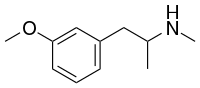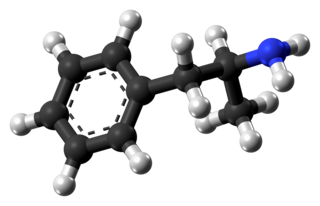
Amphetamine is a strong central nervous system (CNS) stimulant that is used in the treatment of attention deficit hyperactivity disorder (ADHD), narcolepsy, and obesity. It is also commonly used as a recreational drug. Amphetamine was discovered in 1887 and exists as two enantiomers: levoamphetamine and dextroamphetamine. Amphetamine properly refers to a specific chemical, the racemic free base, which is equal parts of the two enantiomers in their pure amine forms. The term is frequently used informally to refer to any combination of the enantiomers, or to either of them alone. Historically, it has been used to treat nasal congestion and depression. Amphetamine is also used as an athletic performance enhancer and cognitive enhancer, and recreationally as an aphrodisiac and euphoriant. It is a prescription drug in many countries, and unauthorized possession and distribution of amphetamine are often tightly controlled due to the significant health risks associated with recreational use.

Phenethylamine (PEA) is an organic compound, natural monoamine alkaloid, and trace amine, which acts as a central nervous system stimulant in humans. In the brain, phenethylamine regulates monoamine neurotransmission by binding to trace amine-associated receptor 1 (TAAR1) and inhibiting vesicular monoamine transporter 2 (VMAT2) in monoamine neurons. To a lesser extent, it also acts as a neurotransmitter in the human central nervous system. In mammals, phenethylamine is produced from the amino acid L-phenylalanine by the enzyme aromatic L-amino acid decarboxylase via enzymatic decarboxylation. In addition to its presence in mammals, phenethylamine is found in many other organisms and foods, such as chocolate, especially after microbial fermentation.

Dextroamphetamine is a central nervous system (CNS) stimulant and an amphetamine enantiomer that is prescribed for the treatment of attention deficit hyperactivity disorder (ADHD) and narcolepsy. It is also used as an athletic performance and cognitive enhancer, and recreationally as an aphrodisiac and euphoriant.

Adderall and Mydayis are trade names for a combination drug called mixed amphetamine salts containing four salts of amphetamine. The mixture is composed of equal parts racemic amphetamine and dextroamphetamine, which produces a (3:1) ratio between dextroamphetamine and levoamphetamine, the two enantiomers of amphetamine. Both enantiomers are stimulants, but differ enough to give Adderall an effects profile distinct from those of racemic amphetamine or dextroamphetamine, which are marketed as Evekeo and Dexedrine/Zenzedi, respectively. Adderall is used in the treatment of attention deficit hyperactivity disorder (ADHD) and narcolepsy. It is also used illicitly as an athletic performance enhancer, cognitive enhancer, appetite suppressant, and recreationally as a euphoriant. It is a central nervous system (CNS) stimulant of the phenethylamine class.

Dimethoxybromoamphetamine (DOB), also known as brolamfetamine (INN) and bromo-DMA, is a psychedelic drug and substituted amphetamine of the phenethylamine class of compounds. DOB was first synthesized by Alexander Shulgin in 1967. Its synthesis and effects are documented in Shulgin's book PiHKAL: A Chemical Love Story.

3,4-Methylenedioxyamphetamine is an empathogen-entactogen, psychostimulant, and psychedelic drug of the amphetamine family that is encountered mainly as a recreational drug. In terms of pharmacology, MDA acts most importantly as a serotonin–norepinephrine–dopamine releasing agent (SNDRA). In most countries, the drug is a controlled substance and its possession and sale are illegal.

2,5-Dimethoxy-4-ethylamphetamine is a psychedelic drug of the phenethylamine and amphetamine chemical classes. It was first synthesized by Alexander Shulgin, and was described in his book PiHKAL.

The solute carrier family 18 member 2 (SLC18A2) also known as vesicular monoamine transporter 2 (VMAT2) is a protein that in humans is encoded by the SLC18A2 gene. SLC18A2 is an integral membrane protein that transports monoamines—particularly neurotransmitters such as dopamine, norepinephrine, serotonin, and histamine—from cellular cytosol into synaptic vesicles. In nigrostriatal pathway and mesolimbic pathway dopamine-releasing neurons, SLC18A2 function is also necessary for the vesicular release of the neurotransmitter GABA.
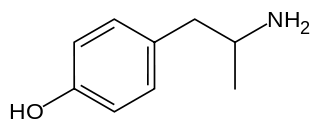
4-Hydroxyamphetamine (4HA), also known as hydroxyamfetamine, hydroxyamphetamine, oxamphetamine, norpholedrine, para-hydroxyamphetamine, and α-methyltyramine, is a drug that stimulates the sympathetic nervous system.

Trace amines are an endogenous group of trace amine-associated receptor 1 (TAAR1) agonists – and hence, monoaminergic neuromodulators – that are structurally and metabolically related to classical monoamine neurotransmitters. Compared to the classical monoamines, they are present in trace concentrations. They are distributed heterogeneously throughout the mammalian brain and peripheral nervous tissues and exhibit high rates of metabolism. Although they can be synthesized within parent monoamine neurotransmitter systems, there is evidence that suggests that some of them may comprise their own independent neurotransmitter systems.

Cocaine- and amphetamine-regulated transcript, also known as CART, is a neuropeptide protein that in humans is encoded by the CARTPT gene. CART appears to have roles in reward, feeding, and stress, and it has the functional properties of an endogenous psychostimulant.

Methamphetamine is a potent central nervous system (CNS) stimulant that is mainly used as a recreational drug and less commonly as a second-line treatment for attention deficit hyperactivity disorder and obesity. Methamphetamine was discovered in 1893 and exists as two enantiomers: levo-methamphetamine and dextro-methamphetamine. Methamphetamine properly refers to a specific chemical substance, the racemic free base, which is an equal mixture of levomethamphetamine and dextromethamphetamine in their pure amine forms. It is rarely prescribed over concerns involving human neurotoxicity and potential for recreational use as an aphrodisiac and euphoriant, among other concerns, as well as the availability of safer substitute drugs with comparable treatment efficacy such as Adderall and Vyvanse. Dextromethamphetamine is a stronger CNS stimulant than levomethamphetamine.

Norfenfluramine, or 3-trifluoromethylamphetamine, is a never-marketed drug of the amphetamine family that behaves as a serotonin and norepinephrine releasing agent and potent 5-HT2A, 5-HT2B, and 5-HT2C agonist. The action of norfenfluramine on 5-HT2B receptors on heart valves leads to a characteristic pattern of heart failure following proliferation of cardiac fibroblasts on the tricuspid valve, known as cardiac fibrosis. This side effect led to the withdrawal of fenfluramine as an anorectic agent worldwide, and to the withdrawal of benfluorex in Europe, as both fenfluramine and benfluorex form norfenfluramine as an active metabolite. It is a human TAAR1 agonist.

Trace amine-associated receptor 1 (TAAR1) is a trace amine-associated receptor (TAAR) protein that in humans is encoded by the TAAR1 gene. TAAR1 is an intracellular amine-activated Gs-coupled and Gq-coupled G protein-coupled receptor (GPCR) that is primarily expressed in several peripheral organs and cells, astrocytes, and in the intracellular milieu within the presynaptic plasma membrane of monoamine neurons in the central nervous system (CNS). TAAR1 was discovered in 2001 by two independent groups of investigators, Borowski et al. and Bunzow et al. TAAR1 is one of six functional human trace amine-associated receptors, which are so named for their ability to bind endogenous amines that occur in tissues at trace concentrations. TAAR1 plays a significant role in regulating neurotransmission in dopamine, norepinephrine, and serotonin neurons in the CNS; it also affects immune system and neuroimmune system function through different mechanisms.
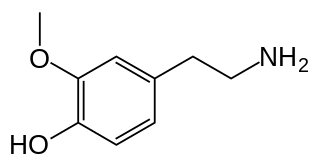
3-Methoxytyramine (3-MT), also known as 3-methoxy-4-hydroxyphenethylamine, is a human trace amine that occurs as a metabolite of the neurotransmitter dopamine. It is formed by the introduction of a methyl group to dopamine by the enzyme catechol-O-methyl transferase (COMT). 3-MT can be further metabolized by the enzyme monoamine oxidase (MAO) to form homovanillic acid (HVA), which is then typically excreted in the urine.

RTI-126 is a phenyltropane derivative which acts as a potent monoamine reuptake inhibitor and stimulant drug, and has been sold as a designer drug. It is around 5 times more potent than cocaine at inhibiting monoamine reuptake in vitro, but is relatively unselective. It binds to all three monoamine transporters, although still with some selectivity for the dopamine transporter. RTI-126 has a fast onset of effects and short duration of action, and its pharmacological profile in animals is among the closest to cocaine itself out of all the drugs in the RTI series. Its main application in scientific research has been in studies investigating the influence of pharmacokinetics on the abuse potential of stimulant drugs, with its rapid entry into the brain thought to be a key factor in producing its high propensity for development of dependence in animals.

3-Methoxy-4-methylamphetamine (MMA) is an entactogen and psychedelic drug of the phenethylamine and amphetamine classes. It was first synthesized in 1970 and was encountered as a street drug in Italy in the same decade. MMA was largely forgotten until being reassayed by David E. Nichols as a non-neurotoxic MDMA analogue in 1991, and has subsequently been sold as a designer drug on the internet since the late 2000s (decade).

meta-Methoxyamphetamine (MMA), also known as 3-methoxyamphetamine (3-MA), is a stimulant drug from the amphetamine family. It has similar effects in animal drug discrimination tests to the more widely known derivative 4-methoxyamphetamine (PMA), although with a slightly different ratio of monoamine release, being a combined serotonin, dopamine, and norepinephrine releasing agent rather than a fairly selective serotonin releaser like PMA. 3-Methoxyamphetamine has similarly appeared on the illicit market as a designer drug alternative to MDMA, although far more rarely than its infamous positional isomer. It produces gepefrine, a cardiac stimulant, as one of its major metabolites.

JWH-007 is an analgesic chemical from the naphthoylindole family, which acts as a cannabinoid agonist at both the CB1 and CB2 receptors. It was first reported in 1994 by a group including the noted cannabinoid chemist John W. Huffman. It was the most active of the first group of N-alkyl naphoylindoles discovered by the team led by John W Huffman, several years after the family was initially described with the discovery of the N-morpholinylethyl compounds pravadoline (WIN 48,098), JWH-200 (WIN 55,225) and WIN 55,212-2 by the Sterling Winthrop group. Several other N-alkyl substituents were found to be active by Huffman's team including the n-butyl, n-hexyl, 2-heptyl, and cyclohexylethyl groups, but it was subsequently determined that the 2-methyl group on the indole ring is not required for CB1 binding, and tends to increase affinity for CB2 instead. Consequently, the 2-desmethyl derivative of JWH-007, JWH-018, has slightly higher binding affinity for CB1, with an optimum binding of 9.00 nM at CB1 and 2.94 nM at CB2, and JWH-007 displayed optimum binding of 9.50 nM at CB1 and 2.94 nM at CB2.