
Monoamine neurotransmitters are neurotransmitters and neuromodulators that contain one amino group connected to an aromatic ring by a two-carbon chain (such as -CH2-CH2-). Examples are dopamine, norepinephrine and serotonin.

Phenethylamine (PEA) is an organic compound, natural monoamine alkaloid, and trace amine, which acts as a central nervous system stimulant in humans. In the brain, phenethylamine regulates monoamine neurotransmission by binding to trace amine-associated receptor 1 (TAAR1) and inhibiting vesicular monoamine transporter 2 (VMAT2) in monoamine neurons. To a lesser extent, it also acts as a neurotransmitter in the human central nervous system. In mammals, phenethylamine is produced from the amino acid L-phenylalanine by the enzyme aromatic L-amino acid decarboxylase via enzymatic decarboxylation. In addition to its presence in mammals, phenethylamine is found in many other organisms and foods, such as chocolate, especially after microbial fermentation.

Tryptamine is an indolamine metabolite of the essential amino acid, tryptophan. The chemical structure is defined by an indole—a fused benzene and pyrrole ring, and a 2-aminoethyl group at the second carbon (third aromatic atom, with the first one being the heterocyclic nitrogen). The structure of tryptamine is a shared feature of certain aminergic neuromodulators including melatonin, serotonin, bufotenin and psychedelic derivatives such as dimethyltryptamine (DMT), psilocybin, psilocin and others. Tryptamine has been shown to activate trace amine-associated receptors expressed in the mammalian brain, and regulates the activity of dopaminergic, serotonergic and glutamatergic systems. In the human gut, symbiotic bacteria convert dietary tryptophan to tryptamine, which activates 5-HT4 receptors and regulates gastrointestinal motility. Multiple tryptamine-derived drugs have been developed to treat migraines, while trace amine-associated receptors are being explored as a potential treatment target for neuropsychiatric disorders.
A biogenic amine is a biogenic substance with one or more amine groups. They are basic nitrogenous compounds formed mainly by decarboxylation of amino acids or by amination and transamination of aldehydes and ketones. Biogenic amines are organic bases with low molecular weight and are synthesized by microbial, vegetable and animal metabolisms. In food and beverages they are formed by the enzymes of raw material or are generated by microbial decarboxylation of amino acids.
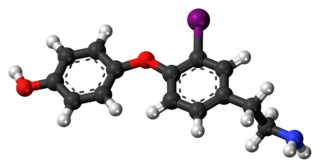
3-Iodothyronamine (T1AM) is an endogenous thyronamine. T1AM is a high-affinity ligand for the trace amine-associated receptor TAAR1 (TAR1, TA1), a recently discovered G protein-coupled receptor. T1AM is the most potent endogenous TAAR1 agonist yet discovered. Activation of TAAR1 by T1AM results in the production of large amounts of cAMP. This effect is coupled with decreased body temperature and cardiac output. Wu et al. have pointed out that this relationship is not typical of the endocrine system, indicating that TAAR1 activity may not be coupled to G-proteins in some tissues, or that T1AM may interact with other receptor subtypes.

Trace amines are an endogenous group of trace amine-associated receptor 1 (TAAR1) agonists – and hence, monoaminergic neuromodulators – that are structurally and metabolically related to classical monoamine neurotransmitters. Compared to the classical monoamines, they are present in trace concentrations. They are distributed heterogeneously throughout the mammalian brain and peripheral nervous tissues and exhibit high rates of metabolism. Although they can be synthesized within parent monoamine neurotransmitter systems, there is evidence that suggests that some of them may comprise their own independent neurotransmitter systems.
Trace amine-associated receptors (TAARs), sometimes referred to as trace amine receptors, are a class of G protein-coupled receptors that were discovered in 2001. TAAR1, the first of six functional human TAARs, has gained considerable interest in academic and proprietary pharmaceutical research due to its role as the endogenous receptor for the trace amines phenylethylamine, tyramine, and tryptamine – metabolic derivatives of the amino acids phenylalanine, tyrosine and tryptophan, respectively – ephedrine, as well as the synthetic psychostimulants, amphetamine, methamphetamine and methylenedioxymethamphetamine. In 2004, it was shown that mammalian TAAR1 is also a receptor for thyronamines, decarboxylated and deiodinated relatives of thyroid hormones. TAAR2–TAAR9 function as olfactory receptors for volatile amine odorants in vertebrates.
Cutamesine (SA 4503) is a synthetic sigma receptor agonist which is selective for the σ1 receptor, a chaperone protein mainly found in the endoplasmic reticulum of cells in the central nervous system. These σ1 receptors play a key role in the modulation of Ca2+ release and apoptosis. Cutamesine's activation of the σ1 receptor is tied to a variety of physiological phenomena in the CNS, including activation of dopamine-releasing neurons and repression of the MAPK/ERK pathway.

N-Methylphenethylamine (NMPEA) is a naturally occurring trace amine neuromodulator in humans that is derived from the trace amine, phenethylamine (PEA). It has been detected in human urine and is produced by phenylethanolamine N-methyltransferase with phenethylamine as a substrate, which significantly increases PEA's effects. PEA breaks down into phenylacetaldehyde which is further broken down into phenylacetic acid by monoamine oxidase. When this is inhibited by monoamine oxidase inhibitors, it allows more of the PEA to be metabolized into nymphetamine (NMPEA) and not wasted on the weaker inactive metabolites.

The human muscarinic acetylcholine receptor M5, encoded by the CHRM5 gene, is a member of the G protein-coupled receptor superfamily of integral membrane proteins. It is coupled to Gq protein. Binding of the endogenous ligand acetylcholine to the M5 receptor triggers a number of cellular responses such as adenylate cyclase inhibition, phosphoinositide degradation, and potassium channel modulation. Muscarinic receptors mediate many of the effects of acetylcholine in the central and peripheral nervous system. The clinical implications of this receptor have not been fully explored; however, stimulation of this receptor is known to effectively decrease cyclic AMP levels and downregulate the activity of protein kinase A (PKA).
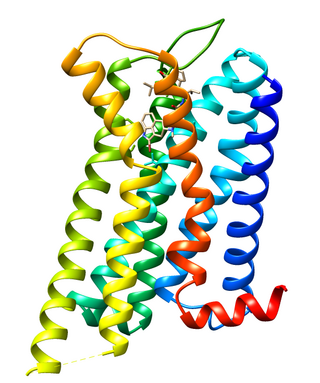
Dopamine receptor D2, also known as D2R, is a protein that, in humans, is encoded by the DRD2 gene. After work from Paul Greengard's lab had suggested that dopamine receptors were the site of action of antipsychotic drugs, several groups, including those of Solomon Snyder and Philip Seeman used a radiolabeled antipsychotic drug to identify what is now known as the dopamine D2 receptor. The dopamine D2 receptor is the main receptor for most antipsychotic drugs. The structure of DRD2 in complex with the atypical antipsychotic risperidone has been determined.

Dopamine receptor D1, also known as DRD1. It is one of the two types of D1-like receptor family — receptors D1 and D5. It is a protein that in humans is encoded by the DRD1 gene.

Trace amine-associated receptor 2 (TAAR2), formerly known as G protein-coupled receptor 58 (GPR58), is a protein that in humans is encoded by the TAAR2 gene. TAAR2 is coexpressed with Gα proteins; however, as of February 2017, its signal transduction mechanisms have not been determined.

Trace amine-associated receptor 5 is a protein that in humans is encoded by the TAAR5 gene. In vertebrates, TAAR5 is expressed in the olfactory epithelium.

Trace amine-associated receptor 1 (TAAR1) is a trace amine-associated receptor (TAAR) protein that in humans is encoded by the TAAR1 gene. TAAR1 is an intracellular amine-activated Gs-coupled and Gq-coupled G protein-coupled receptor (GPCR) that is primarily expressed in several peripheral organs and cells, astrocytes, and in the intracellular milieu within the presynaptic plasma membrane of monoamine neurons in the central nervous system (CNS). TAAR1 was discovered in 2001 by two independent groups of investigators, Borowski et al. and Bunzow et al. TAAR1 is one of six functional human trace amine-associated receptors, which are so named for their ability to bind endogenous amines that occur in tissues at trace concentrations. TAAR1 plays a significant role in regulating neurotransmission in dopamine, norepinephrine, and serotonin neurons in the CNS; it also affects immune system and neuroimmune system function through different mechanisms.

Alazocine, also known more commonly as N-allylnormetazocine (NANM), is a synthetic opioid analgesic of the benzomorphan family related to metazocine, which was never marketed. In addition to its opioid activity, the drug is a sigma receptor agonist, and has been used widely in scientific research in studies of this receptor. Alazocine is described as a potent analgesic, psychotomimetic or hallucinogen, and opioid antagonist. Moreover, one of its enantiomers was the first compound that was found to selectively label the σ1 receptor, and led to the discovery and characterization of the receptor.

Phenylethanolamine, or β-hydroxyphenethylamine, is a trace amine with a structure similar to those of other trace phenethylamines as well as the catecholamine neurotransmitters dopamine, norepinephrine, and epinephrine. As an organic compound, phenylethanolamine is a β-hydroxylated phenethylamine that is also structurally related to a number of synthetic drugs in the substituted phenethylamine class. In common with these compounds, phenylethanolamine has strong cardiovascular activity and, under the name Apophedrin, has been used as a drug to produce topical vasoconstriction.

A monoamine releasing agent (MRA), or simply monoamine releaser, is a drug that induces the release of a monoamine neurotransmitter from the presynaptic neuron into the synapse, leading to an increase in the extracellular concentrations of the neurotransmitter. Many drugs induce their effects in the body and/or brain via the release of monoamine neurotransmitters, e.g., trace amines, many substituted amphetamines, and related compounds.

RO-5166017 is a drug developed by Hoffmann-La Roche which acts as a potent and selective agonist for the trace amine-associated receptor 1, with no significant activity at other targets. This is important for the study of the TAAR1 receptor, as while numerous other compounds are known which act as TAAR1 agonists, such as methamphetamine, MDMA and 3-iodothyronamine, all previously known TAAR1 agonists are either weak and rapidly metabolized, or have strong pharmacological activity at other targets, making it very difficult to assess which effects are due to TAAR1 activation. The discovery of RO-5166017 allows purely TAAR1 mediated effects to be studied, and in animal studies it was shown to prevent stress-induced hyperthermia and block dopamine-dependent hyperlocomotion, as well as blocking the hyperactivity which would normally be induced by an NMDA antagonist. The experiment was done in dopamine transporter knockout mice, and since TAAR1 affects the dopamine transporter, the results could be very different in humans.

o-Phenyl-3-iodotyramine (o-PIT) is a drug which acts as a selective agonist for the trace amine-associated receptor 1. It has reasonable selectivity for TAAR1 but relatively low potency, and is rapidly metabolised in vivo, making it less useful for research than newer ligands such as RO5166017.
















