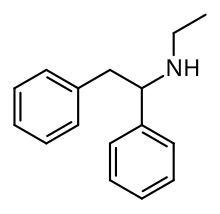
Lefetamine (Santenol) is a drug which is a stimulant and also an analgesic with effects comparable to codeine.

Arylcyclohexylamines, also known as arylcyclohexamines or arylcyclohexanamines, are a chemical class of pharmaceutical, designer, and experimental drugs.

3',4'-Methylenedioxy-α-pyrrolidinobutyrophenone (MDPBP) is a stimulant of the cathinone class developed in the 1960s, which has been reported as a novel designer drug. MDPBP is sometimes sold under the name "NRG-1" as a mixture with other cathinone derivatives, including flephedrone, pentylone, MαPPP and its higher homologue MDPV. As with other cathinones, MDPBP has been shown to have reinforcing effects in rats.

4-Methoxyphencyclidine is a dissociative anesthetic drug that has been sold online as a research chemical. The synthesis of 4-MeO-PCP was first reported in 1965 by the Parke-Davis medicinal chemist Victor Maddox. A 1999 review published by a chemist using the pseudonym John Q. Beagle suggested the potency of 4-MeO-PCP in man was reduced relative to PCP, two years later Beagle published a detailed description of the synthesis and qualitative effects of 4-MeO-PCP, which he said possessed 70% the potency of PCP. 4-MeO-PCP was the first arylcyclohexylamine research chemical to be sold online, it was introduced in late 2008 by a company trading under the name CBAY and was followed by several related compounds such as 3-MeO-PCP and methoxetamine. 4-MeO-PCP has lower affinity for the NMDA receptor than PCP, but higher affinity than ketamine, it is orally active in a dosage range similar to ketamine, with some users requiring doses in excess of 100 mg for desired effects. Users have reported substantial differences in active dose, these discrepancies can be partially explained by the presence of unreacted PCC and other impurities in samples sold on the grey market. 4-MeO-PCP has Ki values of 404 nM for the NMDA receptor, 713 nM for the norepinephrine transporter, 844 nM for the serotonin transporter, 296 nM for the σ1 receptor and 143 nM for the σ2 receptor.

MT-45 (IC-6) is an opioid analgesic drug invented in the 1970s by Dainippon Pharmaceutical Co. It is chemically a 1-substituted-4-(1,2-diphenylethyl) piperazine derivative, which is structurally unrelated to most other opioid drugs. Racemic MT-45 has around 80% the potency of morphine, with almost all opioid activity residing in the (S) enantiomer. It has been used as a lead compound from which a large family of potent opioid drugs have been developed, including full agonists, partial agonists, and antagonists at the three main opioid receptor subtypes. Fluorinated derivatives of MT-45 such as 2F-MT-45 are significantly more potent as μ-opioid receptor agonists, and one of its main metabolites 1,2-diphenylethylpiperazine also blocks NMDA receptors.

AD-1211 is an opioid analgesic drug invented in the 1970s by Dainippon Pharmaceutical Co. It is chemically a 1-substituted-4-prenyl-piperazine derivative, which is structurally unrelated to most other opioid drugs. The (S)-enantiomers in this series are more active as opioid agonists, but the less active (R)-enantiomer of this compound, AD-1211, is a mixed agonist–antagonist at opioid receptors with a similar pharmacological profile to pentazocine, and has atypical opioid effects with little development of tolerance or dependence seen after extended administration in animal studies.

Lanicemine (AZD6765) is a low-trapping NMDA receptor antagonist that was under development by AstraZeneca for the management of severe and treatment-resistant depression. Lanicemine differs from ketamine in that it is a low-trapping NMDA receptor antagonist, showing similar rapid-acting antidepressant effects to ketamine in clinical trials but with little or no psychotomimetic side effects. However, lanicemine did not meet study endpoints, and its development was terminated by AstraZeneca in 2013.

Hamilton Morris is an American journalist, documentarian, and scientific researcher. He is the creator and director of the television series Hamilton's Pharmacopeia, in which he investigates the chemistry, history, and cultural impact of various psychoactive drugs.
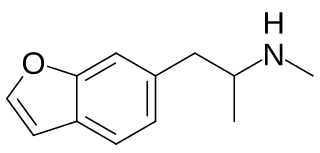
6-MAPB is a psychedelic and entactogenic drug which is structurally related to 6-APB and MDMA. It is not known to have been widely sold as a "designer drug" but has been detected in analytical samples taken from individuals hospitalised after using drug combinations that included other benzofuran derivatives. 6-MAPB was banned in the UK in June 2013, along with 9 other related compounds which were thought to produce similar effects.
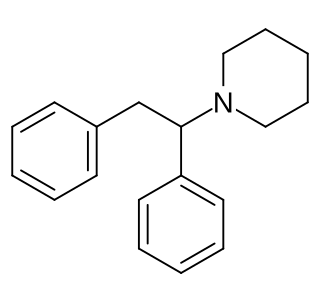
Diphenidine is a dissociative anesthetic that has been sold as a designer drug. The synthesis of diphenidine was first reported in 1924, and employed a Bruylants reaction analogous to the one that would later be used to discover phencyclidine in 1956. Shortly after the 2013 UK ban on arylcyclohexylamines, diphenidine and the related compound methoxphenidine became available on the grey market. Anecdotal reports describe high doses of diphenidine producing "bizarre somatosensory phenomena and transient anterograde amnesia." Diphenidine and related diarylethylamines have been studied in vitro as treatments for neurotoxic injury and are antagonists of the NMDA receptor. In dogs diphenidine exhibits greater antitussive potency than codeine phosphate.
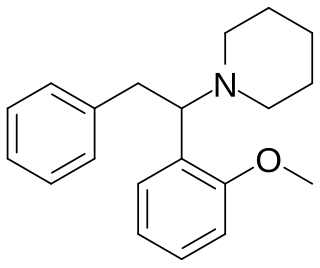
Methoxphenidine is a dissociative of the diarylethylamine class that has been sold online as a designer drug. Methoxphenidine was first reported in a 1989 patent where it was tested as a treatment for neurotoxic injury. Shortly after the 2013 UK ban on arylcyclohexylamines methoxphenidine and the related compound diphenidine became available on the gray market, where it has been encountered as a powder and in tablet form. Though diphenidine possesses higher affinity for the NMDA receptor, anecdotal reports suggest methoxphenidine has greater oral potency. Of the three isomeric anisyl-substituents methoxphenidine has affinity for the NMDA receptor that is higher than 4-MeO-Diphenidine but lower than 3-MeO-Diphenidine, a structure–activity relationship shared by the arylcyclohexylamines.

3-Fluorophenmetrazine is a phenylmorpholine-based stimulant and fluorinated analogue of phenmetrazine that has been sold online as a designer drug.

3-Methoxyeticyclidine (3-MeO-PCE), also known as methoxieticyclidine, is a dissociative anesthetic that is qualitatively similar to PCE and PCP and has been sold online as a designer drug.
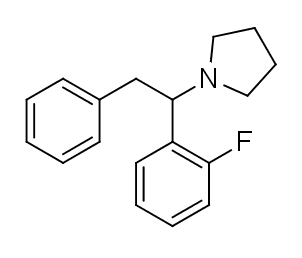
Fluorolintane is a dissociative anesthetic drug that has been sold online as a designer drug.

UWA-001 is a phenethylamine derivative invented at the University of Western Australia as non-toxic alternative to 3,4-methylenedioxy-N-methylamphetamine (MDMA) and researched as a potential treatment for Parkinson's disease.
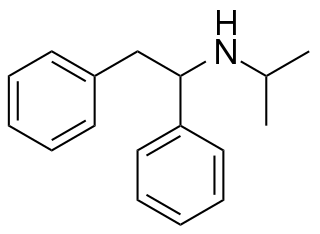
NPDPA is a dissociative anesthetic that has been sold online as a designer drug. It was first identified in Germany in 2008, and while it has never been as widely sold as related compounds such as diphenidine and ephenidine, it has continued to show up in seized drug samples occasionally, and was banned in Sweden in 2015.

Diphenpipenol is an opioid analgesic drug invented in the 1970s by Dainippon Pharmaceutical Co. It is chemically a 1-substituted-4-(1,2-diphenylethyl)piperazine derivative related to compounds such as MT-45 and AD-1211, but diphenpipenol is the most potent compound in the series, with the more active (S) enantiomer being around 105 times the potency of morphine in animal studies. This makes it a similar strength to fentanyl and its analogues, and consequently diphenpipenol can be expected to pose a significant risk of producing life-threatening respiratory depression, as well as other typical opioid side effects such as sedation, itching, nausea and vomiting.
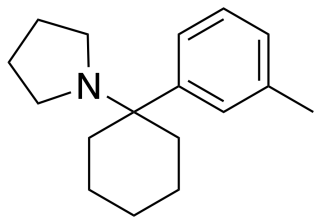
3-Methyl-PCPy (3-Me-PCPy) is an arylcyclohexylamine derivative with an unusual spectrum of pharmacological effects, acting as both a potent NMDA antagonist and also a triple reuptake inhibitor which inhibits reuptake of all three monoamine neurotransmitters serotonin, dopamine and noradrenaline. It also acts as a high affinity sigma receptor ligand, selective for the σ2 subtype. It produces both stimulant and dissociative effects in animal behavioural studies.

4-EA-NBOMe is a substituted amphetamine and 25-NB derivative which has been sold as a designer drug. It was first identified by a forensic laboratory in Germany in 2014, but while its analytical properties and metabolism have been studied, its pharmacology remains unknown.

Propylphenidine is a compound which has been sold as a designer drug, first identified in Australia in 2023. It is unclear what kind of activity it has, as it is claimed to be a stimulant in a patent along with related compounds such as phenylpropylaminopentane, but other structurally similar compounds such as ephenidine and isophenidine are dissociative anesthetic drugs, so it may produce either kind of activity or perhaps a mixture of both.