
Ants are eusocial insects of the family Formicidae and, along with the related wasps and bees, belong to the order Hymenoptera. Ants evolved from vespoid wasp ancestors in the Cretaceous period. More than 13,800 of an estimated total of 22,000 species have been classified. They are easily identified by their geniculate (elbowed) antennae and the distinctive node-like structure that forms their slender waists.

Pogonomyrmex barbatus is a species of harvester ant from the genus Pogonomyrmex. Its common names include red ant and red harvester ant. These large ants prefer arid chaparral habitats and are native to the Southwestern United States. Nests are made underground in exposed areas. Their diets consist primarily of seeds, and they consequently participate in myrmecochory, an ant-plant interaction through which the ants gain nutrients and the plants benefit through seed dispersal. Red harvester ants are often mistaken for fire ants, but are not closely related to any fire ant species, native or introduced.

Dorylus, also known as driver ants, safari ants, or siafu, is a large genus of army ants found primarily in central and east Africa, although the range also extends to southern Africa and tropical Asia. The term siafu is a loanword from Swahili, and is one of numerous similar words from regional Bantu languages used by indigenous peoples to describe various species of these ants. Unlike the New World members of the former subfamily Ecitoninae, members of this genus form temporary subterranean bivouacs in underground cavities which they excavate and inhabit - either for a few days or up to three months. Also unlike some New World army ants, driver ants are not specialized predators of other species of ant, instead being more generalistic with a diet consisting of a diversity of arthropods. Colonies are enormous compared to other army ants and can contain over 20 million individuals. As with their American counterparts, workers exhibit caste polymorphism with the soldiers having particularly large heads that power their scissor-like mandibles. They are capable of stinging, but very rarely do so, relying instead on their powerful shearing jaws. Driver ant queens are the largest living ants known, with the largest measuring between 40 - 63 millimeters in total body length depending on their physiological condition.

The pharaoh ant is a small (2 mm) yellow or light brown, almost transparent ant notorious for being a major indoor nuisance pest, especially in hospitals. A cryptogenic species, it has now been introduced to virtually every area of the world, including Europe, the Americas, Australasia and Southeast Asia. It is a major pest in the United States, Australia, and Europe. The ant's common name is said to be derived from the mistaken belief that it was one of the biblical Egyptian (pharaonic) plagues.

Bombus terrestris, the buff-tailed bumblebee or large earth bumblebee, is one of the most numerous bumblebee species in Europe. It is one of the main species used in greenhouse pollination, and so can be found in many countries and areas where it is not native, such as Tasmania. Moreover, it is a eusocial insect with an overlap of generations, a division of labour, and cooperative brood care. The queen is monogamous which means she mates with only one male. B. terrestris workers learn flower colours and forage efficiently.

Vespula vulgaris, known as the common wasp, is a species found in regions that include the United Kingdom, Ireland, Germany, India, China, New Zealand and Australia. It is sometimes known in English as the European wasp, but the same name is used for the species Vespula germanica or German wasp. In 2010, the ostensible Vespula vulgaris wasps in North America were found to be a different species, Vespula alascensis.

Eciton burchellii is a species of New World army ant in the genus Eciton. This species performs expansive, organized swarm raids that give it the informal name, Eciton army ant. This species displays a high degree of worker polymorphism. Sterile workers are of four discrete size-castes: minors, medias, porters (sub-majors), and soldiers (majors). Soldiers have much larger heads and specialized mandibles for defense. In lieu of underground excavated nests, colonies of E. burchellii form temporary living nests known as bivouacs, which are composed of hanging live worker bodies and which can be disassembled and relocated during colony emigrations. Eciton burchellii colonies cycle between stationary phases and nomadic phases when the colony emigrates nightly. These alternating phases of emigration frequency are governed by coinciding brood developmental stages. Group foraging efforts known as "raids" are maintained by the use of pheromones, can be 200 metres (660 ft) long, and employ up to 200,000 ants. Workers are also adept at making living structures out of their own bodies to improve efficiency of moving as a group across the forest floor while foraging or emigrating. Workers can fill "potholes" in the foraging trail with their own bodies, and can also form living bridges. Numerous antbirds prey on the Eciton burchellii by using their raids as a source of food. In terms of geographical distribution, this species is found in the Amazon jungle and Central America.

Nothomyrmecia, also known as the dinosaur ant or dawn ant, is an extremely rare genus of ants consisting of a single species, Nothomyrmecia macrops. These ants live in South Australia, nesting in old-growth mallee woodland and Eucalyptus woodland. The full distribution of Nothomyrmecia has never been assessed, and it is unknown how widespread the species truly is; its potential range may be wider if it does favour old-growth mallee woodland. Possible threats to its survival include habitat destruction and climate change. Nothomyrmecia is most active when it is cold because workers encounter fewer competitors and predators such as Camponotus and Iridomyrmex, and it also increases hunting success. Thus, the increase of temperature may prevent them from foraging and very few areas would be suitable for the ant to live in. As a result, the IUCN lists the ant as Critically Endangered.

Eciton is a New World army ant genus that contains the most familiar species of army ants. The most predominant and well-known species is Eciton burchellii, which is also more commonly known as the army ant and is considered the type species.

Nuptial flight is an important phase in the reproduction of most ant, termite, and some bee species. It is also observed in some fly species, such as Rhamphomyia longicauda.
Myrmecia inquilina is a species of ant endemic to Australia in the subfamily Myrmeciinae, first discovered in 1955 and described by Athol Douglas and William Brown Jr. in 1959. These ants are large, measuring 21.4 millimetres (0.84 in). During the time of its discovery, Douglas and Brown announced M. inquilina as the first social parasite among the primitive subfamilies, and today it is one of the two known Myrmecia species to have no worker caste. Two host species are known, Myrmecia nigriceps and Myrmecia vindex. Aggression between M. inquilina and its host species does not occur, and colonies may only produce M. inquilina brood months after the inquiline queens begin to lay their eggs. Queens eat the colony brood or trophic eggs, and other Myrmecia species may kill M. inquilina queens if they reject them. Due to its restricted distribution and threats to its habitat, the ant is "vulnerable" according to the IUCN Red List.

Eusociality is the highest level of organization of sociality. It is defined by the following characteristics: cooperative brood care, overlapping generations within a colony of adults, and a division of labor into reproductive and non-reproductive groups. The division of labor creates specialized behavioral groups within an animal society which are sometimes referred to as 'castes'. Eusociality is distinguished from all other social systems because individuals of at least one caste usually lose the ability to perform behaviors characteristic of individuals in another caste. Eusocial colonies can be viewed as superorganisms.

The eastern yellow jacket or eastern yellowjacket is a wasp found in eastern North America. Although most of their nests are subterranean, they are often considered a pest due to their nesting in recreational areas and buildings. This yellow jacket is a social insect, living in colonies of hundreds to thousands of individuals. Along with their subfamily, Vespinae, this species demonstrates supportive parental care for offspring, separation of reproductive and sterile castes, and overlapping generations. They aggressively defend their hives from threats and are known to inflict painful stings.

The green-head ant is a species of ant that is endemic to Australia. It was described by British entomologist Frederick Smith in 1858 as a member of the genus Rhytidoponera in the subfamily Ectatomminae. These ants measure between 5 and 7 mm. The queens and workers look similar, differing only in size, with the males being the smallest. They are well known for their distinctive metallic appearance, which varies from green to purple or even reddish-violet. Among the most widespread of all insects in Australia, green-head ants are found in almost every Australian state, but are absent in Tasmania. They have also been introduced in New Zealand, where several populations have been established.
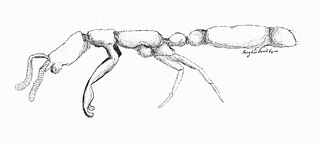
Leptanilla japonica is an uncommon highly migratory, subterranean ant found in Japan. They are tiny insects, with workers measuring about 1.2 mm and queens reaching to about 1.8 mm, and live in very small colonies of only a few hundred individuals at a time Its sexual development follows a seasonal cycle that affects the colony's migration and feeding habits, and vice versa. L. japonica exhibits specialized predation, with prey consisting mainly of geophilomorph centipedes, a less reliable food source that also contributes to their high rate of nest migration. Like ants of genera Amblyopone and Proceratium, the genus Leptanilla engages in larval hemolymph feeding (LHF), with the queen using no other form of sustenance. LHF is an advantageous alternative to the more costly cannibalism. Unlike any other ant, however, members of Leptanilla, including L. japonica, have evolved a specialized organ dubbed the “larval hemolymph tap” that reduces the damage LHF inflicts on the larvae. LHF has become this species' main form of nutrition.

Polistes exclamans, the Guinea paper wasp, is a social wasp and is part of the family Vespidae of the order Hymenoptera. It is found throughout the United States, Mexico, the Bahamas, Jamaica and parts of Canada. Due to solitary nest founding by queens, P. exclamans has extended its range in the past few decades and now covers the eastern half of the United States, as well as part of the north. This expansion is typically attributed to changing global climate and temperatures. P. exclamans has three specific castes, including males, workers, and queens, but the dominance hierarchy is further distinguished by age. The older the wasp is, the higher it is in ranking within the colony. In most P. exclamans nests, there is one queen who lays all the eggs in the colony. The physiological similarities between the worker and queen castes have led to experiments attempting to distinguish the characteristics of these two castes and how they are determined, though males have easily identifiable physiological characteristics. Since P. exclamans live in relatively small, open combed nests, they are often subject to predators and parasites, such as Chalcoela iphitalis, Elasmus polistis, and birds. P. exclamans have defense and recognition strategies that help protect against these predators and parasites.

Ooceraea biroi, the clonal raider ant, is a queenless clonal ant in the genus Ooceraea. Native to the Asian mainland, this species has become invasive on tropical and subtropical islands throughout the world. Unlike most ants, which have reproductive queens and mostly nonreproductive workers, all individuals in a O. biroi colony reproduce clonally via thelytokous parthenogenesis. Like most dorylines, O. biroi are obligate myrmecophages and raid nests of other ant species to feed on the brood.

Novomessor cockerelli is a species of ant in the subfamily Myrmicinae. It is native to the deserts of the Southwestern United States and Mexico. It lives in large underground colonies in which there is a single queen. The worker ants leave the nest daily to forage for seeds, plant material and dead insects.
This is a glossary of terms used in the descriptions of ants.

Nymphister kronaueri is a species of histerid beetle native to Costa Rica. It was first discovered in 2014. The discovery of the beetle received much media attention due to its unusual habit of hitchhiking on army ants. It was formally described in 2017.
























