
Phenmetrazine is a stimulant drug first synthesized in 1952 and originally used as an appetite suppressant, but withdrawn from the market in the 1980s due to widespread abuse. It was initially replaced by its analogue phendimetrazine which functions as a prodrug to phenmetrazine, but now it is rarely prescribed, due to concerns of abuse and addiction. Chemically, phenmetrazine is a substituted amphetamine containing a morpholine ring.
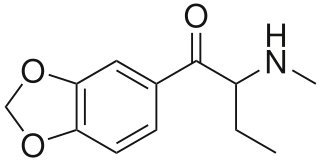
Butylone, also known as β-keto-N-methylbenzodioxolylbutanamine (βk-MBDB), is an entactogen, psychedelic, and stimulant psychoactive drug of the phenethylamine chemical class. It is the β-keto analogue of MBDB and the substituted methylenedioxyphenethylamine analogue of buphedrone.

Methallylescaline (4-Methylallyloxy-3,5-dimethoxyphenethylamine) is a lesser-known psychedelic drug. It is the 4-methyl analog of allylescaline. Methallylescaline was first synthesized by Alexander Shulgin. In his book PiHKAL, the dosage range is listed as 40–65 mg and the duration is listed as 12–16 hours. Little data exists about the pharmacological properties, metabolism, and toxicity of methallylescaline, with effects comparable to that of other mescaline analogs and has been sold as a designer drug. Contrary to mescaline (IC50 = 6.3 μM), methallylescaline is unable to bind the serotonin receptor 5-HT2A, but can bind 5-HT2C with the same strength as mescaline (~17 μM). At very high doses (30 μg/kg) methallylescaline, as well as the more common psychedelic DOI, causes neuroinflammation and loss of serotonergic neurons in mice.

Dimethocaine, also known as DMC or larocaine, is a compound with a stimulatory effect. This effect resembles that of cocaine, although dimethocaine appears to be less potent. Just like cocaine, dimethocaine is addictive due to its stimulation of the reward pathway in the brain. However, dimethocaine is a legal cocaine replacement in some countries and is even listed by the European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) under the category “synthetic cocaine derivatives”. The structure of dimethocaine, being a 4-aminobenzoic acid ester, resembles that of procaine. It is found as a white powder at room temperature.

5-Iodo-2-aminoindane (5-IAI) is a drug which acts as a releasing agent of serotonin, norepinephrine, and dopamine. It was developed in the 1990s by a team led by David E. Nichols at Purdue University. 5-IAI fully substitutes for MDMA in rodents and is a putative entactogen in humans. Unlike related aminoindane derivatives like MDAI and MMAI, 5-IAI causes some serotonergic neurotoxicity in rats, but is substantially less toxic than its corresponding amphetamine homologue pIA, with the damage observed barely reaching statistical significance.

5-(2-Aminopropyl)indole is an indole and phenethylamine derivative with empathogenic effects. Its preparation was first reported by Albert Hofmann in 1962. It is a designer drug that has been openly sold as a recreational drug by online vendors since 2011.

3-Fluoroamphetamine is a stimulant drug from the amphetamine family which acts as a monoamine releaser with similar potency to methamphetamine but more selectivity for dopamine and norepinephrine release over serotonin. It is self-administered by mice to a similar extent to related drugs such as 4-fluoroamphetamine and 3-methylamphetamine.

Pentylone is a stimulant developed in the 1960s. It is a substituted cathinone. It has been identified in some samples of powders sold as "NRG-1", along with varying blends of other cathinone derivatives including flephedrone, MDPBP, MDPV and 4-MePPP. It was also found in combination with 4-MePPP being sold as "NRG-3". Reports indicate side effects include feelings of paranoia, agitation and inability to sleep, with effects lasting for several days at high doses.
6-APB is an empathogenic psychoactive compound of the substituted benzofuran and substituted phenethylamine classes. 6-APB and other compounds are sometimes informally called "Benzofury" in newspaper reports. It is similar in structure to MDA, but differs in that the 3,4-methylenedioxyphenyl ring system has been replaced with a benzofuran ring. 6-APB is also the unsaturated benzofuran derivative of 6-APDB. It may appear as a tan grainy powder. While the drug never became particularly popular, it briefly entered the rave and underground clubbing scene in the UK before its sale and import were banned. It falls under the category of research chemicals, sometimes called "legal highs." Because 6-APB and other substituted benzofurans have not been explicitly outlawed in some countries, they are often technically legal, contributing to their popularity.

Pseudophenmetrazine is a psychostimulant compound of the morpholine class. It is the N-demethylated and cis-configured analogue of phendimetrazine as well as the cis-configured stereoisomer of phenmetrazine. In addition, along with phenmetrazine, it is believed to be one of the active metabolites of phendimetrazine, which itself is inactive and behaves merely as a prodrug. Relative to phenmetrazine, pseudophenmetrazine is of fairly low potency, acting as a modest releasing agent of norepinephrine (EC50 = 514 nM), while its (+)-enantiomer is a weak releaser of dopamine (EC50 = 1,457 nM) whereas its (−)-enantiomer is a weak reuptake inhibitor of dopamine (Ki = 2,691 nM); together as a racemic mixture with the two enantiomers combined, pseudophenmetrazine behaves overall more as a dopamine reuptake inhibitor (Ki = 2,630 nM), possibly due to the (+)-enantiomer blocking the uptake of the (−)-enantiomer into dopaminergic neurons and thus preventing it from inducing dopamine release. Neither enantiomer has any significant effect on serotonin reuptake or release (both Ki = >10,000 nM and EC50 = >10,000 nM, respectively).

βk-2C-B (βeta-keto-4-bromo-2,5-dimethoxyphenylamine), also known as bk-2C-B, is a novel psychedelic substance. It is the beta (β) ketone structural analogue of 2C-B, a psychedelic drug of the 2C family. It is used as a recreational drug, usually taken orally. βk-2C-B is a controlled substance in Canada, Germany, Switzerland, and the United Kingdom.

Ephenidine is a dissociative anesthetic that has been sold online as a designer drug. It is illegal in some countries as a structural isomer of the banned opioid drug lefetamine, but has been sold in countries where it is not yet banned.

3-Methoxyeticyclidine (3-MeO-PCE), also known as methoxieticyclidine, is a dissociative anesthetic that is qualitatively similar to PCE and PCP and has been sold online as a designer drug.

MDMB-CHMINACA (also known as MDMB(N)-CHM) is an indazole-based synthetic cannabinoid that acts as a potent agonist of the CB1 receptor, and has been sold online as a designer drug. It was invented by Pfizer in 2008, and is one of the most potent cannabinoid agonists known, with a binding affinity of 0.0944 nM at CB1, and an EC50 of 0.330 nM. It is closely related to MDMB-FUBINACA, which caused at least 1000 hospitalizations and 40 deaths in Russia as consequence of intoxication.
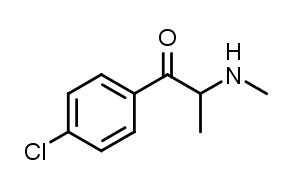
4-Chloromethcathinone is a stimulant drug of the cathinone class that has been sold online as a designer drug.

Substituted phenylmorpholines, or substituted phenmetrazines alternatively, are chemical derivatives of phenylmorpholine or of the psychostimulant drug phenmetrazine. Most such compounds act as releasers of monoamine neurotransmitters, and have stimulant effects. Some also act as agonists at serotonin receptors, and compounds with an N-propyl substitution act as dopamine receptor agonists. A number of derivatives from this class have been investigated for medical applications, such as for use as anorectics or medications for the treatment of ADHD. Some compounds have also become subject to illicit use as designer drugs.

Mexedrone is a stimulant and an entactogen drug of the cathinone class that has been sold online as a designer drug. It is the alpha-methoxy derivative of Mephedrone.
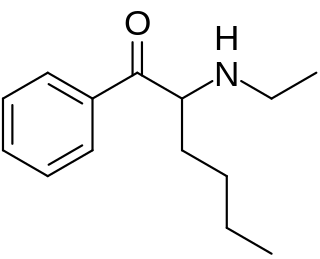
N-Ethylhexedrone (also known as α-ethylaminocaprophenone, N-ethylnorhexedrone, hexen, and NEH) is a stimulant of the cathinone class that acts as a norepinephrine–dopamine reuptake inhibitor (NDRI) with IC50 values of 0.0978 and 0.0467 μM, respectively. N-Ethylhexedrone was first mentioned in a series of patents by Boehringer Ingelheim in the 1960s which led to the development of the better-known drug methylenedioxypyrovalerone (MDPV). Since the mid-2010s, N-ethylhexedrone has been sold online as a designer drug. In 2018, N-ethylhexedrone was the second most common drug of the cathinone class to be identified in Drug Enforcement Administration seizures.

ortho-Methylphenylpiperazine (also known as oMPP, oMePP, 1-(2-methylphenyl)piperazine, 2-MPP, and 2-MePP) is a psychoactive designer drug of the phenylpiperazine group. It acts as a serotonin–norepinephrine–dopamine releasing agent (SNDRA), with EC50 values for induction of monoamine release of 175 nM for serotonin, 39.1 nM for norepinephrine, and 296–542 nM for dopamine. As such, it has about 4.5-fold preference for induction of norepinephrine release over serotonin, and about 7.6- to 13.9-fold preference for induction of norepinephrine release over dopamine.

4-Methylphenmetrazine is a recreational designer drug with stimulant effects. It is a substituted phenylmorpholine derivative, closely related to better known drugs such as phenmetrazine and 3-fluorophenmetrazine. It was first identified in Slovenia in 2015, and has been shown to act as a monoamine releaser with some preference for serotonin release.




















