








