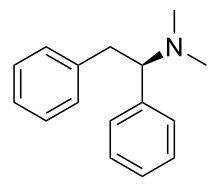
Thebaine (paramorphine), also known as codeine methyl enol ether, is an opiate alkaloid, its name coming from the Greek Θῆβαι, Thēbai (Thebes), an ancient city in Upper Egypt. A minor constituent of opium, thebaine is chemically similar to both morphine and codeine, but has stimulatory rather than depressant effects. At high doses, it causes convulsions similar to strychnine poisoning. The synthetic enantiomer (+)-thebaine does show analgesic effects apparently mediated through opioid receptors, unlike the inactive natural enantiomer (−)-thebaine. While thebaine is not used therapeutically, it is the main alkaloid extracted from Papaver bracteatum and can be converted industrially into a variety of compounds, including hydrocodone, hydromorphone, oxycodone, oxymorphone, nalbuphine, naloxone, naltrexone, buprenorphine, butorphanol and etorphine.

Tramadol, sold under the brand name Ultram among others, is an opioid pain medication and a serotonin–norepinephrine reuptake inhibitor (SNRI) used to treat moderately severe pain. When taken by mouth in an immediate-release formulation, the onset of pain relief usually begins within an hour. It is also available by injection. It is available in combination with paracetamol (acetaminophen).

Hydromorphone, also known as dihydromorphinone, and sold under the brand name Dilaudid among others, is a morphinan opioid used to treat moderate to severe pain. Typically, long-term use is only recommended for pain due to cancer. It may be used by mouth or by injection into a vein, muscle, or under the skin. Effects generally begin within half an hour and last for up to five hours. A 2016 Cochrane review found little difference in benefit between hydromorphone and other opioids for cancer pain.

Opioids are a class of drugs that derive from, or mimic, natural substances found in the opium poppy plant. Opioids work in the brain to produce a variety of effects, including pain relief. As a class of substances, they act on opioid receptors to produce morphine-like effects.

Opioid use disorder (OUD) is a substance use disorder characterized by cravings for opioids, continued use despite physical and/or psychological deterioration, increased tolerance with use, and withdrawal symptoms after discontinuing opioids. Opioid withdrawal symptoms include nausea, muscle aches, diarrhea, trouble sleeping, agitation, and a low mood. Addiction and dependence are important components of opioid use disorder.

Buprenorphine, sold under the brand name Subutex among others, is an opioid used to treat opioid use disorder, acute pain, and chronic pain. It can be used under the tongue (sublingual), in the cheek (buccal), by injection, as a skin patch (transdermal), or as an implant. For opioid use disorder, the patient must have moderate opioid withdrawal symptoms before buprenorphine can be administered under direct observation of a health-care provider.
Physical dependence is a physical condition caused by chronic use of a tolerance-forming drug, in which abrupt or gradual drug withdrawal causes unpleasant physical symptoms. Physical dependence can develop from low-dose therapeutic use of certain medications such as benzodiazepines, opioids, stimulants, antiepileptics and antidepressants, as well as the recreational misuse of drugs such as alcohol, opioids and benzodiazepines. The higher the dose used, the greater the duration of use, and the earlier age use began are predictive of worsened physical dependence and thus more severe withdrawal syndromes. Acute withdrawal syndromes can last days, weeks or months. Protracted withdrawal syndrome, also known as post-acute-withdrawal syndrome or "PAWS", is a low-grade continuation of some of the symptoms of acute withdrawal, typically in a remitting-relapsing pattern, often resulting in relapse and prolonged disability of a degree to preclude the possibility of lawful employment. Protracted withdrawal syndrome can last for months, years, or depending on individual factors, indefinitely. Protracted withdrawal syndrome is noted to be most often caused by benzodiazepines. To dispel the popular misassociation with addiction, physical dependence to medications is sometimes compared to dependence on insulin by persons with diabetes.
Substance dependence, also known as drug dependence, is a biopsychological situation whereby an individual's functionality is dependent on the necessitated re-consumption of a psychoactive substance because of an adaptive state that has developed within the individual from psychoactive substance consumption that results in the experience of withdrawal and that necessitates the re-consumption of the drug. A drug addiction, a distinct concept from substance dependence, is defined as compulsive, out-of-control drug use, despite negative consequences. An addictive drug is a drug which is both rewarding and reinforcing. ΔFosB, a gene transcription factor, is now known to be a critical component and common factor in the development of virtually all forms of behavioral and drug addictions, but not dependence.

Nalbuphine, sold under the brand names Nubain among others, is an opioid analgesic which is used in the treatment of pain. It is given by injection into a vein, muscle, or fat.
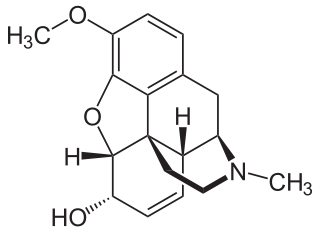
Codeine is an opiate and prodrug of morphine mainly used to treat pain, coughing, and diarrhea. It is also commonly used as a recreational drug. It is found naturally in the sap of the opium poppy, Papaver somniferum. It is typically used to treat mild to moderate degrees of pain. Greater benefit may occur when combined with paracetamol (acetaminophen) or a nonsteroidal anti-inflammatory drug (NSAID) such as aspirin or ibuprofen. Evidence does not support its use for acute cough suppression in children or adults. In Europe, it is not recommended as a cough medicine in those under 12 years of age. It is generally taken by mouth. It typically starts working after half an hour, with maximum effect at two hours. Its effects last for about four to six hours. Codeine exhibits abuse potential similar to other opioid medications, including a risk of habituation and overdose.

Benzylmorphine (Peronine) is a semi-synthetic opioid narcotic introduced to the international market in 1896 and that of the United States very shortly thereafter. It is much like codeine, containing a benzyl group attached to the morphine molecule just as the methyl group creates codeine and the ethyl group creates ethylmorphine or dionine. It is about 90% as strong as codeine by weight.

Oripavine is an opioid and the major metabolite of thebaine. It is the parent compound from which a series of semi-synthetic opioids are derived, which includes the compounds etorphine and buprenorphine. Although its analgesic potency is comparable to morphine, it is not used clinically due to its severe toxicity and low therapeutic index. Due to its use in manufacture of strong opioids, oripavine is a controlled substance in some jurisdictions.

Propiram is a partial μ-opioid receptor agonist and weak μ antagonist analgesic from the ampromide family of drugs related to other drugs such as phenampromide and diampromide. It was invented in 1963 in the United Kingdom by Bayer but was not widely marketed, although it saw some limited clinical use, especially in dentistry. Propiram reached Phase III clinical trials in the United States and Canada.
Drug detoxification is variously construed or interpreted as a type of "medical" intervention or technique in regards to a physical dependence mediated by a drug; as well as the process and experience of a withdrawal syndrome or any of the treatments for acute drug overdose (toxidrome). The first definition however, in relation to substance dependence and its treatment is arguably a misnomer and even directly contradictory since withdrawal is neither contingent upon nor alleviated through biological excretion or clearance of the drug. In fact, excretion of a given drug from the body is one of the very processes that leads to withdrawal since the syndrome arises largely due to the cessation itself and the drug being absent from the body; especially the blood plasma, not from ‘leftover toxins’ or traces of the drug still being in the system.

An opiate is an alkaloid substance derived from opium. It differs from the similar term opioid in that the latter is used to designate all substances, both natural and synthetic, that bind to opioid receptors in the brain. Opiates are alkaloid compounds naturally found in the opium poppy plant Papaver somniferum. The psychoactive compounds found in the opium plant include morphine, codeine, and thebaine. Opiates have long been used for a variety of medical conditions, with evidence of opiate trade and use for pain relief as early as the eighth century AD. Most opiates are considered drugs with moderate to high abuse potential and are listed on various "Substance-Control Schedules" under the Uniform Controlled Substances Act of the United States of America.

MT-45 (IC-6) is an opioid analgesic drug invented in the 1970s by Dainippon Pharmaceutical Co. It is chemically a 1-substituted-4-(1,2-diphenylethyl) piperazine derivative, which is structurally unrelated to most other opioid drugs. Racemic MT-45 has around 80% the potency of morphine, with almost all opioid activity residing in the (S) enantiomer. It has been used as a lead compound from which a large family of potent opioid drugs have been developed, including full agonists, partial agonists, and antagonists at the three main opioid receptor subtypes. Fluorinated derivatives of MT-45 such as 2F-MT-45 are significantly more potent as μ-opioid receptor agonists, and one of its main metabolites 1,2-diphenylethylpiperazine also blocks NMDA receptors.

AD-1211 is an opioid analgesic drug invented in the 1970s by Dainippon Pharmaceutical Co. It is chemically a 1-substituted-4-prenyl-piperazine derivative, which is structurally unrelated to most other opioid drugs. The (S)-enantiomers in this series are more active as opioid agonists, but the less active (R)-enantiomer of this compound, AD-1211, is a mixed agonist–antagonist at opioid receptors with a similar pharmacological profile to pentazocine, and has atypical opioid effects with little development of tolerance or dependence seen after extended administration in animal studies.

Buprenorphine/naloxone, sold under the brand name Suboxone among others, is a fixed-dose combination medication that includes buprenorphine and naloxone. It is used to treat opioid use disorder, and reduces the mortality of opioid use disorder by 50%. It relieves cravings to use and withdrawal symptoms. Buprenorphine/naloxone is available for use in two different forms, under the tongue or in the cheek.
Clinical Opiate Withdrawal Scale (COWS) is a method used by registered practitioners to measure the severity of a patient's opioid withdrawal symptoms. This method consists of a series of 11 topics each comprising 4 - 5 common symptoms experienced by a patient undergoing opioid withdrawal. In each topic a rank is given depending on what the patient responds to. Generally, 0 is considered to be no symptom shown and 4 or 5 is considered to be the most common and severe symptom shown. These results are then added up and a final diagnosis is made based on the value obtained. This test is crucial as it allows the practitioner to assess the physiological and psychological behaviours of the patient as well as the severity of each symptom during the duration of the examination. The results are grouped into 3 categories of mild, moderately severe and severe. Mild consists of 5 to 12 points, moderately severe consists of 13 to 24 points and anything above 36 points is severe and requires direct medical attention.

Opioid withdrawal is a set of symptoms arising from the sudden withdrawal or reduction of opioids where previous usage has been heavy and prolonged. Signs and symptoms of withdrawal can include drug craving, anxiety, restless legs, nausea, vomiting, diarrhea, sweating, and an increased heart rate. Opioid use triggers a rapid adaptation in cellular signalling pathways that means, when rapidly withdrawn, there can be adverse physiological effects. All opioids, both recreational drugs and medications, when reduced or stopped, can lead to opioid withdrawal symptoms. When withdrawal symptoms are due to recreational opioid use, the term opioid use disorder is used, whereas when due to prescribed medications, the term prescription opioid use disorder is used. Opioid withdrawal can be helped by the use of opioid replacement therapy, and symptoms may be relieved by the use of medications including lofexidine and clonidine.