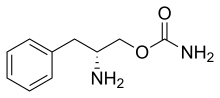
Modafinil, sold under the brand name Provigil among others, is a wakefulness-promoting medication used primarily to treat narcolepsy. Modafinil is also approved for stimulating wakefulness in people with sleep apnea and shift work sleep disorder. It is taken by mouth. Modafinil is not approved by the US Food and Drug Administration (FDA) for use in people under age 17.

Methylphenidate, sold under the brand names Ritalin and Concerta among others, is a potent central nervous system (CNS) stimulant used medically to treat attention deficit hyperactivity disorder (ADHD) and, to a lesser extent, narcolepsy. It is a primary medication for ADHD ; it may be taken by mouth or applied to the skin, and different formulations have varying durations of effect, commonly ranging from 2-4 hours. For ADHD, the effectiveness of methylphenidate is comparable to atomoxetine but modestly lower than amphetamines.
A dopamine reuptake inhibitor (DRI) is a class of drug which acts as a reuptake inhibitor of the monoamine neurotransmitter dopamine by blocking the action of the dopamine transporter (DAT). Reuptake inhibition is achieved when extracellular dopamine not absorbed by the postsynaptic neuron is blocked from re-entering the presynaptic neuron. This results in increased extracellular concentrations of dopamine and increase in dopaminergic neurotransmission.
Hypersomnia is a neurological disorder of excessive time spent sleeping or excessive sleepiness. It can have many possible causes and can cause distress and problems with functioning. In the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5), hypersomnolence, of which there are several subtypes, appears under sleep-wake disorders.
Upper airway resistance syndrome (UARS) is a sleep disorder characterized by the narrowing of the airway that can cause disruptions to sleep. The symptoms include unrefreshing sleep, fatigue, sleepiness, chronic insomnia, and difficulty concentrating. UARS can be diagnosed by polysomnograms capable of detecting Respiratory Effort-related Arousals. It can be treated with lifestyle changes, functional orthodontics, surgery, mandibular repositioning devices or CPAP therapy. UARS is considered a variant of sleep apnea, although some scientists and doctors believe it to be a distinct disorder.
A microsleep is a sudden temporary episode of sleep or drowsiness which may last for a few seconds where an individual fails to respond to some arbitrary sensory input and becomes unconscious. Episodes of microsleep occur when an individual loses and regains awareness after a brief lapse in consciousness, often without warning, or when there are sudden shifts between states of wakefulness and sleep. In behavioural terms, MSs may manifest as droopy eyes, slow eyelid-closure, and head nodding. In electrical terms, microsleeps are often classified as a shift in electroencephalography (EEG) during which 4–7 Hz activity replaces the waking 8–13 Hz background rhythm.

Pemoline, sold under the brand name Cylert among others, is a stimulant medication which has been used in the treatment of attention-deficit hyperactivity disorder (ADHD) and narcolepsy. It has been discontinued in most countries due to rare but serious problems with liver toxicity. The medication was taken by mouth.

Viloxazine, sold under the brand name Qelbree and formerly as Vivalan among others, is a selective norepinephrine reuptake inhibitor medication which is used in the treatment of attention deficit hyperactivity disorder (ADHD) in children and adults. It was marketed for almost 30 years as an antidepressant for the treatment of depression before being discontinued and subsequently repurposed as a treatment for ADHD. Viloxazine is taken orally. It was used as an antidepressant in an immediate-release form and is used in ADHD in an extended-release form.

Armodafinil (trade name Nuvigil) is the enantiopure compound of the eugeroic modafinil (Provigil). It consists of only the (R)-(−)-enantiomer of the racemic modafinil. Armodafinil is produced by the pharmaceutical company Cephalon Inc. and was approved by the U.S. Food and Drug Administration (FDA) in June 2007. In 2016, the FDA granted Mylan rights for the first generic version of Cephalon's Nuvigil to be marketed in the U.S.
Excessive daytime sleepiness (EDS) is characterized by persistent sleepiness and often a general lack of energy, even during the day after apparently adequate or even prolonged nighttime sleep. EDS can be considered as a broad condition encompassing several sleep disorders where increased sleep is a symptom, or as a symptom of another underlying disorder like narcolepsy, circadian rhythm sleep disorder, sleep apnea or idiopathic hypersomnia.
Christian Guilleminault was a French physician and researcher in the field of sleep medicine who played a central role in the early discovery of obstructive sleep apnea and made seminal discoveries in many other areas of sleep medicine.

Narcolepsy is a chronic neurological disorder that impairs the ability to regulate sleep–wake cycles, and specifically impacts REM sleep. The pentad symptoms of narcolepsy include excessive daytime sleepiness (EDS), sleep related hallucinations, sleep paralysis, disturbed nocturnal sleep (DNS) and cataplexy. There are two recognized forms of narcolepsy, narcolepsy type 1 and type 2. Narcolepsy type 1 (NT1) can be clinically characterized by symptoms of EDS and cataplexy, and/or will have CSF orexin levels of less than 110 pg/ml. Cataplexy are transient episodes of aberrant tone, most typically loss of tone, that can be associated with strong emotion. In pediatric onset narcolepsy, active motor phenomena are not uncommon. Cataplexy may be mistaken for syncope, tic disorder or seizures. Narcolepsy type 2 (NT2) does not have features of cataplexy and CSF orexin levels are normal. Sleep related hallucinations, also known as hypnogogic and hypnopompic are vivid hallucinations that can be auditory, visual or tactile and may occur independent of or in combination with an inability to move. People with narcolepsy tend to sleep about the same number of hours per day as people without it, but the quality of sleep is typically compromised.
Pitolisant, sold under the brand name Wakix among others, is a medication used for the treatment of excessive daytime sleepiness in adults with narcolepsy. It is a histamine 3 (H3) receptor antagonist/inverse agonist (an antihistamine drug specific to that kind of receptors). It represents the first commercially available medication in its class, so that the US Food and Drug Administration (FDA) declares it a first-in-class medication. Pitolisant enhances the activity of histaminergic neurons in the brain that function to improve a person's wakefulness. It was approved by the European Medicines Agency (EMA) in March 2016 for narcolepsy with or without cataplexy, and for excessive daytime sleepiness by the FDA in August 2019. The most common side effects include difficulty sleeping, nausea, and feeling worried.
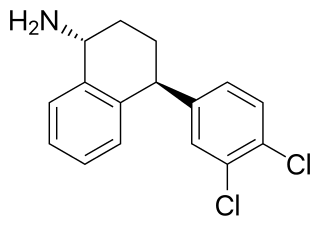
Dasotraline is a serotonin-norepinephrine-dopamine reuptake inhibitor (SNDRI) that was under development by Sunovion for the treatment of attention-deficit hyperactivity disorder (ADHD) and binge eating disorder (BED). Structurally, dasotraline is a stereoisomer of desmethylsertraline (DMS), which is an active metabolite of the marketed selective serotonin reuptake inhibitor (SSRI) antidepressant sertraline (Zoloft).

A norepinephrine releasing agent (NRA), also known as an adrenergic releasing agent, is a catecholaminergic type of drug that induces the release of norepinephrine (noradrenaline) and epinephrine (adrenaline) from the pre-synaptic neuron into the synapse. This in turn leads to increased extracellular concentrations of norepinephrine and epinephrine therefore an increase in adrenergic neurotransmission.

Eugeroics, also known as wakefulness-promoting agents and wakefulness-promoting drugs, are a class of drugs that promote wakefulness and alertness. They are medically indicated for the treatment of certain sleep disorders including excessive daytime sleepiness (EDS) in narcolepsy or obstructive sleep apnea (OSA). Eugeroics are also often prescribed off-label for the treatment of EDS in idiopathic hypersomnia. In contrast to classical psychostimulants, such as methylphenidate and amphetamine, which are also used in the treatment of these disorders, eugeroics typically do not produce marked euphoria, and, consequently, have a lower addictive potential.

A norepinephrine–dopamine reuptake inhibitor (NDRI) is a drug used for the treatment of clinical depression, attention deficit hyperactivity disorder (ADHD), narcolepsy, and the management of Parkinson's disease. The drug acts as a reuptake inhibitor for the neurotransmitters norepinephrine and dopamine by blocking the action of the norepinephrine transporter (NET) and the dopamine transporter (DAT), respectively. This in turn leads to increased extracellular concentrations of both norepinephrine and dopamine and, therefore, an increase in adrenergic and dopaminergic neurotransmission.
Idiopathic hypersomnia(IH) is a neurological disorder which is characterized primarily by excessive sleep and excessive daytime sleepiness (EDS). Idiopathic hypersomnia was first described by Bedrich Roth in 1976, and it can be divided into two forms: polysymptomatic and monosymptomatic. The condition typically becomes evident in early adulthood and most patients diagnosed with IH will have had the disorder for many years prior to their diagnosis. As of August 2021, an FDA-approved medication exists for IH called Xywav, which is oral solution of calcium, magnesium, potassium, and sodium oxybates; in addition to several off-label treatments (primarily FDA-approved narcolepsy medications).