
In mammalian cells, vinculin is a membrane-cytoskeletal protein in focal adhesion plaques that is involved in linkage of integrin adhesion molecules to the actin cytoskeleton. Vinculin is a cytoskeletal protein associated with cell-cell and cell-matrix junctions, where it is thought to function as one of several interacting proteins involved in anchoring F-actin to the membrane.

Merlin is a cytoskeletal protein. In humans, it is a tumor suppressor protein involved in neurofibromatosis type II. Sequence data reveal its similarity to the ERM protein family.

Cortactin is a monomeric protein located in the cytoplasm of cells that can be activated by external stimuli to promote polymerization and rearrangement of the actin cytoskeleton, especially the actin cortex around the cellular periphery. It is present in all cell types. When activated, it will recruit Arp2/3 complex proteins to existing actin microfilaments, facilitating and stabilizing nucleation sites for actin branching. Cortactin is important in promoting lamellipodia formation, invadopodia formation, cell migration, and endocytosis.

Leukosialin also known as sialophorin or CD43 is a transmembrane cell surface protein that in humans is encoded by the SPN (sialophorin) gene.
Talin is a high-molecular-weight cytoskeletal protein concentrated at regions of cell–substratum contact and, in lymphocytes, at cell–cell contacts. Discovered in 1983 by Keith Burridge and colleagues, talin is a ubiquitous cytosolic protein that is found in high concentrations in focal adhesions. It is capable of linking integrins to the actin cytoskeleton either directly or indirectly by interacting with vinculin and α-actinin.

Ezrin also known as cytovillin or villin-2 is a protein that in humans is encoded by the EZR gene.

Zonula occludens-1 ZO-1, also known as Tight junction protein-1 is a 220-kD peripheral membrane protein that is encoded by the TJP1 gene in humans. It belongs to the family of zonula occludens proteins, which are tight junction-associated proteins and of which, ZO-1 is the first to be cloned. It was first isolated in 1986 by Stevenson and Goodenough using a monoclonal antibody raised in rodent liver to recognise a 225-kD polypeptide in whole liver homogenates and in tight junction-enriched membrane fractions. It has a role as a scaffold protein which cross-links and anchors Tight Junction (TJ) strand proteins, which are fibril-like structures within the lipid bilayer, to the actin cytoskeleton.

Sodium-hydrogen antiporter 3 regulator 1 is a regulator of Sodium-hydrogen antiporter 3. It is encoded by the gene SLC9A3R1. It is also known as ERM Binding Protein 50 (EBP50) or Na+/H+ Exchanger Regulatory Factor (NHERF1). It is believed to interact via long-range allostery, involving significant protein dynamics.

Intercellular adhesion molecule 2 (ICAM2), also known as CD102, is a human gene, and the protein resulting from it.

Sodium-hydrogen exchange regulatory cofactor NHE-RF2 (NHERF-2) also known as tyrosine kinase activator protein 1 (TKA-1) or SRY-interacting protein 1 (SIP-1) is a protein that in humans is encoded by the SLC9A3R2 gene.

Moesin is a protein that in humans is encoded by the MSN gene.

Vasodilator-stimulated phosphoprotein is a protein that in humans is encoded by the VASP gene.

Radixin is a protein that in humans is encoded by the RDX gene.

Zyxin is a protein that in humans is encoded by the ZYX gene.

Microtubule-actin cross-linking factor 1, isoforms 1/2/3/5 is a protein that in humans is encoded by the MACF1 gene.

Myosin X, also known as MYO10, is a protein that in humans is encoded by the MYO10 gene.

Myosin regulatory light chain interacting protein, also known as MYLIP, is a protein that in humans is encoded by the MYLIP gene.

αE-catenin, also known as Catenin alpha-1 is a protein that in humans is encoded by the CTNNA1 gene. αE-catenin is highly expressed in cardiac muscle and localizes to adherens junctions at intercalated disc structures where it functions to mediate the anchorage of actin filaments to the sarcolemma. αE-catenin also plays a role in tumor metastasis and skin cell function.
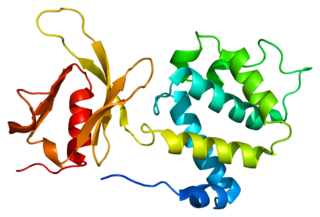
Talin-1 is a protein that in humans is encoded by the TLN1 gene. Talin-1 is ubiquitously expressed, and is localized to costamere structures in cardiac and skeletal muscle cells, and to focal adhesions in smooth muscle and non-muscle cells. Talin-1 functions to mediate cell-cell adhesion via the linkage of integrins to the actin cytoskeleton and in the activation of integrins. Altered expression of talin-1 has been observed in patients with heart failure, however no mutations in TLN1 have been linked with specific diseases.
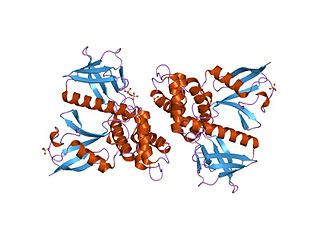
In molecular biology, the FERM domain is a widespread protein module involved in localising proteins to the plasma membrane. FERM domains are found in a number of cytoskeletal-associated proteins that associate with various proteins at the interface between the plasma membrane and the cytoskeleton. The FERM domain is located at the N terminus in the majority of proteins in which it is found.



















