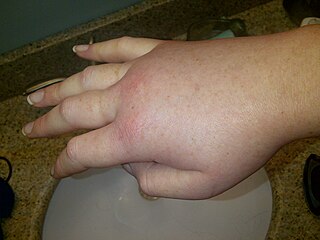
Hereditary angioedema (HAE) is a disorder that results in recurrent attacks of severe swelling. The swelling most commonly affects the arms, legs, face, intestinal tract, and airway. If the intestinal tract is affected, abdominal pain and vomiting may occur. Swelling of the airway can result in its obstruction and trouble breathing. Without preventive treatment, attacks typically occur every two weeks and last for a few days.

Angioedema is an area of swelling (edema) of the lower layer of skin and tissue just under the skin or mucous membranes. The swelling may occur in the face, tongue, larynx, abdomen, or arms and legs. Often it is associated with hives, which are swelling within the upper skin. Onset is typically over minutes to hours.

Icatibant, sold under the brand name Firazyr, is a medication for the symptomatic treatment of acute attacks of hereditary angioedema (HAE) in adults with C1-esterase-inhibitor deficiency. It is not effective in angioedema caused by medication from the ACE inhibitor class.
Ecallantide is a drug used for the treatment of hereditary angioedema (HAE) and in the prevention of blood loss in cardiothoracic surgery. It is an inhibitor of the protein kallikrein and a 60-amino acid polypeptide which was developed from a Kunitz domain through phage display to mimic antibodies inhibiting kallikrein.
BioCryst Pharmaceuticals, Inc. is an American pharmaceutical company headquartered in Durham, North Carolina. The company is a late stage biotech company that focuses on oral drugs for rare and serious diseases. BioCryst's antiviral drug peramivir (Rapivab) was approved by FDA in December 2014. It has also been approved in Japan, Korea, and China.

Lasmiditan, sold under the brand name Reyvow, is a medication used for the acute treatment of migraine with or without aura in adults. It is not useful for prevention. It is taken by mouth.
Ixekizumab, sold under the brand name Taltz, is an injectable medication for the treatment of autoimmune diseases. Chemically, it is a form of a humanized monoclonal antibody. The substance acts by binding interleukin 17A and neutralizing it, reducing inflammation.

Cabotegravir, sold under the brand name Vocabria among others, is a antiretroviral medication used for the treatment of HIV/AIDS. It is available in the form of tablets and as an intramuscular injection, as well as in an injectable combination with rilpivirine under the brand name Cabenuva.

Semaglutide is an antidiabetic medication used for the treatment of type 2 diabetes and an anti-obesity medication used for long-term weight management. It is a peptide similar to the hormone glucagon-like peptide-1 (GLP-1), modified with a side chain. It can be administered by subcutaneous injection or taken orally. It is sold under the brand names Ozempic and Rybelsus for diabetes, and under the brand name Wegovy for weight loss.
Inebilizumab, sold under the brand name Uplizna, is a medication for the treatment of neuromyelitis optica spectrum disorder (NMOSD) in adults. Inebilizumab is a humanized mAb that binds to and depletes CD19+ B cells including plasmablasts and plasma cells.

Galcanezumab, sold under the brand name Emgality, is a humanized monoclonal antibody used for the prevention of migraine. It is also used for cluster headaches.
Lanadelumab, sold under the brand name Takhzyro, is a human monoclonal antibody that targets plasma kallikrein (pKal) in order to promote prevention of angioedema in people with hereditary angioedema. Lanadelumab, was approved in the United States as the first monoclonal antibody indicated for prophylactic treatment to prevent hereditary angioedema attacks. Lanadelumab is the first treatment for hereditary angioedema prevention made by using cells within a lab, not human plasma.
Crizanlizumab, sold under the brand name Adakveo among others, is a monoclonal antibody medication that binds to P-selectin. It is a medication used to reduce the frequency of vaso-occlusive crisis in people aged 16 years and older who have sickle cell anemia. It is given by injection into a vein.
Satralizumab, sold under the brand name Enspryng, is a humanized monoclonal antibody medication that is used for the treatment of neuromyelitis optica spectrum disorder (NMOSD), a rare autoimmune disease. The drug is being developed by Chugai Pharmaceutical, a subsidiary of Roche.
Bempedoic acid, sold under the brand name Nexletol among others, is a medication for the treatment of hypercholesterolemia.
Inotersen, sold under the brand name Tegsedi, is a 2'-O-(2-methoxyethyl) (2'-MOE) antisense oligonucleotide medication used for the treatment of nerve damage in adults with hereditary transthyretin-mediated amyloidosis. The sequence is TCTTG GTTACATGAA ATCCC, where C is methylated C, and the first and third section are MOE-modified.

Ripretinib, sold under the brand name Qinlock, is a medication for the treatment of adults with advanced gastrointestinal stromal tumor (GIST), a type of tumor that originates in the gastrointestinal tract. It is taken by mouth. Ripretinib inhibits the activity of the kinases KIT and PDGFRA, which helps keep cancer cells from growing.
Somapacitan, sold under the brand name Sogroya, is a growth hormone medication. Somapacitan is a human growth hormone analog. Somapacitan-beco is produced in Escherichia coli by recombinant DNA technology.

Avacopan, sold under the brand name Tavneos, is a medication used to treat anti-neutrophil cytoplasmic autoantibody-associated vasculitis. Avacopan is a complement 5a receptor antagonist and a cytochrome P450 3A4 inhibitor.

Nirmatrelvir/ritonavir, sold under the brand name Paxlovid, is a co-packaged medication used as a treatment for COVID‑19. It contains the antiviral medications nirmatrelvir and ritonavir and was developed by Pfizer. Nirmatrelvir inhibits SARS-CoV-2 main protease, while ritonavir is a strong CYP3A inhibitor, slowing down nirmatrelvir metabolism and therefore boosting its effect. It is taken by mouth.