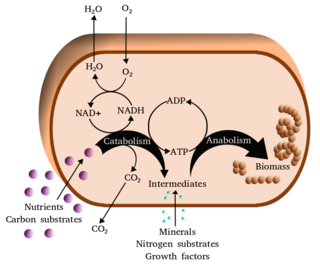
Metabolism is the set of life-sustaining chemical reactions in organisms. The three main functions of metabolism are: the conversion of the energy in food to energy available to run cellular processes; the conversion of food to building blocks for proteins, lipids, nucleic acids, and some carbohydrates; and the elimination of metabolic wastes. These enzyme-catalyzed reactions allow organisms to grow and reproduce, maintain their structures, and respond to their environments. The word metabolism can also refer to the sum of all chemical reactions that occur in living organisms, including digestion and the transportation of substances into and between different cells, in which case the above described set of reactions within the cells is called intermediary metabolism.
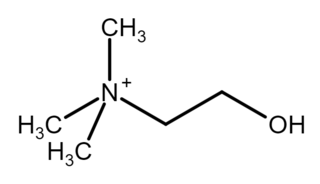
Choline is a cation with the chemical formula [(CH3)3NCH2CH2OH]+. Choline forms various salts, for example choline chloride and choline bitartrate.

The human microbiome is the aggregate of all microbiota that reside on or within human tissues and biofluids along with the corresponding anatomical sites in which they reside, including the skin, mammary glands, seminal fluid, uterus, ovarian follicles, lung, saliva, oral mucosa, conjunctiva, biliary tract, and gastrointestinal tract. Types of human microbiota include bacteria, archaea, fungi, protists, and viruses. Though micro-animals can also live on the human body, they are typically excluded from this definition. In the context of genomics, the term human microbiome is sometimes used to refer to the collective genomes of resident microorganisms; however, the term human metagenome has the same meaning.

A transferase is any one of a class of enzymes that catalyse the transfer of specific functional groups from one molecule to another. They are involved in hundreds of different biochemical pathways throughout biology, and are integral to some of life's most important processes.
Trimethylamine (TMA) is an organic compound with the formula N(CH3)3. It is a colorless, hygroscopic, and flammable tertiary amine. It is a gas at room temperature but is usually sold as a 40% solution in water. (It is also sold in pressurized gas cylinders.) TMA is a nitrogenous base and can be readily protonated to give the trimethylammonium cation. Trimethylammonium chloride is a hygroscopic colorless solid prepared from hydrochloric acid. Trimethylamine is a good nucleophile, and this reaction is the basis of most of its applications. TMA is widely used in industry: it is used in the synthesis of choline, tetramethylammonium hydroxide, plant growth regulators or herbicides, strongly basic anion exchange resins, dye leveling agents, and a number of basic dyes. At higher concentrations it has an ammonia-like odor, and can cause necrosis of mucous membranes on contact. At lower concentrations, it has a "fishy" odor, the odor associated with rotting fish.

Gut microbiota, gut microbiome, or gut flora, are the microorganisms, including bacteria, archaea, fungi, and viruses, that live in the digestive tracts of animals. The gastrointestinal metagenome is the aggregate of all the genomes of the gut microbiota. The gut is the main location of the human microbiome. The gut microbiota has broad impacts, including effects on colonization, resistance to pathogens, maintaining the intestinal epithelium, metabolizing dietary and pharmaceutical compounds, controlling immune function, and even behavior through the gut–brain axis.
Joanna Sigfred Fowler is a scientist emeritus at the U.S. Department of Energy's Brookhaven National Laboratory in New York. She served as professor of psychiatry at Mount Sinai School of Medicine and director of Brookhaven's Radiotracer Chemistry, Instrumentation and Biological Imaging Program. Fowler studied the effect of disease, drugs, and aging on the human brain and radiotracers in brain chemistry. She has received many awards for her pioneering work, including the National Medal of Science.
Jeffrey I. Gordon is a biologist and the Dr. Robert J. Glaser Distinguished University Professor and Director of the Center for Genome Sciences and Systems Biology at Washington University in St. Louis. He is internationally known for his research on gastrointestinal development and how gut microbial communities affect normal intestinal function, shape various aspects of human physiology including our nutritional status, and affect predisposition to diseases. He is a member of the National Academy of Sciences, the American Academy of Arts and Sciences, the Institute of Medicine of the National Academies, and the American Philosophical Society.

The Human Microbiome Project (HMP) was a United States National Institutes of Health (NIH) research initiative to improve understanding of the microbiota involved in human health and disease. Launched in 2007, the first phase (HMP1) focused on identifying and characterizing human microbiota. The second phase, known as the Integrative Human Microbiome Project (iHMP) launched in 2014 with the aim of generating resources to characterize the microbiome and elucidating the roles of microbes in health and disease states. The program received $170 million in funding by the NIH Common Fund from 2007 to 2016.

A microbiome is the community of microorganisms that can usually be found living together in any given habitat. It was defined more precisely in 1988 by Whipps et al. as "a characteristic microbial community occupying a reasonably well-defined habitat which has distinct physio-chemical properties. The term thus not only refers to the microorganisms involved but also encompasses their theatre of activity". In 2020, an international panel of experts published the outcome of their discussions on the definition of the microbiome. They proposed a definition of the microbiome based on a revival of the "compact, clear, and comprehensive description of the term" as originally provided by Whipps et al., but supplemented with two explanatory paragraphs. The first explanatory paragraph pronounces the dynamic character of the microbiome, and the second explanatory paragraph clearly separates the term microbiota from the term microbiome.
B. Brett Finlay, is a Canadian microbiologist well known for his contributions to understanding how microbes cause disease in people and developing new tools for fighting infections, as well as the role the microbiota plays in human health and disease. Science.ca describes him as one of the world's foremost experts on the molecular understanding of the ways bacteria infect their hosts. He also led the SARS Accelerated Vaccine Initiative (SAVI) and developed vaccines to SARS and a bovine vaccine to E. coli O157:H7. His current research interests focus on pathogenic E. coli and Salmonella pathogenicity, and the role of the microbiota in infections, asthma, and malnutrition. He is currently the UBC Peter Wall Distinguished Professor and a Professor in the Michael Smith Laboratories, Microbiology and Immunology, and Biochemistry and Molecular Biology, and Co-director and Senior Fellow for the CIFAR Humans and Microbes program. He is also co-author of the book Let Them Eat Dirt: Saving Your Child from an Oversanitized World and The Whole-Body Microbiome: How to Harness Microbes - Inside and Out - For Lifelong Health. Finlay is the author of over 500 publications in peer-reviewed journals and served as editor of several professional publications for many years.
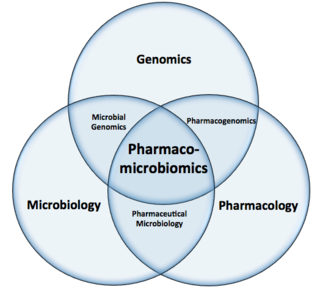
Pharmacomicrobiomics, proposed by Prof. Marco Candela for the ERC-2009-StG project call, and publicly coined for the first time in 2010 by Rizkallah et al., is defined as the effect of microbiome variations on drug disposition, action, and toxicity. Pharmacomicrobiomics is concerned with the interaction between xenobiotics, or foreign compounds, and the gut microbiome. It is estimated that over 100 trillion prokaryotes representing more than 1000 species reside in the gut. Within the gut, microbes help modulate developmental, immunological and nutrition host functions. The aggregate genome of microbes extends the metabolic capabilities of humans, allowing them to capture nutrients from diverse sources. Namely, through the secretion of enzymes that assist in the metabolism of chemicals foreign to the body, modification of liver and intestinal enzymes, and modulation of the expression of human metabolic genes, microbes can significantly impact the ingestion of xenobiotics.
Alison Butler is a Distinguished Professor in the Department of Chemistry and Biochemistry at the University of California, Santa Barbara. She works on bioinorganic chemistry and metallobiochemistry. She is a Fellow of the American Association for the Advancement of Science (1997), the American Chemical Society (2012), the American Academy of Arts and Sciences (2019), and the Royal Society of Chemistry (2019). She was elected a member of the National Academy of Sciences in 2022.
Elizabeth S. Sattely is an American scientist and biotechnology engineer. She is an Associate Professor of Chemical Engineering in the Department of Chemical Engineering, an HHMI investigator, and a ChEM-H Faculty Fellow at Stanford University.

Janelle S. Ayres is an American immunologist and microbiologist, member of the NOMIS Center for Immunobiology and Microbial Pathogenesis and Helen McLoraine Developmental Chair at the Salk Institute for Biological Sciences. Her research focuses on the relation of host-pathogen interactions with the microbiome.
Emily A. Weiss is the Mark and Nancy Ratner Professor of Chemistry and Director of the Photo-Sciences Research Center at Northwestern University. Her research considers the optical and electronic properties of nanostructures, including hybrid organic–inorganic quantum dots. She was a two-time finalist in the Blavatnik Awards for Young Scientists.

Peter J. Turnbaugh is a microbiologist and a professor at University of California, San Francisco. He is known for his research on the metabolic activities performed by the trillions of microbes that colonize humans' adult bodies. Turnbaugh and his research group use interdisciplinary approaches in preclinical models and human cohorts to study the mechanisms through which the gut microbiome influences nutrition and pharmacology.
Catalina Cuellar Gempeler is a Colombian microbial ecologist and marine microbiologist, currently teaching and doing research at Cal Poly Humboldt. Her research focuses mainly on understanding microbial meta-community, eco-evolutionary dynamics, and ecosystem dynamics. The Catalina Cuellar-Gempeler lab is currently focused on studying the interactions between hosts and their microbial communities. The lab's main emphasis is on the microbes used in digestion in the Californian and Eastern, carnivorous pitcher plants. In March 2021, Cuellar-Gempeler was awarded an Early Career grant for $1 million by the National Science Foundation.
Lesley Hoyles is a Welsh microbiologist who is Professor of Microbiome and Systems Biology at Nottingham Trent University. She combines in vivo and in vitro microbiology and bioinformatics research to better understand how the gut microbiota influences health and disease.

Elaine Yih-Nien Hsiao is an American biologist who is Professor in Biological Sciences at University of California, Los Angeles. Her research considers the microbes that impact human health. She was a 2022 Laureate for the Blavatnik Awards for Young Scientists.










